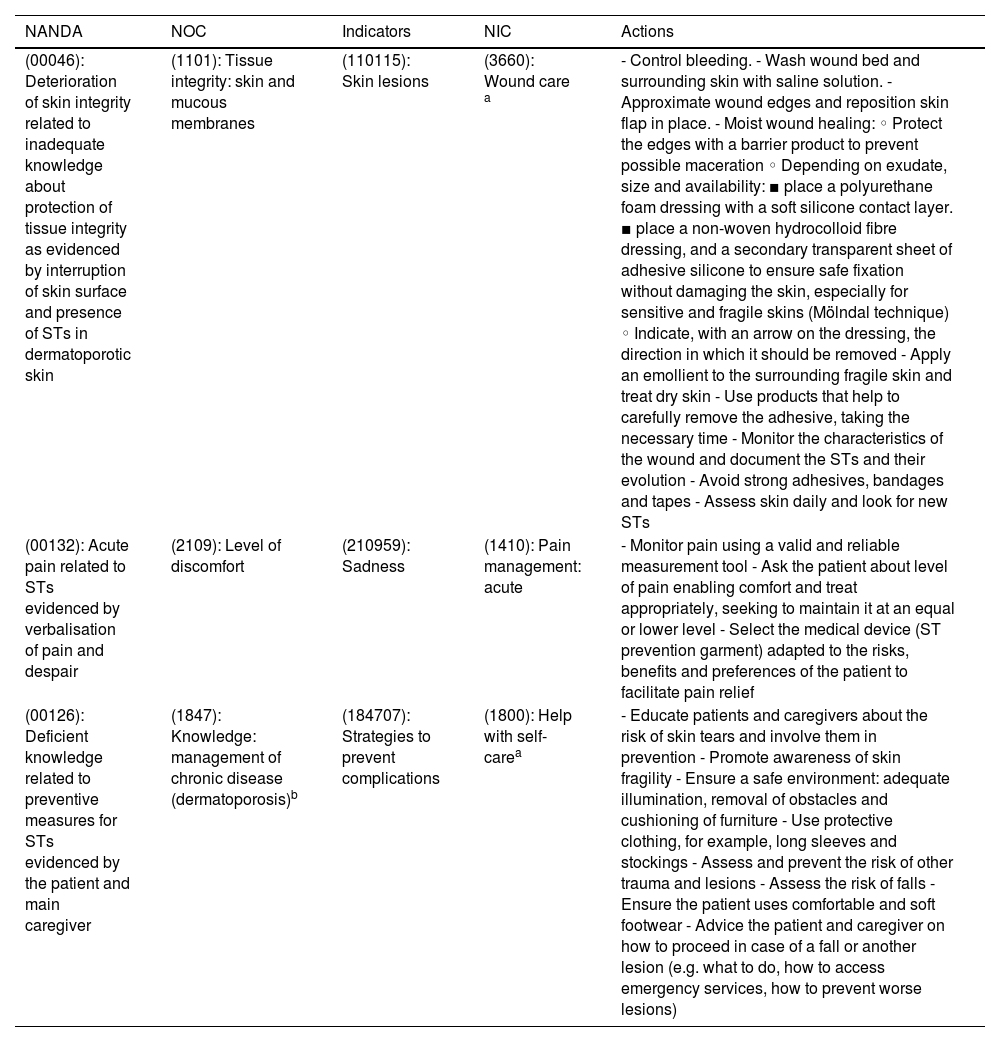
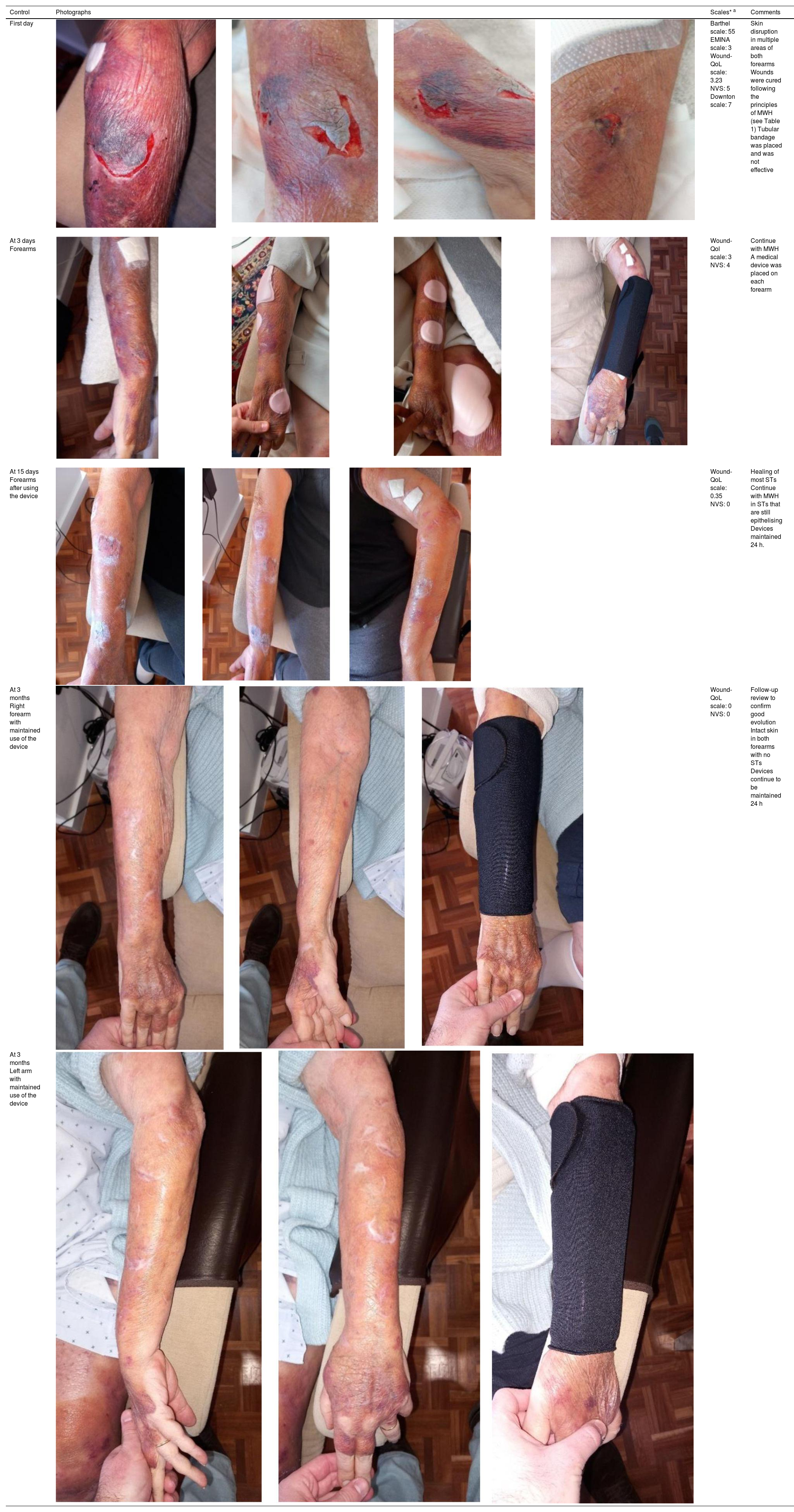
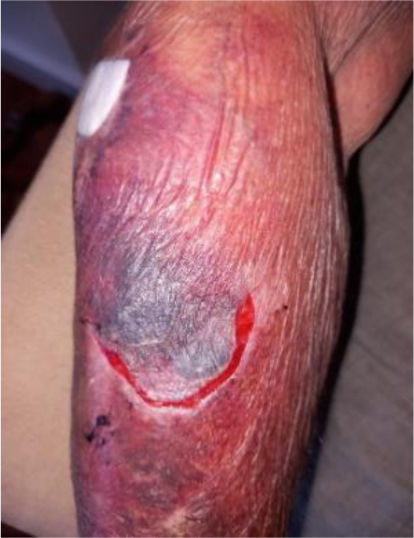
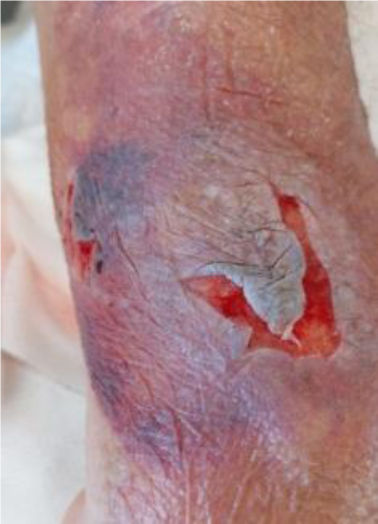
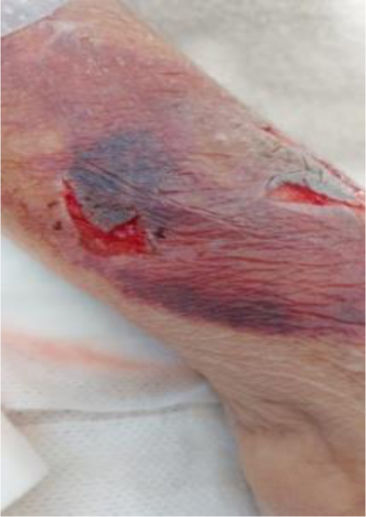
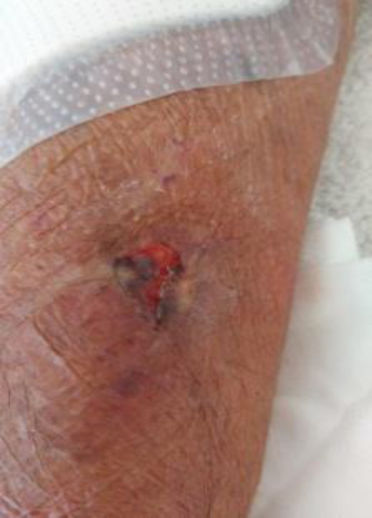
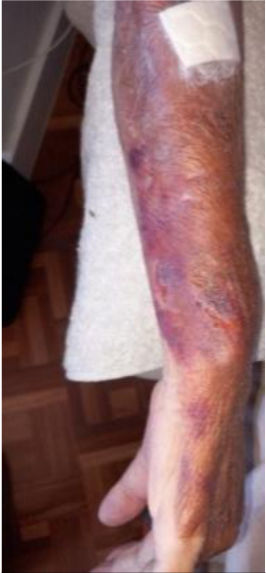
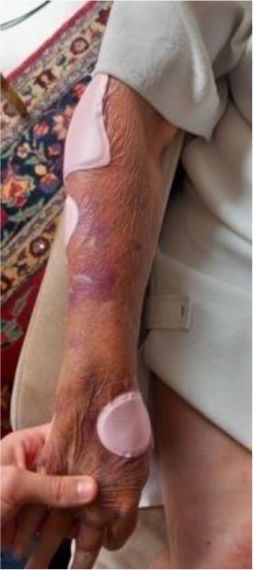
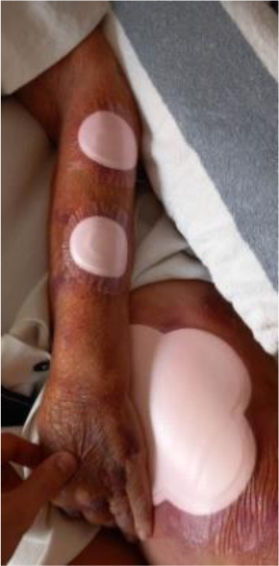
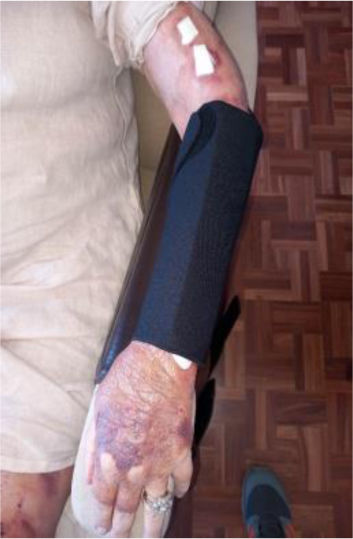
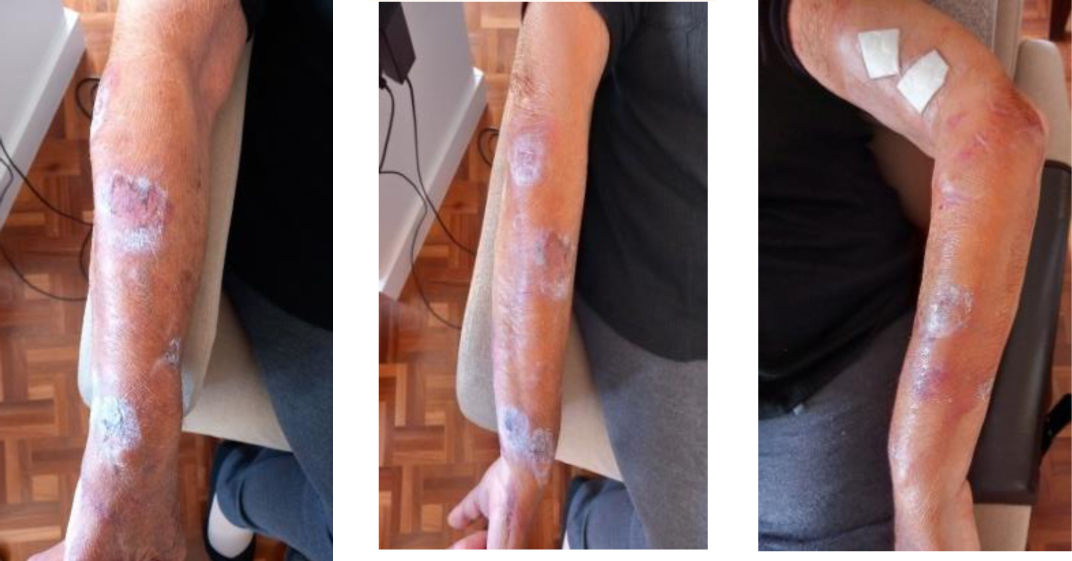
