Acromegaly is a disease characterized by excess growth hormone (GH), starting after the closure of the epiphyseal plates of long bones. Somatostatin is a peptide hormone with a short half-life (2–3min) which is synthesized in multiple tissues, including the hypothalamus, to inhibit GH secretion. This hormone mediates its actions through five receptor subtypes (SSRs), of which SSR2 and SSR5 are the most widely expressed in pituitary adenomas.1 Octreotide is a synthetic analog of somatostatin with an inhibitory action on the secretion of GH and various gastrointestinal and pancreatic hormones.1
We report the case of a 37-year-old male patient from Mérida (Venezuela) with no personal or familial pathological history who was referred to the endocrinology unit for physiognomic changes over the previous seven years, highly severe holocranial headache, and bitemporal heteronymous hemianopsia starting five months before.
The physical examination findings included: a body weight of 102kg, a height of 185.0cm, a body mass index (BMI) of 29.8kg/m2, a heart rate of 72bpm, blood pressure of 130/80mmHg, and coarse facial features characterized by nose enlargement, prognathism, bulging of the forehead, and prominent cheeks. Thick skin with an oily texture, upper thoracic lordosis with compensatory lumbar hyperlordosis, prominence of the lower part of the sternum, and big hands and feet were also found, and confrontation campimetry showed bitemporal heteronymous hemianopsia. No cardiovascular or respiratory changes were found.
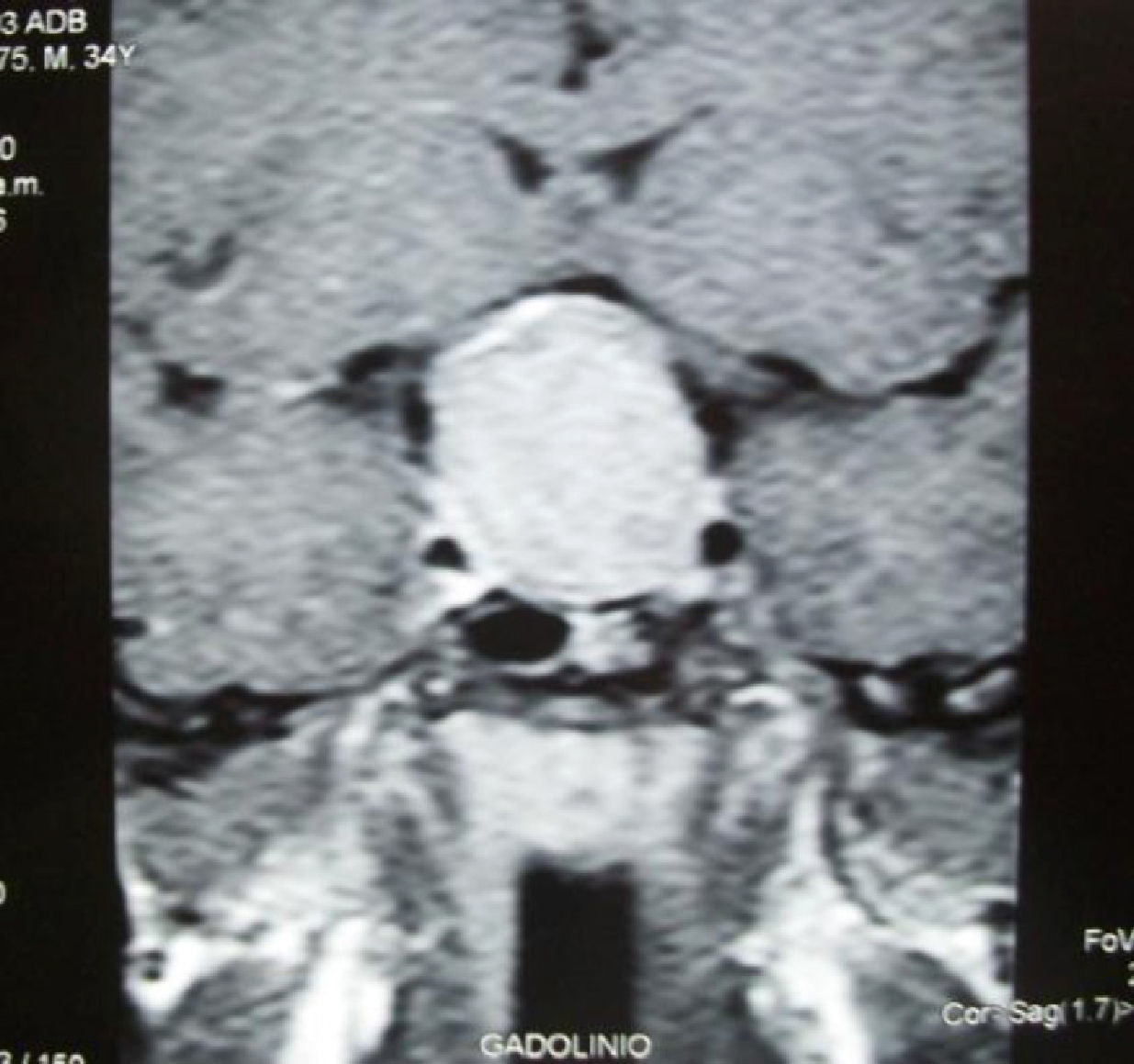
Laboratory tests revealed no hematological changes. Blood chemistry showed normal glucose, kidney and liver function, calcium, and phosphorus levels. Additional test results included: basal plasma GH, 9.7μg/L (NR, 0–2.5); GH 2h after a 75g oral glucose tolerance test, 8.2μg/L (NR, less than 1); IGF-1 (insulin-like growth factor), 355ng/mL (NR for age, 109–284); thyroid-stimulating hormone (TSH), 2.45μIU/mL (NR, 0.3–4.2); free thyroxine (FT4), 1.20ng/dL (NR, 0.8–2); and prolactin, 12.8ng/mL (NR in males, 0–15). Magnetic resonance imaging with gadolinium contrast revealed a pituitary macroadenoma with suprasellar extension and optic chiasm compression (Fig. 1).
