The use of antiplatelet and anticoagulant drugs is common in patients undergoing elective endoscopic procedures. To minimize both bleeding and thromboembolic risks, an individualized assessment is essential. For this purpose, the Societat Catalana de Digestologia (SCD) commissioned a consensus document aimed at providing practical recommendations based on the best available evidence and expert agreement.
Material and methodsA panel of 10 specialists from the fields of Gastroenterology, Cardiology, Hematology, and Anesthesiology were involved in the development of the document. A non-systematic review of the literature was conducted using a modified Delphi method through online voting. Recommendations were reviewed and discussed during each round until consensus was reached. Recommendations with ≥80% agreement were accepted.
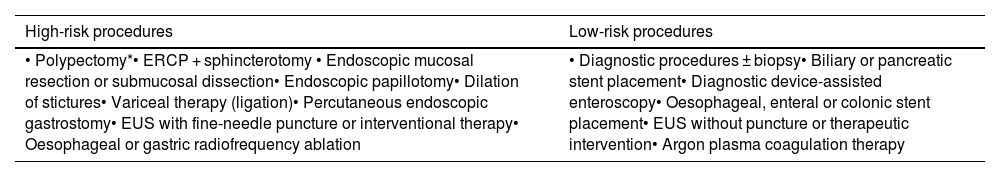
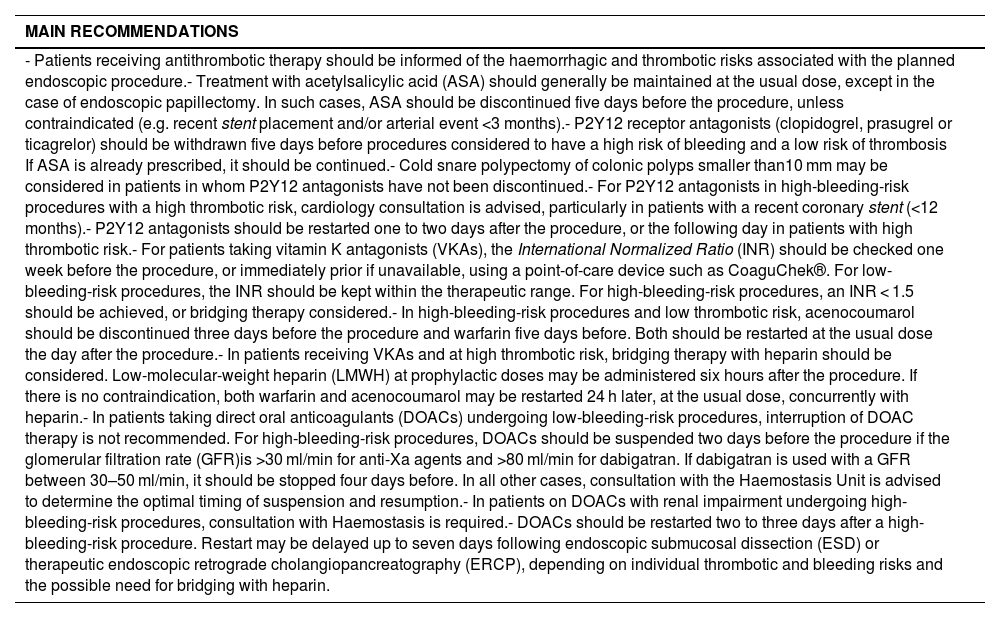
ResultsA total of 16 endoscopic procedures were assessed based on their hemorrhagic risk, and tailored management guidelines were defined to different groups of antithrombotic agents. The document includes a management algorithm and stratification tables for bleeding and thrombotic risk.
ConclusionThe recommendations developed by the SCD integrate international evidence and are adapted to the local healthcare context, providing a valuable tool for clinical practice. The importance of multidisciplinary coordination and effective communication with the patient is emphasized to ensure safety throughout the periprocedural period.
El uso de fármacos antiagregantes y anticoagulantes es habitual en pacientes que se realizan un procedimiento endoscópico electivo. Para minimizar el riesgo tanto de hemorragia como de trombosis, es fundamental ofrecer una evaluación individualizada. Para ello, la Societat Catalana de Digestología (SCD) encargó un documento de consenso con el objetivo de ofrecer unas recomendaciones prácticas basadas en la mejor evidencia disponible y en consenso de expertos.
Material y métodosDiez especialistas en Aparato Digestivo, Cardiología, Hematología y Anestesiología participaron en la elaboración del documento. Se llevó a cabo una revisión no sistemática de la literatura y se aplicó el método Delphi modificado con votación telemática. Las recomendaciones se revisaron y comentaron en cada ronda para llegar a un consenso, aceptándose aquellas que alcanzaron un nivel de acuerdo del 80 %.
ResultadosSe evaluaron 16 procedimientos endoscópicos según su riesgo hemorrágico, y se definieron pautas específicas de manejo adaptadas a los distintos grupos de fármacos antitrombóticos. El documento incluye un algoritmo de manejo y tablas de estratificación de riesgo hemorrágico y trombótico.
ConclusiónLas recomendaciones de la SCD integran la evidencia internacional y se adaptan a nuestro entorno sanitario, ofreciendo una herramienta útil para la práctica clínica. Se resalta la necesidad de coordinación multidisciplinaria y de una buena comunicación con el paciente para garantizar la seguridad durante el periodo periprocedimiento.