To show our results with the use of a polypropylene mesh at the stoma site, as prophylaxis of parastomal hernias in patients with rectal cancer when a terminal colostomy is performed.
MethodsFrom January 2010 until March 2014, 45 consecutive patients with rectal cancer, underwent surgical treatment with the need of a terminal colostomy. A prophylactic mesh was placed in a sublay position at the stoma site in all cases. We analyse Demographics, technical issues and effectiveness of the procedure, as well as subsequent complications.
ResultsA prophylactic mesh was placed in 45 patients, 35 male and 10 females, mean age of 66.2 (47–88) and Body Mass Index 29.1 (20.4–40.6). A total of 7 middle rectal carcinoma, 36 low rectal carcinoma, one rectal melanoma and one squamous cell anal carcinoma were electively treated with identical protocol. Abdominoperineal resection was performed in 38 patients, and low anterior resection with terminal colostomy in 7. An open approach was elected in 39 patients and laparoscopy in 6, with 2 conversions to open surgery. Medium follow up was 22 months (2.1–53). Overall, 3 parastomal hernias (6.66%) were found, one of which was a radiological finding with no clinical significance. No complications related to the mesh or the colostomy was found.
ConclusionsThe use of a prophylactic polypropylene mesh placed in a sublay position at the stoma site is a safe and feasible technique. It lowers the incidence of parastomal hernias with no increased morbidity.
Presentar nuestra experiencia en la implantación periestomal de malla de polipropileno en espacio preperitoneal como profilaxis de hernia paraestomal en colostomías terminales en pacientes intervenidos por neoplasia rectal.
MétodosDesde enero de 2010 hasta marzo de 2014, 45 pacientes consecutivos afectados de neoplasia de recto que requirieron implantación de colostomía terminal definitiva fueron intervenidos y analizados. En todos ellos se implantó una malla de polipropileno profiláctica en espacio preperitoneal periestomal. Analizamos variables demográficas, aspectos técnicos y efectividad de la técnica así como complicaciones consecuentes.
ResultadosSe implantó malla profiláctica en 45 pacientes, 35 varones y 10 mujeres, con una media edad de 66,2 años (47–88) y un Índice Masa Corporal de 29,19 (20,4–40,6). Se intervinieron de manera programada y con idéntico protocolo 7 adenocarcinomas de recto medio, 36 de recto bajo, un melanoma de recto y un carcinoma de células escamosas de ano; realizándose una amputación abdominoperineal en 38 pacientes y resección anterior baja con colostomía terminal en 7 pacientes. La vía de abordaje fue laparotómica en 39 casos y laparoscópica en 6 casos, 2 de los cuales se convirtieron a laparotomía. La mediana del tiempo de seguimiento fue de 22 meses (2,1–53). Se evidenciaron 3 hernias paraestomales (6,6%), siendo un hallazgo radiológico durante tomografía computarizada de control. No hubo complicaciones asociadas a la colostomía ni a la implantación de la malla.
ConclusionesLa colocación de una malla de polipropileno en localización paraestomal preperitoneal es fácilmente reproducible, disminuyendo la incidencia de hernia paraestomal sin aumentar la morbilidad ni la mortalidad.
A parastomal hernia is an incisional hernia developed in the vicinity of a stoma, that causes the passing of intestinal loops through the aponeurotic orifice.1 The incidence of parastomal hernia is about 50% in the first year after construction of a colostomy.2 Its symptoms may vary from discomfort to potentially fatal complications.
Up to one third of parastomal hernias may require surgical intervention.3 The surgical procedures used have a high failure rate. Recurrence after simple aponeurotic repair or relocation of the stoma is 46%–100%,4 while after the use of prosthetic materials it is 8%–56%.5–8 This is why several authors have proposed that the best strategy to fight it may be to prevent its onset.3,6,9,10 For this purpose, a mesh is implanted when preparing the colostomy.
