To assess the efficacy of a central venous catheter adhesive fixation device (CVC) to prevent associated complications. To establish the need for dressing changes, number of days’ catheterization and reasons for catheter removal in both study groups. To assess the degree of satisfaction of personnel with the adhesive system.
MethodA, randomised, prospective and open pilot study, of parallel groups, with comparative evaluation between CVC fixation with suture and with an adhesive safety system. The study was performed in the Coronary Unit of the Universitari de Bellvitge Hospital, between April and November 2016. The population studied were patients with a CVC. The results were analysed using SPSS Statistics software. The study was approved by the Clinical Research Ethics Committee.
Results100 patients (47 adhesive system and 53 suture) were analysed. Both groups were homogeneous in terms of demographic variables, anticoagulation and days of catheterization. The frequency of complications in the adhesive system group was 21.3%, while in the suture group it was 47.2% (p=.01). The suture group had a higher frequency of local signs of infection (p=.006), catheter displacement (p=.005), and catheter-associated bacteraemia (p=.05). The use of adhesive fixation was associated with a lower requirement for dressing changes due to bleeding (p=.006). Ninety-six point seven percent of the staff recommended using the adhesive safety system.
ConclusionsThe catheters fixed with adhesive systems had fewer infectious complications and less displacement.
El objetivo principal fue valorar la eficacia de un dispositivo de fijación adhesiva de catéter venoso central (CVC) para prevenir complicaciones asociadas. Los objetivos secundarios fueron: determinar la frecuencia de cambios de apósito, días de cateterización y motivos de retirada de los catéteres en ambos grupos del estudio, y evaluar el grado de satisfacción del personal en base al uso del sistema adhesivo.
MétodoEstudio piloto, aleatorizado, prospectivo y abierto, de grupos paralelos realizándose una evaluación comparativa entre fijación de CVC con sutura y un sistema de seguridad adhesivo. El estudio se realizó en la Unidad Coronaria del Hospital Universitari de Bellvitge, entre abril y noviembre de 2016. La población estudiada fueron aquellos pacientes portadores de CVC. Los resultados se analizaron mediante SPSS 18.0. El estudio fue aprobado por el Comité Ético de Investigación Clínica.
ResultadosFueron analizados 100 pacientes (47 con sistema adhesivo y 53 con sutura). Ambos grupos fueron homogéneos en cuanto a variables demográficas, anticoagulación y días de cateterización. La frecuencia de aparición de complicaciones en el sistema adhesivo fue del 21,3%, mientras que con la sutura fue del 47,2% (p=0,01). El grupo sutura presentó mayor frecuencia de signos locales de infección (p=0,006), desplazamiento del catéter (p=0,005) y bacteriemias asociadas a catéter (p=0,05). El uso de la fijación adhesiva se asoció a menor requerimiento de cambios de apósito por sangrado (p=0,006). El 96,7% del personal recomendó la utilización del sistema de seguridad adhesivo.
ConclusionesLos catéteres fijados con sistema adhesivo presentaron menos complicaciones infecciosas y grado de desplazamiento.
The clinical practice guideline on vascular care, Disease Control and Prevention, recommends, with a Category II level of evidence, the use of suture-free devices to reduce the risk of infection by intravascular catheters. Central venous catheter (CVC) securement is considered a measure to ensure stability of the catheter, and reduce the risk of phlebitis and associated infections.
What does this paper contribute?The results of our study highlight the advantages of CVC securement using an adhesive system compared to a suture, reducing the onset of catheter-related infections and degree of displacement of the catheter. They also show that using an adhesive fixation system is less gruelling for the patient, avoids the risk of bleeding associated with the suture and consequently requires fewer dressing changes.
Implications of the studyThe use of a CVC fixation system reduces infectious complications, ensuring stability of the catheters and promoting better wellbeing for patients.
Central venous catheterisation is an invasive technique that comprises the insertion of a catheter into the patient's intravascular space, the distal end of which is placed in the lower third portion of the superior vena cava or just at the inlet of the right atrium.1
Its principal indications are: the measurement of central venous pressure, administration of vasoactive or high osmolarity drugs, long-term antibiotherapy, total parenteral nutrition, chemotherapy, haemodialysis, plasmapheresis, endocavitary pacemaker implantation or when peripheral venous access is not possible.
The technique is gradually being used more often in hospitals due to increased age, comorbidity and severity in hospitalised patients, especially in critical care units. According to the national data from the study programme of the prevalence of nosocomial infections in Spain, around 7% of patients admitted to hospital have a central venous catheter (CVC).2
An increased onset of secondary complications has been associated with the use of CVC, occurring in more than 15% of admitted patients.3 The most common complications from CVC are divided into two large groups: mechanical and infectious.