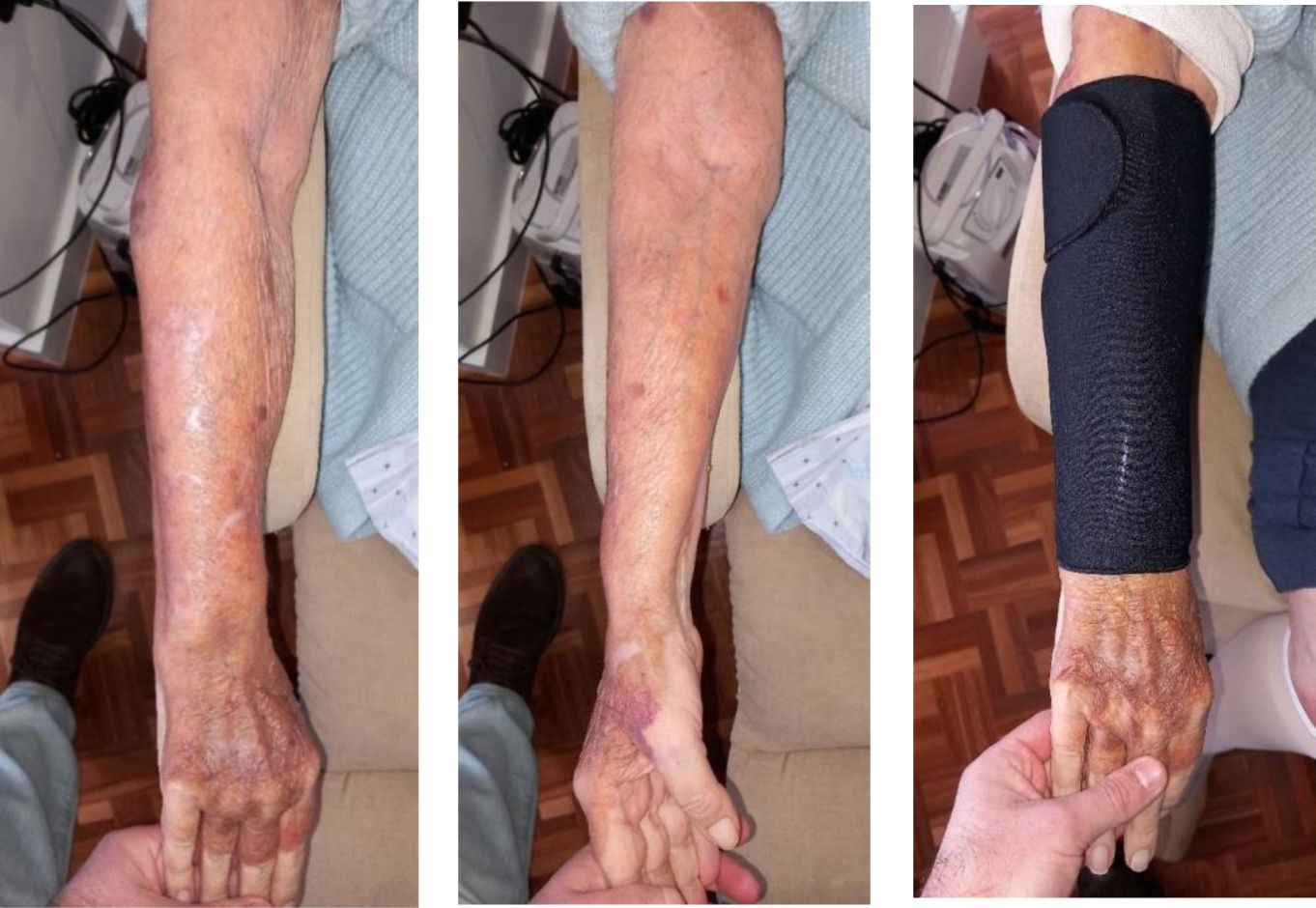
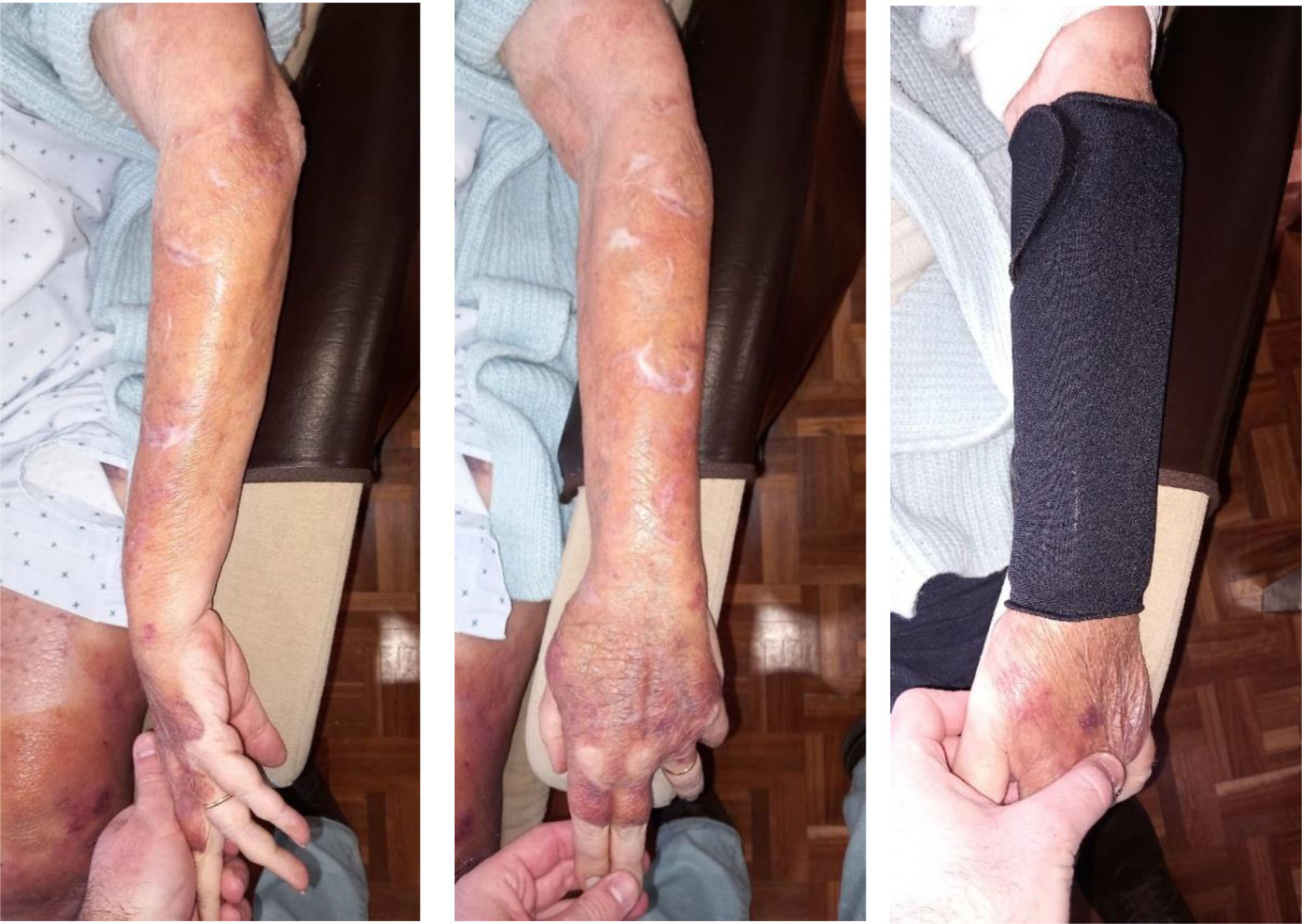
We present the clinical case of a 64-year-old multi-pathological patient who presents numerous skin tears in the upper extremities.
The aim is to restore skin integrity in a patient with numerous skin tears (ST) on both forearms using a new class I medical device composed of elastic and adjustable medical grade encapsulated gel.
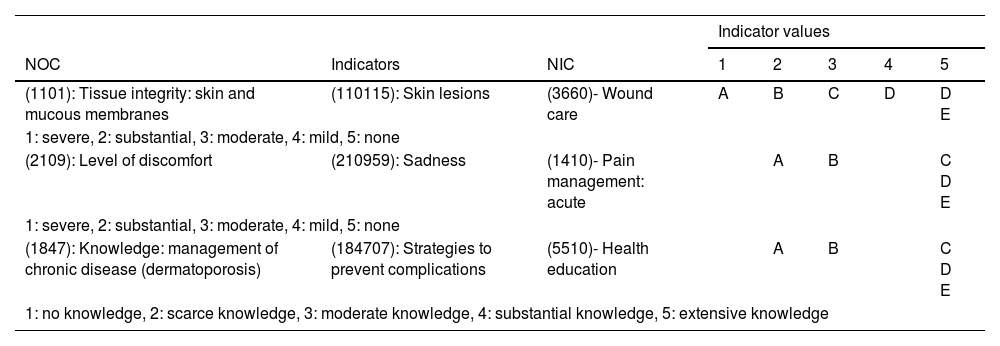