Bayer et al. (1986) were the first to describe the implantation of a mesh at the time of colostomy construction.11 The locations and types of prophylactic mesh vary according to different studies. Therefore, the different types of mesh used are politetrafluoroethylene, polypropylene and composite mesh which may be implanted in intraperitoneal cavity (IPOM), preperitoneal (sublay) or supraaponeurotic (onlay).12
Two recent reviews have shown that parastomal hernia can be prevented by placing a polypropylene mesh in the peristomal preperitoneal space at the time of creation of the colostomy.13,14 In the clinical trials of Jänes and Serra-Aracil, a lower rate of parastomal hernia was evidenced in groups with prophylactic parastomal mesh (13.3 and 14.8%) in relation to the control group with no mesh (80 and 40.7%).15,16
The purpose of this article is to present our experience with the implantation of polypropylene in the peristomal preperitoneal space as prophylaxis of parastomal hernia in terminal colostomies in patients with rectal neoplasms.
MethodsFrom January 2010 to March 2014, the Colorectal Surgery Unit of Hospital Virgen Macarena carried out scheduled surgery on 45 patients with rectal neoplasia who required construction of a definitive terminal colostomy. A prophylactic polypropylene mesh was implanted in all patients in the peristomal preperitoneal space.
The inclusion criteria of our study were patients with rectal neoplasia operated on by scheduled surgery and who, because they required a definitive colostomy, had a prophylactic parastomal mesh implanted.
This series does not include patients operated on as an emergency, or those who had a lateral colostomy or, temporary stomas.
The approach used was both laparotomy and laparoscopy. All patients included in the study were diagnosed with malignant rectal disease. The surgical technique selected for each patient was determined by the colorectal cancer protocol of our unit. Tumour extension directly to the sphincter or pelvic muscle floor and tumours immediately adjacent to the pectineal line not eligible for local or radiotherapy treatment were directly treated by abdominoperineal amputation. Furthermore, a low anterior rectal resection with terminal colostomy was performed in patients with elevated anastomotic risk, due to the comorbidities or because local conditions made it not advisable.
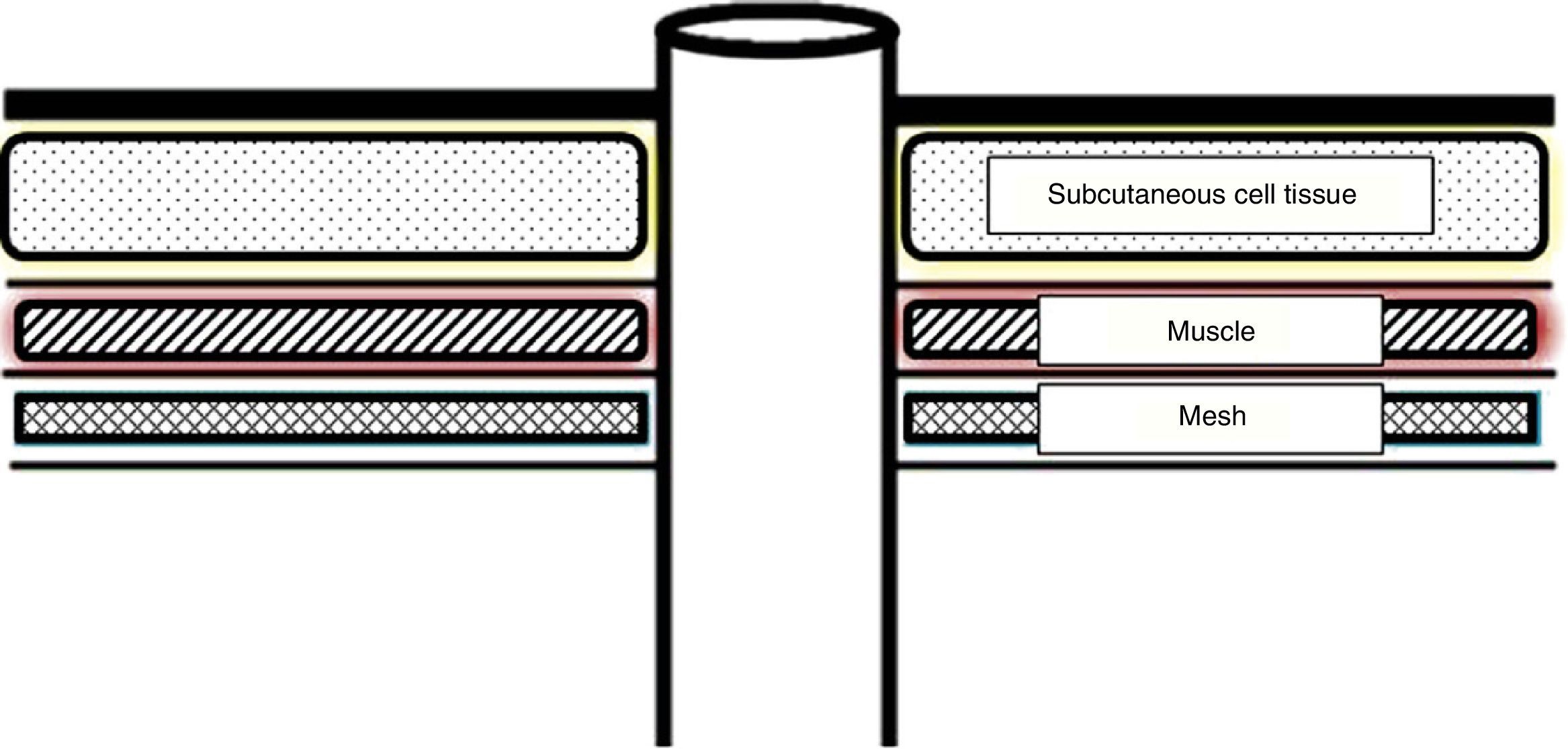
The same colostomy creation method was followed in all cases. By making a change in the Jänes and Israelsson technique, the colon subject to the colostomy is closed with a mechanical suture, reducing the possibility of contamination to a minimum. An oval incision is performed in the skin of the left iliac fossa, marked by the stomatherapist before the intervention. After exposing the left rectum anterior sheath, a cross-shaped incision is performed. The abdominal rectus muscle is dilacerated in the direction of the fibres. The preperitoneal retromuscular cavity is dissected digitally, where the Alexis® type separator is