There are associated risk factors that favour the onset of both types of complications: operator-dependent factors (experience and insertion technique), patient-dependent factors (age, weight, comorbidity or associated treatment) and catheter-dependent factors (material, type, diameter, number of lumens and dwell time).4–6 Moreover, insertion of the catheter itself can cause mechanical complications such as pneumothorax, arterial puncture or catheterisation.7 The access route is a clear conditioning factor for infectious complications from CVC. Therefore, catheters inserted in the femoral or jugular vein have a greater risk of colonisation and infection than those in the subclavian vein or the arm.8
Infectious complications depend to a large extent on the care and maintenance of the catheter; of note are local infection at the point of insertion and the sutures, phlebitis, and vascular catheter-related bacteraemia (VCRB).
There are different mechanisms that can lead to contamination of a CVC. Extraluminal colonisation due to migration of the skin flora through the fibrin sheath around the catheter is most common in catheters that have been in situ for less than 8 days. Handling causes direct contamination of the catheter by endoluminal colonisation through the external connections. There are also cases where the catheters can be haematologically colonised by a different focus of infection, and therefore have to be removed.9,10
The incidence of infectious complications in CVC varies between 5% and 26%.3,7 The frequency of VCRB is gradually increasing due to extensive use, prolonged duration of CVC, increased hospital stays, and patient morbidity.
It is currently calculated that between 15% and 30% of all nosocomial bacteraemias are related to the use of percutaneous intravascular devices.11,12 In critical care units this type of infection has been associated with high morbidity and attributable mortality, increased hospital stay, and very relevant added health costs.
Incorrect securement of the catheter is another very important aspect that can result in one of these pathogenic complications. This can cause the catheter to move inside and outside the intravascular space through the insertion point.13
The clinical practice guidelines recommend, with a Category II level of evidence, the use of suture-free securement devices to reduce the risk of infection by intravascular catheters.6 Stabilising the CVC is recognised as a measure that can reduce the risk of phlebitis, potential migration and/or accidental removal of the catheter, and can also help prevent infections.
Patients admitted to coronary units usually require anticoagulant treatment due to the underlying disease itself (ischaemic heart disease, valvulopathies, myocardopathies, etc.) as well as associated medical treatment (ventricular assist devices, pacemakers, haemodiafiltration, etc.). Both anticoagulation and the skin puncture itself to fix the catheter are factors that predispose to bleeding through the suture site. Using an adhesive securement system prevents secondary bleeding from the puncture for the suture.
Following the recommendations of the clinical practice guidelines, and bearing in mind the good results of the latest studies published on the use of sutureless CVC securement systems,14,15 we decided to undertake a research study in our unit with the principal aim of evaluating the efficacy of an adhesive CVC securement device in preventing the onset of associated complications. Our secondary aims were to determine the frequency of dressing changes, days of catheterisation and reason for removing the catheters in both of the study groups, and to assess the level of staff satisfaction with the adhesive system.
MethodologyStudy design and scopeA pilot study performed in the Coronary Care Unit of the Bellvitge University Hospital (HUB), an adult tertiary hospital of the Catalan Health Institute. The data were collected between April and November, 2016. The study was approved by the hospital's Clinical Research Ethics Committee.
A randomised, prospective and open, parallel-group clinical trial was performed of a healthcare product, with a comparative evaluation between the CVC fixation method using sutures and an adhesive safety system.
The sutureless fixation system used in this study was 3M Tegaderm CVC/PICC™ (Ref: 1837-2100/1839-2100), comprising a silicone adhesive device together with a reinforced transparent Tegaderm dressing.
SubjectsAll the patients admitted to the Coronary Care Unit who required the insertion of a CVC were included. The catheters used in the study were Teleflex-Arrow® 3-lumen CVC and 7 French.
The exclusion criteria were:
- 1.
Patients with congenital haematological diseases (haemophilia, thrombocytopenia, polycythaemia, etc.).
- 2.
CVC not inserted in the unit itself.
- 3.
CVC with a dwell time of less than 48h.
- 4.
Patients with a CVC transferred to hospitals or areas of the HUB, other than the Cardiology area.
In order to guarantee that they were familiar and able to place the new adhesive system correctly, theoretical-practical workshops were organised for all the unit staff. A pilot test was then performed with patients over a period of 15 days. The pilot test enabled the data collection sheet to be redesigned to make it more agile and intuitive to complete.
The patients that met the inclusion criteria were randomised using random allocation sequence software (RndSeq-Statistical Package for the Social Sciences [SPSS]).