Once the NANDA International diagnostic labels were identified using the NNNConsult online tool, the expected results were planned along with the nursing interventions.
The care plan and cure guidelines are detailed based on the consensus evidence of the International Skin Tear Advisory Panel, also including preventive activities in risk areas (General Health, Mobility and Skin), in addition to the implementation of a new class I medical device. Finally, skin integrity is restored.
This work shows the positive preventive and therapeutic results in fragile skin and ST when carrying out a multiple care strategy with a new medical device.
Se presenta el caso clínico de una paciente pluripatológica de 64 años con numerosos desgarros cutáneos (DC) en extremidades superiores.
El objetivo es restablecer la integridad cutánea en una paciente con numerosos desgarros cutáneos en ambos antebrazos utilizando un nuevo dispositivo médico clase I compuesto por gel encapsulado de grado médico elástico y ajustable.
Una vez identificadas las etiquetas diagnósticas NANDA Internacional utilizando la herramienta online NNNConsult, se planificaron los resultados esperados junto con las intervenciones enfermeras.
Se detalla el plan de cuidados y pauta de curas basadas en las evidencias de consenso del Internacional Skin Tear Advisory Panel, recogiendo también las actividades preventivas en las áreas de riesgo (Salud general, Movilidad y Piel), además de la implementación de un nuevo dispositivo médico clase I. Finalmente, se consigue restablecer la integridad cutánea.
Este trabajo muestra los resultados positivos preventivos y terapéuticos en piel frágil y DC al llevar a cabo una estrategia de cuidados múltiple con un nuevo dispositivo médico.