Acromegaly was diagnosed based on the clinical, laboratory, and imaging findings, and surgery was performed, but only partial resection of the pituitary macroadenoma could be performed. The patient was assessed again at three months. He reported significant improvements in headache and visual field disturbances, and was found to have a basal GH level of 3.2μg/L and a plasma IGF-1 level of 298ng/mL. Thus, since the biochemical criteria for disease control had not been met, treatment with octreotide 100μg by the subcutaneous route every 8h was prescribed, but after 10 days the patient started to experience dizziness and generalized weakness, and a physical examination revealed a heart rate of 41bpm.
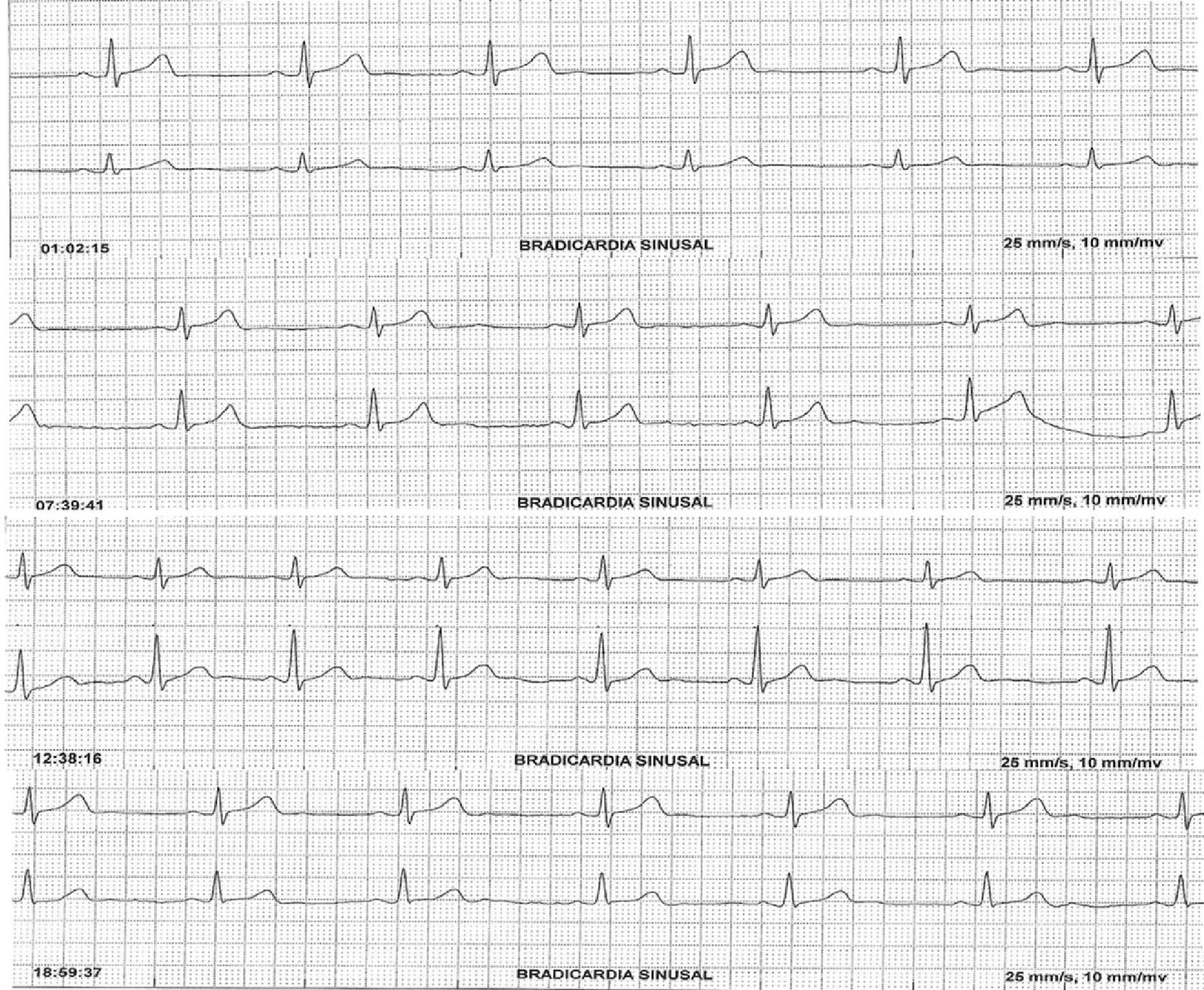
The patient was referred to the cardiology department, where 24-h Holter monitoring revealed sinus bradycardia (Fig. 2) with a mean heart rate of 44bpm. Octreotide was discontinued, and his heart rate normalized.
Somatostatin analogs are the main therapeutic agents for the management of acromegaly.1 They have traditionally been used as an adjuvant therapy in the event of surgery failure in order to achieve biochemical control and to decrease tumor volume, as well as to provide partial or total disease control from the time of the administration of radiotherapy to the start of the maximum benefit derived from it.2 The use of these agents as first-line drugs for pituitary macroadenomas with suprasellar extension to decrease tumor volume before surgery has also been postulated.2 However, they were not prescribed for this purpose in our patient due to the presence of compression signs indicating immediate surgery.
Various factors possibly accounting for the individual response to somatostatin analogs have been proposed, including: age and sex, GH levels before treatment, the GH suppression test, histopathology (tumors with dense granulation), immunohistochemical detection of SSR2, the presence of the gsp oncogene, and treatment dosage and duration.3