VariablesThe independent variable of the study was the catheter fixation method used. The dependent study variables were the presence of a complication associated with the catheter, defined as the presence of signs of infection, phlebitis, VCRB, migration or complete accidental removal. The remaining variables were: sex, age, BMI, reason for admission, Charlson index, anticoagulation, level of consciousness and pain scale of the patient during insertion of the CVC, the patient's level of mobility during the dwell time of the CVC, catheter dwell time in days, number and reason for dressing changes, skin integrity after removing the adhesive device, accidental punctures by the professional during fixation of the CVC, reason for removing the CVC.
Data collectionIf a patient required CVC catheterisation and met the inclusion criteria they were asked to participate in the study. Once they had signed their informed consent, they were randomised to either the suture fixation system or the adhesive system.
A data collection sheet was then completed (Appendix A) with in situ evaluation of other variables relating to the CVC catheterisation itself, such as the insertion site (humeral, subclavian, jugular or femoral); centimetres of CVC inserted measured with a measuring tape (from the puncture site to the suture or the adhesive device), and whether or not ultrasound had been used to guide insertion of the catheter. Pain was measured exclusively in conscious patients using the visual analogue scale (VAS, from 0 to 10).
The healthcare professional decided the access route and whether to use ultrasound for catheterisation according to their own experience and the anatomy and clinical situation of the patient. During the dwell time of the catheter, the nurse responsible for the patient assessed the stability of the catheter once per shift (degree of displacement in centimetres), checked for local signs of infection at the insertion site or suture site (erythema, pain and purulent drainage) and whether a dressing change was necessary for any of the following reasons: due to the protocol (7 days after placement), due to blood staining, due to loss of dressing adherence, or to replace the catheter after chest X-ray. The degree of displacement was measured using a measuring tape after inserting the catheter and then measured again each time that the dressing was changed or the catheter removed. The distance was measured in centimetres from the insertion site of the CVC to the place of securement, either by suture or adhesive device.
The data collection sheet was fully completed once the CVC had been removed, either because the treatment had been completed, due to complete accidental removal, insertion or suture site infection, phlebitis, fever of unknown origin with suspected infection of the catheter, pericatheter bleeding, or the death of the patient. In the group using the adhesive safety system, skin integrity was also assessed when the CVC was finally removed using the Norton ulcer assessment scale (from 0 to 3 grades, grade 0 being no injury; grade I redness, grade II redness and blistering; and grade III the presence of an ulcer).
A CVC tip culture was taken from those removed due to high suspicion of infection, as well as blood cultures. When the data collection was complete, the satisfaction of the staff who participated in the study was measured using a questionnaire, evaluating aspects such as the ease of placement and removal of the different fixation types, and the comfort and safety for the patient using a Likert-type assessment scale (scoring from 0 to 5 points, 0 being the lowest grade of ease, safety or comfort and 5 the highest).
Data analysisBecause this was a pilot study, we did not calculate the sample size. The results were studied using SPSS (version 18.0, SPSS Inc., Chicago, IL). Once the data had been collected a descriptive analysis was performed of the recruited sample using the mean, median, percentiles 25% and 75%, and minimum and maximum values.
The chi-squared test was use to detect significant differences between each group or Fisher's exact test for categorical variables, and the Student's t-test or U Mann–Whitney test for continuous variables, depending on the results of the Kolmogorov–Smirnov normality test. The results were presented as 95% confidence interval (CI). Statistical significance was set at a bilateral p<.05.