adjusted to the size of the orifice. In this way, we have the necessary surgical field to operate comfortably and get a correct expansion and fixation of the mesh. Into the space a 10cm×10cm low molecular weight polypropylene mesh is placed, with a cross-shaped incision in the centre of the prosthetic to allow the colon to pass through (Fig. 1). The mesh is fixed to the 4 cardinal points of the anterior aponeurosis with Vicryl 2/0 suture. Then, the underlying peritoneum is split to the mesh and the colon is passed through it. The stoma is fixed to the skin using absorbable sutures.
The implantation of the prophylactic mesh in the preperitoneal space is a technique of minimal difficulty, which can easily be reproduced, and does not add more than 20min to the total time of surgery.
Patients are examined in a specific consultation at the Colorectal Surgery Unit one month after the surgical intervention and afterwards every 3 months during the first 2 years and every 6 months afterwards, with a control CT each year. In turn, from the immediate postoperative period, we have a stomatherapist nurse who, in addition to educating patients in the management and care of the colostomy, performs a weekly review of each patient during the first 3 months, monthly during the first year and every six months thereafter. That stomatherapist notifies the Unit of any complications or incidents relating to the colostomy.
Parastomal hernia episodes found both clinically and radiologically were recorded.
Demographic variables were also analysed (gender, age, Body Mass Index), technical aspects (viability and reproducibility of the technique) and effectiveness of the technique, as well as related complications (infection of the wound, disinsertion of the stoma, necrosis of the stoma, stenosis of the stoma and fistula).
We compared the results of our series with the global incidence of parastomal hernia.
ResultsProphylactic mesh was used in 45 patients. The characteristics of the group, duration of the intervention and mean stay are reflected in Table 1.
Scheduled surgery with identical protocol was carried out on 7 mid rectal adenocarcinomas, 36 low rectal adenocarcinomas, one rectal melanoma and one anal squamous cell carcinoma. An abdominoperineal amputation was performed in 38 patients and low anterior resection with end colostomy in 7 patients.
The approach was mid-line suprainfraumbilical laparotomy in 39 cases and laparoscopic in 6 cases, of which, 2 patients required conversion to laparotomy due to technical difficulties. The mean surgical time was 188min and the mean stay was 10.54 days (5–34).
During the immediate postoperative period, the following complications appeared: one perineal infection, one infection of pelvic haematoma, 2 episodes of evisceration and one episode of intestinal obstruction caused by adhesions. No complications associated to the colostomy or complications related to the implantation of the mesh were described.
The follow-up time of patients varied from 2.1 months to 53 months, with a mean of 22 months.