The adverse effects most commonly associated with somatostatin analogs are gastrointestinal in nature and include nausea, vomiting, flatulence, and diarrhea. They are of variable severity and often resolve upon drug discontinuation.4 Octreotide-induced bradycardia has previously been reported as an unusual finding in different clinical situations.5,6 As in our case, Herrington et al.5 reported a patient who experienced sinus bradycardia (38bpm) after subcutaneous octreotide administration. Moreover, a significant decrease in heart rate with a concomitant increase in peripheral vascular resistance was reported in a small patient series.7
The most plausible mechanism to explain the negative chronotropic effect of octreotide is the direct action of somatostatin on the heart. Somatostatin coexists with acetylcholine in presynaptic vagal endings, and may therefore be released by vagus nerve stimulation.8 The main cardiovascular effects of somatostatin include decreases in heart rate and myocardial contractility and a slowing of cardiac conduction velocity. Many actions of somatostatin or its analogs result from interaction with SSRs, which belong to the family of G protein-coupled receptors and whose activation causes the inhibition of adenylate cyclase with a resultant decrease in the intracellular concentration of cyclic adenosine monophosphate (cAMP) which causes decreased conduction through voltage-dependent calcium channels and increased membrane permeability to potassium.9 In fact, because of its negative chronotropic effect and its ability to prolong atrioventricular conduction, somatostatin has been successfully used to treat supraventricular tachycardia.10
We conclude that sinus bradycardia was due to the use of octreotide, as was shown by a clear and consistent time sequence in both symptom occurrence and clinical improvement upon drug discontinuation. This potential side effect should therefore be considered when treating patients with acromegaly who require the use of somatostatin analogs.
Please cite this article as: Lima-Martínez MM, López-Méndez G, Mangupli R. Bradicardia sinusal inducida por octreotide en un varón con acromegalia. Endocrinol Nutr. 2013;60:e7–e9.