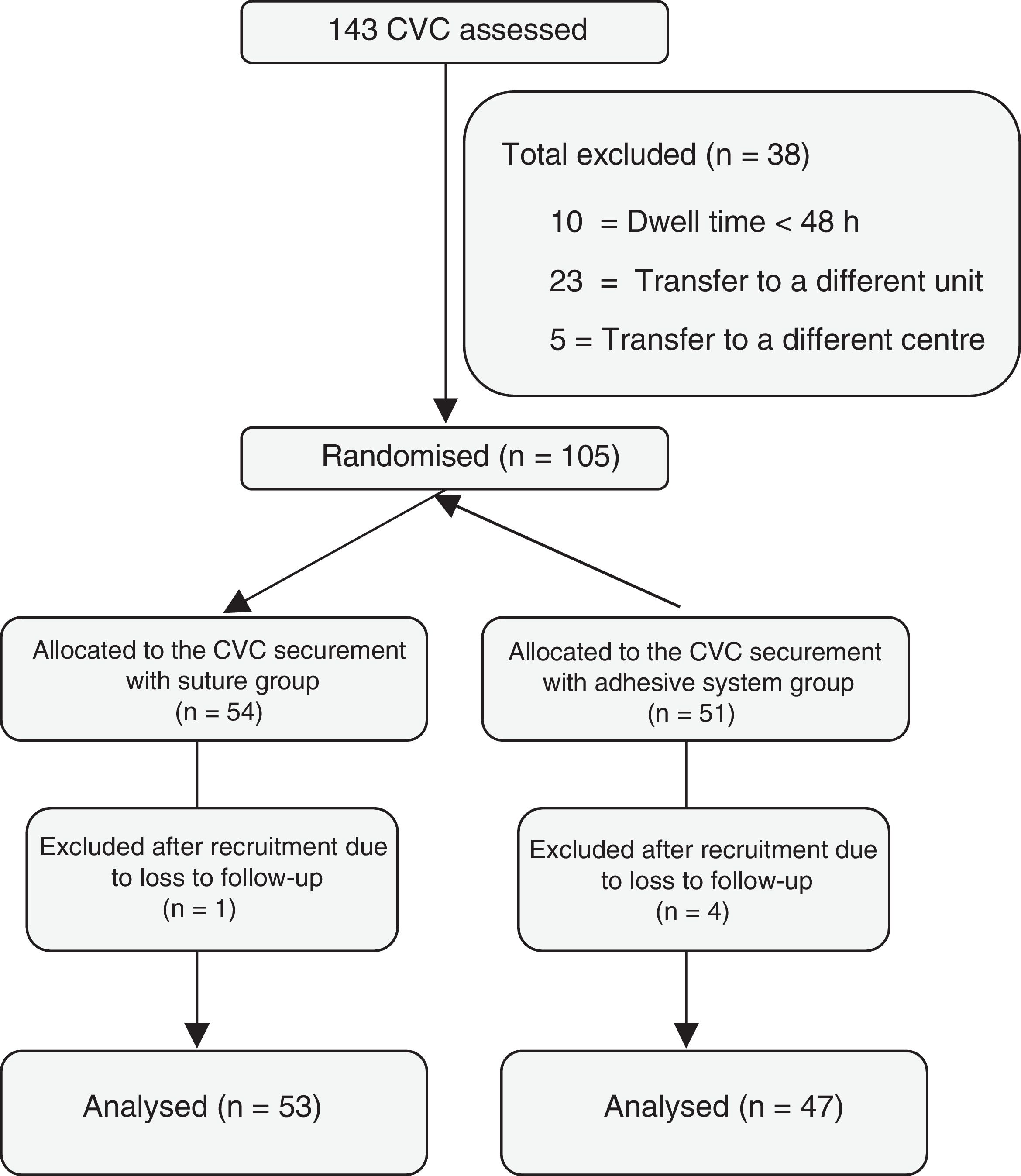
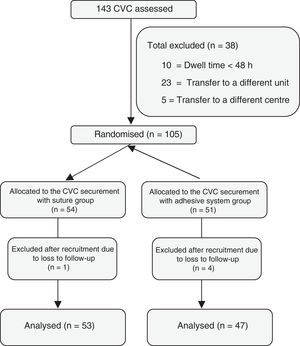
ResultsOne hundred and forty three patients inserted with a CVC were assessed, of whom 38 were excluded. Of the 105 randomised patients, 51 were allocated to the adhesive safety system, and 54 to the suture fixation system. Eventually, after applying the exclusion criteria, a total of 100 patients were studied (47 with the adhesive fixation system, and 53 with the suture fixation system) (Fig. 1).
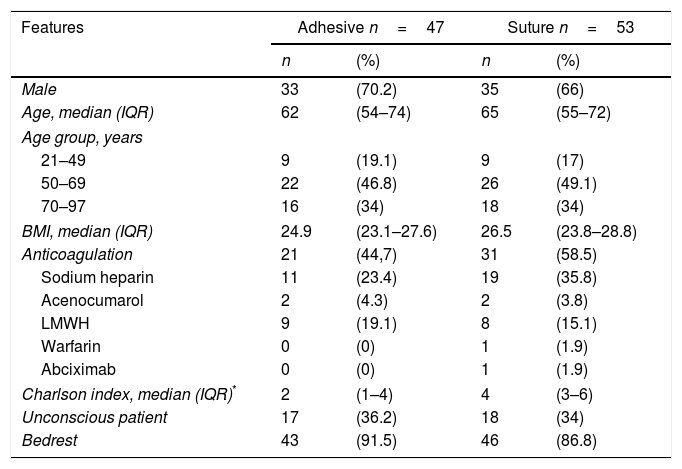
The features of the patients were similar in both of the study groups. There were no significant differences in terms of age, sex or BMI. There were no significant differences in terms of state of consciousness or mobility either. The most commonly used anticoagulation in both groups was sodium heparin, and no significant differences were detected. However, the Charlson index was highest in the suture fixation group (p<.05) (Table 1).
