The diagnosis of genital ulcers in children is of particular importance, as they can be an indication of pathologies that are either non-infectious (Behçet’s disease, inflammatory bowel disease, adverse drug reactions or Lipschütz ulcers) or infectious, and in some cases may be associated with sexual abuse. Of particular note are infections due to herpes simplex virus, cytomegalovirus and Epstein-Barr virus. Bacterial causes include streptococcus and staphylococcus infections, gram-negative bacteria and ecthyma gangrenosum due to Pseudomonas aeruginosa1.
Actinotignum schaalii (A. schaalii) is a gram-positive, facultative anaerobic bacillus that resides in the urogenital mucocutaneous epithelium2. Its true clinical significance has probably continued to be underestimated due to the difficulty of isolating it in routine cultures3. To date, just seven previous cases of paediatric infection have been described, primarily urinary and, less commonly, genital infections4, but never ulcers. We present the first case to be described in the literature of genital ulcers associated with isolation of this microorganism.
The patient was a seven-year-old boy who was seen following a telephone consultation during the COVID-19 lockdown. He presented with ulcers on the glans and balanopreputial region that had appeared 24 h earlier, without fever, history of trauma or recent use of new medications. The patient had been previously diagnosed with primary nocturnal enuresis and daytime wetting, with an increase in the preceding weeks, coinciding with increased anxiety due to the lockdown situation. This patient also had encopresis and Tourette’s syndrome. The was no evidence of sexual abuse/assault.
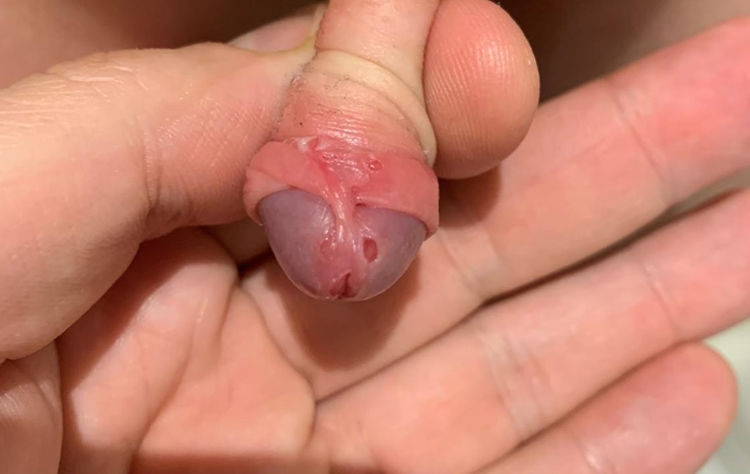
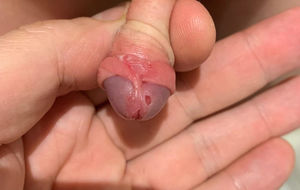
During the examination, an ulcer of 4 mm was found on the glans (Fig. 1) and another of similar size but less deep, mirroring the first contralaterally, as well as others below the level of the balanopreputial frenulum, which was found to be erythematous. He did not present oropharyngeal lesions and the rest of the physical examination was normal. A sample of exudate was taken from the ulcers for a microbiological study. The baseline blood test was normal, including C-reactive protein <0.2 mg/l, blood count: 11,890 leukocytes/mm3 (31% polymorphonuclear neutrophils, 54% lymphocytes, 7% monocytes, 7% eosinophils).
In the microbiological study, after 48 h’ incubation in CO2, a large number of very small (<1 mm) colonies had grown in pure, non-haemolytic, cytochrome-oxidase and catalase-negative cultures in a sheep blood agar medium (BD, Spain), which corresponded to gram-positive bacteria with a slightly curved morphology and traces of corynebacterium, with slow growth. They were correctly identified using MALDI-TOF mass spectrometry (Bruker Biotyper, Billerica, MA, USA) as A. schaalii (with a maximum score of 2.212). The antibiotic sensitivity study, in a blood agar medium in CO2, found the following minimum inhibitory concentration values (mg/l) (interpreted at 48 h, according to the EUCAST criteria for anaerobic gram-positive bacteria and the CLSI 2020 criteria for anaerobic bacteria for tetracycline): ampicillin (0.064), clindamycin (0.023) and tetracycline (0.5) (sensitive), and metronidazole (>256) (resistant). Following isolation of the ulcer, a urine culture was taken to search specifically for A. schaalii, which was negative.
A serology test for Chlamydophila pneumoniae, Mycoplasma pneumoniae, syphilis, Toxoplasma gondii, hepatitis C virus (HCV), hepatitis B virus (HBV) and human immunodeficiency virus (HIV) was negative. Only anti-cytomegalovirus and anti-Epstein-Barr virus IgG were detected. The antinuclear antibodies determination had a speckled pattern (1/160). Treatment was administered orally with amoxicillin (50 mg/kg/day, seven days) and topically with fusidic acid, as well as hygiene measures, with a favourable clinical course and the lesions disappeared at seven days.
Although globally the most common cause of genital lesions is herpes simplex virus infection, in children and non-sexually-active adolescents, other non-infectious and infectious aetiologies should be considered5. The importance of detecting any such process, and of ruling out sexual transmission, necessitates a precise aetiological diagnosis. A. schaalii is a microorganism occasionally isolated in elderly patients with underlying genitourinary disease, and can be responsible for invasive infections in immunosuppressed patients1,2,6. Of the paediatric cases mentioned previously, five were urinary infections, with one case of intradural abscess and another of balanoposthitis4. As in adult patients, there is an association with a history of urogenital disease, which was present in five of the seven cases. Conditions of prolonged moisture in the genital area and anaerobiosis promote its growth. This was associated with four of the seven cases (enuresis, prolonged wearing of nappies or bathing suits). Likewise, our patient suffered from primary enuresis, with a worsening of the clinical picture in the context of prolonged lockdown due to the COVID-19 pandemic. The family described an increase in daytime wetting associated with the stressful situation, which could be considered an adverse effect of the lockdown.
The case presented is, to our knowledge, the first isolation of A. schaalii in genital ulcers in either children or adults. Although the presence of this genus has been demonstrated in the urogenital microbiota of asymptomatic patients2, the species A. schaalii has shown the greatest pathogenic potential. During the patient’s initial care in the paediatric emergency department, sending a urine sample for culture was not considered; the urinalysis using a reactive strip was normal, and the patient did not present signs or symptoms compatible with a urine infection other than his underlying enuresis, nor did he show any alterations of infection parameters in blood tests. Nevertheless, following the identification of the microorganism, on reviewing the patient, a microbiological study in urine was requested specifically to search for this microorganism, which was negative. Although in this case other causes of genital ulcers were plausibly ruled out, as the patient showed no other symptoms and presented an adequate response to antibiotic treatment, further case studies are required to fully explain this new association. For the treatment of infections caused by A. schaalii, amoxicillin or cephalosporins are recommended, as it is generally resistant to fluoroquinolones and cotrimoxazole4.
One limitation of this work is that identification was not performed using a molecular method, although the correct identification of this species with MALDI-TOF has previously been described7.
In conclusion, opportunistic microorganisms such as A. schaalii must be taken into account in the differential diagnosis of genital ulcers in patients with urological disease and, in particular, when there is prolonged exposure to moisture in the genital area.
Conflicts of interestThe authors declare that they have no conflicts of interest.
Please cite this article as: Vázquez-Pérez Á, Santos-Pérez JL, Herrera-Goicoechea A, Gutiérrez-Fernández J. Actinotignum schaalii y úlceras genitales en paciente pediátrico. Enferm Infecc Microbiol Clin. 2021;39:480–481.