During the follow-up of patients in the office, 2 parastomal hernias were diagnosed during physical examination (4.44%). Both hernias appeared in the first 12 months after the surgery. The great symptomatology of one of them made a new surgical intervention necessary to repair the hernia using the Sugarbaker technique.
One of the annual CTs revealed a parastomal hernia with no clinical symptoms, which was not evidenced during physical examination in the office (2.2%). That finding appeared the second year after surgery. This way, the global incidence of parastomal hernia in our series is 6.6%, with 2 episodes diagnosed by physical examination (4.44%) and one by a radiological test (2.2%).
During follow-up of the patients, 3 deaths were recorded as a consequence of the natural evolution of the oncologic disease. None was related to complications derived from surgery. The rest of patients continue with follow-up to this date.
DiscussionThe Colorectal Surgery Unit of Hospital Universitario Virgen Macarena operates a mean of 58 scheduled rectum neoplasms per year, performing abdominoperineal amputation in a mean of 17 patients per year (29.31%). Although the ideal proportion between anterior resection and abdominoperineal amputation cannot be determined, it is recommended that the percentage of rectal tumours treated by abdominoperineal amputation be less than 40%.17
Parastomal hernia is the most frequent complication associated to stomas, especially definitive end colostomies. Its high incidence entails a huge social and health problem because it limits the daily activity of many colostomy carriers, and a third of those require new surgical interventions to treat that complication.3 The surgical procedures to repair it have a high failure rate, with no gold standard to treat this affliction. Therefore, we consider that the best strategy to defeat parastomal hernia is to prevent its onset.
The implantation of the mesh in the preperitoneal space is presented as a simple technique and with no significant increase of surgical time, with the only limitation being the presence of a previous scar in the site for the implantation of the colostomy, which may hinder a proper dissection of the preperitoneal space.
After application of the prophylactic mesh, the incidence of parastomal hernia in our series (6.66%) is comparable to the results obtained by Jänes and Serra-Aracil15,16 and, therefore, significantly lower than the 50% rates reported in the global historical incidence for parastomal hernia published in larger series.2,18 However, we would have to assess whether the appearance of a radiological parastomal hernia in the follow-up of a patient, with no clinical repercussion, and also undetectable in the clinical examination, should be considered a casual finding or if it should be considered as a parastomal hernia per se.
Our group decided to perform parastomal hernia prophylaxis based on previously published experience, without proposing a comparative study, since we consider that scientific evidence sustains the suitability of performing the prophylaxis when performing the stoma, which is far from the standard in many work groups. We provide our experience and results to support that option, which we consider compulsory in our practice based on the results obtained.13–16
The recorded complications, such as pelvic abscess and the infection of the perianal wound, are attributable to the abdominoperineal amputation technique.19
We decided to use a polypropylene prosthesis based on the previous experience of other groups due to its lower cost and because it was a potentially contaminated localisation, with no complication described that was related or attributed to the use of that mesh in our series.
Therefore, we can affirm that the application of a polypropylene mesh in the preperitoneal cavity is a safe and easily reproducible method with no significant increase in operating time, which does not associate morbidity to the surgical technique and evidently decreases the incidence of parastomal hernia, confirming previous studies gathered in the bibliography.
Authors Contribution- -
Dr. Javier Valdés-Hernández: study design, analysis and interpretation of results, article drafting, critical review and final version approval.
- -
Dr. Juan Antonio Díaz Milanés: study design, acquisition and collection of data, analysis and interpretation of results, article drafting, critical review and final version approval.
- -
Prof. Luis Cristóbal Capitán Morales: study design and final version approval.
- -
Dr. Francisco Javier del Río la Fuente: critical review and final version approval.
- -
Dr. Cristina Torres Arcos: study design and critical review.
- -
Dr. Jesús Cañete Gómez: analysis and interpretation of the results and critical review.
- -
Dr. Fernando Oliva Mompeán: study design and final version approval.
- -
Prof. Francisco Javier Padillo Ruiz: study design and final version approval.
The authors declare that there are no conflicts of interest.
Please cite this article as: Valdés-Hernández J, Díaz Milanés JA, Capitán Morales LC, del Río la Fuente FJ, Torres Arcos C, Cañete Gómez J, et al. Profilaxis de la hernia paraestomal mediante malla de polipropileno en espacio preperitoneal. Cir Esp. 2015;93:455–459.