Patient features.
| Features | Adhesive n=47 | Suture n=53 | ||
|---|---|---|---|---|
| n | (%) | n | (%) | |
| Male | 33 | (70.2) | 35 | (66) |
| Age, median (IQR) | 62 | (54–74) | 65 | (55–72) |
| Age group, years | ||||
| 21–49 | 9 | (19.1) | 9 | (17) |
| 50–69 | 22 | (46.8) | 26 | (49.1) |
| 70–97 | 16 | (34) | 18 | (34) |
| BMI, median (IQR) | 24.9 | (23.1–27.6) | 26.5 | (23.8–28.8) |
| Anticoagulation | 21 | (44,7) | 31 | (58.5) |
| Sodium heparin | 11 | (23.4) | 19 | (35.8) |
| Acenocumarol | 2 | (4.3) | 2 | (3.8) |
| LMWH | 9 | (19.1) | 8 | (15.1) |
| Warfarin | 0 | (0) | 1 | (1.9) |
| Abciximab | 0 | (0) | 1 | (1.9) |
| Charlson index, median (IQR)* | 2 | (1–4) | 4 | (3–6) |
| Unconscious patient | 17 | (36.2) | 18 | (34) |
| Bedrest | 43 | (91.5) | 46 | (86.8) |
LMWH: low-molecular-weight heparin; IQR: interquartile range.
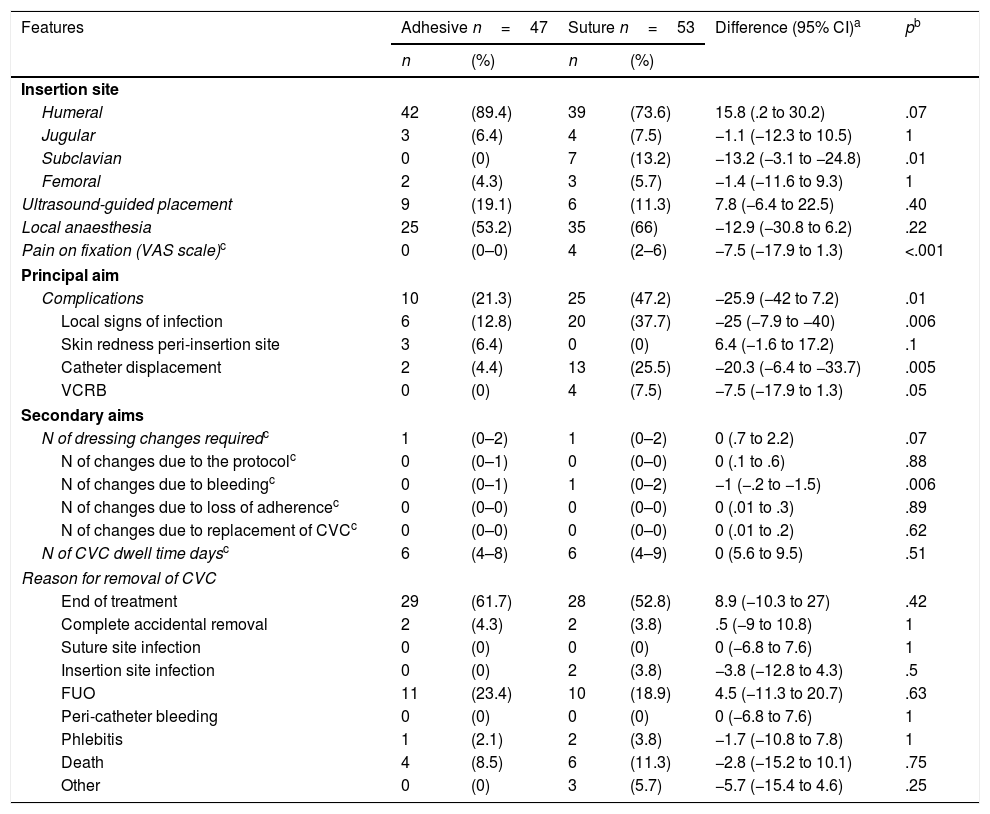
Table 2 details the results of the study. Analysis of our principal aim shows that the frequency of onset of complications with the adhesive safety system was 21.3%, whereas it was 47.2% with the suture fixation system.
Results according to study group.
| Features | Adhesive n=47 | Suture n=53 | Difference (95% CI)a | pb | ||
|---|---|---|---|---|---|---|
| n | (%) | n | (%) | |||
| Insertion site | ||||||
| Humeral | 42 | (89.4) | 39 | (73.6) | 15.8 (.2 to 30.2) | .07 |
| Jugular | 3 | (6.4) | 4 | (7.5) | −1.1 (−12.3 to 10.5) | 1 |
| Subclavian | 0 | (0) | 7 | (13.2) | −13.2 (−3.1 to −24.8) | .01 |
| Femoral | 2 | (4.3) | 3 | (5.7) | −1.4 (−11.6 to 9.3) | 1 |
| Ultrasound-guided placement | 9 | (19.1) | 6 | (11.3) | 7.8 (−6.4 to 22.5) | .40 |
| Local anaesthesia | 25 | (53.2) | 35 | (66) | −12.9 (−30.8 to 6.2) | .22 |
| Pain on fixation (VAS scale)c | 0 | (0–0) | 4 | (2–6) | −7.5 (−17.9 to 1.3) | <.001 |
| Principal aim | ||||||
| Complications | 10 | (21.3) | 25 | (47.2) | −25.9 (−42 to 7.2) | .01 |
| Local signs of infection | 6 | (12.8) | 20 | (37.7) | −25 (−7.9 to −40) | .006 |
| Skin redness peri-insertion site | 3 | (6.4) | 0 | (0) | 6.4 (−1.6 to 17.2) | .1 |
| Catheter displacement | 2 | (4.4) | 13 | (25.5) | −20.3 (−6.4 to −33.7) | .005 |
| VCRB | 0 | (0) | 4 | (7.5) | −7.5 (−17.9 to 1.3) | .05 |
| Secondary aims | ||||||
| N of dressing changes requiredc | 1 | (0–2) | 1 | (0–2) | 0 (.7 to 2.2) | .07 |
| N of changes due to the protocolc | 0 | (0–1) | 0 | (0–0) | 0 (.1 to .6) | .88 |
| N of changes due to bleedingc | 0 | (0–1) | 1 | (0–2) | −1 (−.2 to −1.5) | .006 |
| N of changes due to loss of adherencec | 0 | (0–0) | 0 | (0–0) | 0 (.01 to .3) | .89 |
| N of changes due to replacement of CVCc | 0 | (0–0) | 0 | (0–0) | 0 (.01 to .2) | .62 |
| N of CVC dwell time daysc | 6 | (4–8) | 6 | (4–9) | 0 (5.6 to 9.5) | .51 |
| Reason for removal of CVC | ||||||
| End of treatment | 29 | (61.7) | 28 | (52.8) | 8.9 (−10.3 to 27) | .42 |
| Complete accidental removal | 2 | (4.3) | 2 | (3.8) | .5 (−9 to 10.8) | 1 |
| Suture site infection | 0 | (0) | 0 | (0) | 0 (−6.8 to 7.6) | 1 |
| Insertion site infection | 0 | (0) | 2 | (3.8) | −3.8 (−12.8 to 4.3) | .5 |
| FUO | 11 | (23.4) | 10 | (18.9) | 4.5 (−11.3 to 20.7) | .63 |
| Peri-catheter bleeding | 0 | (0) | 0 | (0) | 0 (−6.8 to 7.6) | 1 |
| Phlebitis | 1 | (2.1) | 2 | (3.8) | −1.7 (−10.8 to 7.8) | 1 |
| Death | 4 | (8.5) | 6 | (11.3) | −2.8 (−15.2 to 10.1) | .75 |
| Other | 0 | (0) | 3 | (5.7) | −5.7 (−15.4 to 4.6) | .25 |
VCRB: vascular catheter-related bacteraemia; CVC: central venous catheter; VAS: visual analogue scale numerically graduated to assess pain intensity; FUO: fever of unknown origin; CI: confidence interval.
Comparing the adhesive safety system group with the suture fixation group, the suture fixation group most often presented signs of local infection (12.8% vs 37.7%, p=.006), catheter displacement (4.4% vs 25.5%, p=.005, with a mean of .14cm vs 1.5cm) and VCRB (0% vs 7.5%, p=.05).
No significant differences were observed with regard to the onset of phlebitis or peri-insertion skin redness.
With regard to the secondary study aims, the number of dressing changes was similar in both groups, although there were more dressing changes due to bleeding in the suture fixation group (range=0–3 vs 0–7). We detected no significant differences in either group in terms of CVC dwell time days or reason for removal.
The principal insertion route of the CVC was humeral (89.4% adhesive safety system vs 73.6% suture fixation system). There were no significant differences with regard to the humeral, jugular and femoral insertion routes. More CVC were inserted via the subclavian routes in the patients with the suture fixation system (p=.01). No significant differences were detected with regard to ultrasound guidance of CVC insertion or the use of local anaesthesia, a low frequency of ultrasound use was observed in both groups (19.1% vs 11.3%). However, the patients with the suture fixation system reported more pain (VAS scale) on securing the CVC (p<.001).
Despite the strong adherence to the skin of the securement devices, practically no skin integrity alterations were documented after its removal. Only 3 cases of skin redness were detected (grade I injury, Norton scale) after removing the adhesive safety system, which did not require any further intervention.
Of the 4 patients with VCRB, 2 had a jugular CVC, one humeral, and the other subclavian. The mean dwell time of these CVC was 10 days (range=6–20 days), and the microorganisms detected were: Staphylococcus coagulase-negative (SCN) (n=2), Serratia marcescens (n=1) and Candida albicans (n=1).
An analysis was also performed on the patients with a high Charlson index (>3 points). With regard to the main variable of the study, no significant differences were detected in the onset of complications between the patients with a low or high Charlson index (34% adhesive safety system vs 32% suture fixation system, p=1).
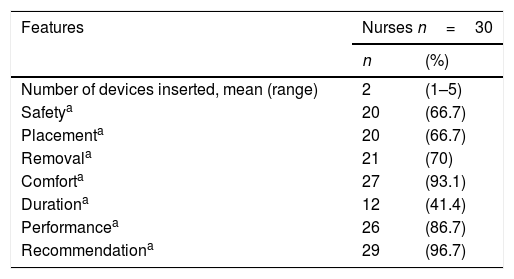
An ad-hoc survey was later designed to assess the level of satisfaction of the staff that used the adhesive safety system. Thirty healthcare professionals used this system (mean=2, range=1–5 systems) (Table 3). Comfort and performance were the most highly assessed characteristics (>85% scored these as superior or very superior), and the lowest score was awarded to the duration of the dressing (41.4% scored this as superior or very superior). Finally, 96.7% of the respondents recommended using the adhesive safety system.
DiscussionThe adhesive securement device is a method of CVC fixation that is much less aggressive for the patient and reduces bleeding through the puncture site. This intervention is of particular interest for critical heart patients with high comorbidity, who are routinely given anticoagulant treatment, because fewer dressing changes are required and there is consequently less handling of the catheters. All of this in turn can result in a reduction in the bacterial colonisation level of the catheter and the insertion sites, as demonstrated in the study by Timsit et al., 16 where they randomised 1.419 patients with 296 catheters that were colonised, 29 major catheter-related infections, and 23 VCRB. Of the 11,036 dressing changes, 7347 (67%) were performed before the anticipated date due to staining or loss of adherence. In the study by Timsit et al. the number of dressing changes directly related to a greater risk of colonisation of the skin around the catheter on removal (p<.001).
The study by Yamamoto et al.15 compared fixation of peripherally inserted catheters (PICC) with a suture fixation system (n=85) to a StatLock securement device (n=85) concluding that catheter-related infections could be significantly reduced with the latter fixation method (p<.032). The results of our study coincide with the literature, in that the patients using the suture fixation system (control group) were more prone to local signs of infection and required more frequent dressing changes due to blood staining.
The study by Crnich and Maki14 assessed the onset of infectious complications in peripherally inserted catheters between a suture fixation system and an adhesive method. The VCRB were significantly fewer in the group of patients using the adhesive fixation device (p<.01). The study concluded that the great potential of the adhesive securement system lies in reducing infections by eliminating the purulent wounds created by skin sutures, which are contiguous with the recently inserted catheter, and in reducing the micro-movements of the catheter. Although the patients included in both of our study groups were homogeneous in terms of level of consciousness and level of mobility (bed rest vs seated), the catheters fixed using the adhesive method were less prone to displacement, and the migration distance was also less.
The access route is directly related to catheter migration and infection. CVC inserted via the jugular and femoral veins are more likely to displace due to movements of the neck or leg and to become infected due to the high level of local humidity or bacterial contamination from inguinal skin.17
The evidence-based recommendations for vascular access care advocate inserting the CVC in the subclavian vein rather than the jugular or femoral veins to minimise the risk of infection (CDC 2011, level of evidence Ib).6 A minimum risk of infectious complications and very good patient comfort are associated with PICC.18 The catheters used in both our study groups were principally inserted peripherally via one of the veins in the arm (basilic, cephalic or brachial). This is because this is the route of first choice, and nurses perform the procedure. Furthermore, the clinical practice guidelines suggest peripheral access since there is no risk of pneumothorax, and the risk of haemorrhage is very low. Studies such as that by Polderman and Girbes,17 show that the risk of bacteraemia with PICC is very low compared to CVC via other access routes.
Dwell time is another factor that might influence the onset of catheter-related infection, there is a risk of 3–5% from the third day which increases to 5–10% from the seventh day.18 In our study there were no differences between the groups in terms of CVC dwell time, with a mean of 6 days. The infection route of a catheter directly relates to the day of onset, extraluminal infections are associated with those detected before the eighth day after insertion of the catheter.19 Bearing these data in mind, 3 of the 4 VCRB detected in our study might suggest that the entry route was extraluminal, which is closely related to the handling of the catheters during dressing changes.
According to the financial report of the VINCat programme for 2015, VCRB infections result in the highest costs, estimated at a mean 31.381€ per infection.
In recent years, rates of VCRB have reduced progressively, from 2.00 per 1000 stay days in 2008 to 1.10 per 1000 in 2013, particularly due to the implementation of the Bacteraemia Zero Project in intensive care units. Nonetheless, they continue to incur a very significant annual cost, up to approximately 12 million Euros.20 This cost is associated with increased hospital stay, drug treatment and the consumption of material and human resources. In our series, analysis of the VCRB aetiology confirmed, as in other studies,21 the leading role of SCN as the primary pathogen responsible for this infection.
The results of our study showed that ultrasound was rarely used to guide CVC insertion, although the literature clearly recommends its use to reduce mechanical complications and facilitate the procedure.6 The reasons that ultrasound was seldom used might be because most of the catheters were peripherally inserted, which is a route with few risks of mechanical complications during insertion, and because the staff who participated in the study were not used to using it.
The guidelines recommend that ultrasound-guided insertion should only be performed by staff fully trained in the procedure (CDC 2011, level of evidence Ib)6.
Although the practitioner who is inserting the CVC administers a local anaesthetic to the conscious patient prior to the procedure, the needle puncture during suturing causes the patient a high level of pain, and therefore increases their discomfort. Our study shows that the use of sutureless CVC securement devices prevents the secondary pain from fixing the catheter to the skin.
The data obtained show greater displacement of CVC fixed with a suture, principally via humeral access, bearing in mind that this type of access was used on most of the sample in both groups. After analysing the data, the greater catheter displacement could be attributed to several factors: on the one hand, more bleeding via the puncture and suture sites, which required more frequent dressing changes and the consequent handling of the catheters, and on the other, in line with the previous point, there were also more local signs of infection, which encourages less stable suture sites.
One of the strengths of this study is that it is one of the first randomised, parallel-group studies to compare the adhesive system with the suture fixation system. It also had a considerable sample, with only 5 losses to follow-up. The remaining data were collected in their entirety. By contrast, it has certain limitations. This study was undertaken in the coronary unit of a single centre, therefore the data are not extrapolable to all patients admitted to critical care units. In addition, it is a pilot study, and although the total sample included comprised 100 patients, the results obtained must be corroborated by further studies. Finally, although a higher Charlson index was detected in the suture fixation system, the post hoc analysis shows that there were no differences in terms of a high Charlson index and the onset of complications. Another limitation of the study is that the experience of the professionals who inserted the CVC was not taken into account, or the number of punctures used to insert it. The level of experience of the staff is a factor that should be born in mind when CVC are inserted, a greater number of punctures might be required and the consequent greater risk of bleeding through the puncture site.
Last but not least, we must promote safety policies for patients as well as practitioners. The use of a sutureless fixation device reduces the risk of injury to healthcare professionals by accidental puncture. No accidental punctures were recorded in our study during suture fixation. However, according to the Occupational Safety and Health Administration (OSHA, 2001), and the guidelines for the prevention of intravascular catheter-related infections of the Healthcare Infection Control Practices Advisory Committee, sutureless devices should be used to secure CVC because they prevent accidental puncture to healthcare professionals.6 Furthermore, we should highlight the high level of satisfaction of the staff who used the adhesive system, and particularly recommended its use.
ConclusionsThe CVC secured using the adhesive system had fewer local infectious complications and VCRB that those fixed by suture. The stability of the catheters was greater for those secured using the adhesive system, and there was less displacement while they were in situ. The catheters were handled more in the group with sutures due to the greater need for dressing changes secondary to bleeding through the puncture and suture sites.
The professionals who took part in the study showed a high level of satisfaction with the adhesive fixation system, highlighted the comfort for the patient, and recommended its use in daily clinical practice.
Conflict of interestsThe authors have no conflict of interests to declare.
We would like to thank all the nursing staff of the Coronary Unit of the Bellvitge University Hospital for their collaboration in the data collection, without which this study would not have been possible. We would also like to thank our hospital's Infection Control Team for their support and advice while we undertook this study.
Please cite this article as: Molina-Mazón CS, Martín-Cerezo X, Domene-Nieves de la Vega G, Asensio-Flores S, Adamuz-Tomás J. Estudio comparativo sobre fijación de catéter venoso central mediante sutura versus dispositivo adhesivo. Enferm Intensiva. 2018;29:103–112.