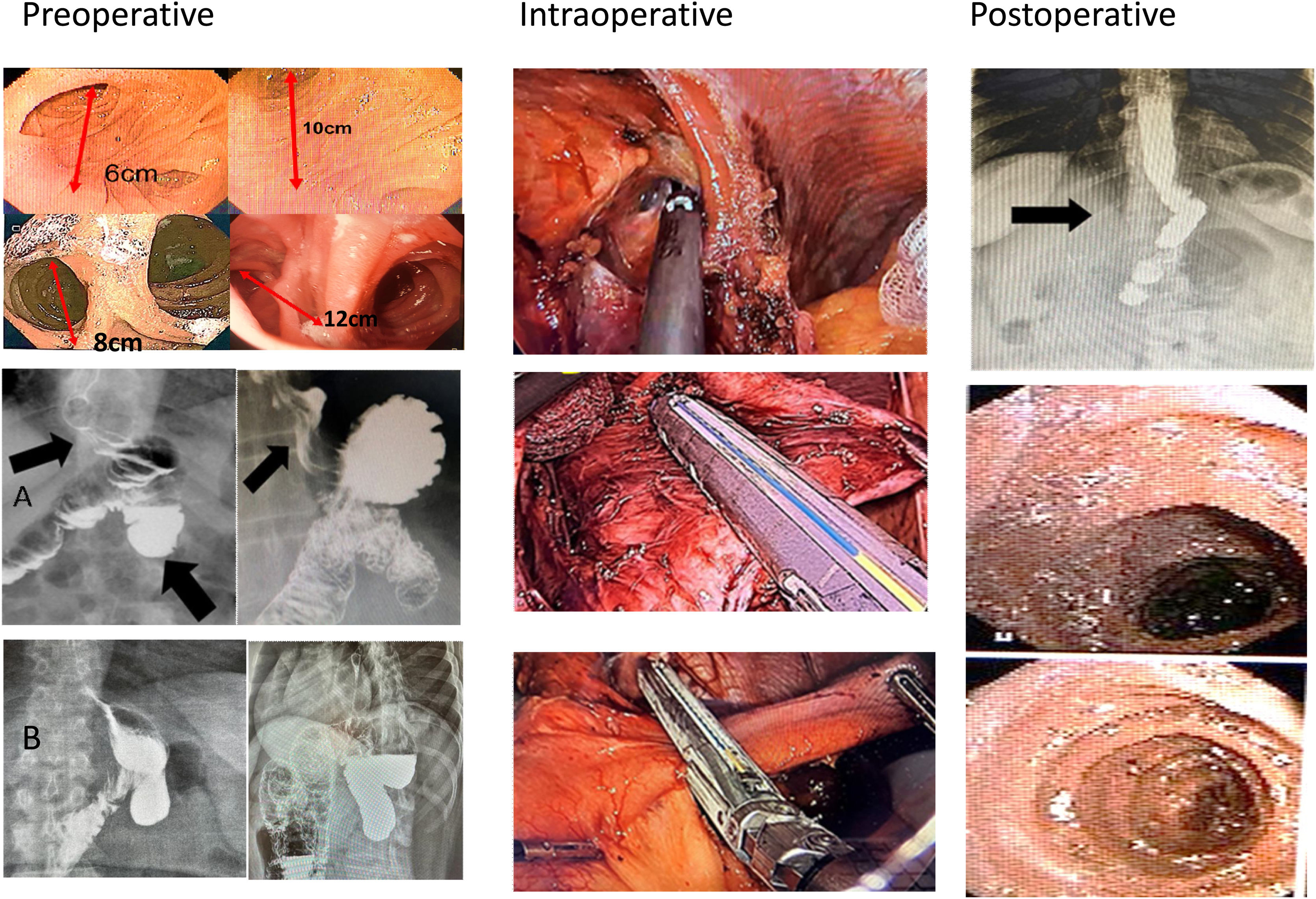
Candy cane syndrome (CCS) is a rare complication of laparoscopic Roux-en-Y gastric bypass (LRYGB). It occurs due to redundancy in the blind loop at the gastro-jejunal anastomosis.
ObjectiveTo evaluate the type of symptoms, anatomic and functional findings, and outcome after treatment.
Material and methodsA prospective case series study was conducted between 2010 and 2022, including symptomatic patients with CCS after LRYGB. Symptoms were correlated with anatomic and functional findings. Big gastric pouch was defined if its size was >5 cm, and a long candy cane loop was diagnosed if its length was >5 cm. Due to failure of medical treatment, revision surgery (RS) was indicated for resection of the elongated blind jejunal loop, resizing the redundant gastric pouch and repairing the hiatal hernia repair (HH) when necessary.
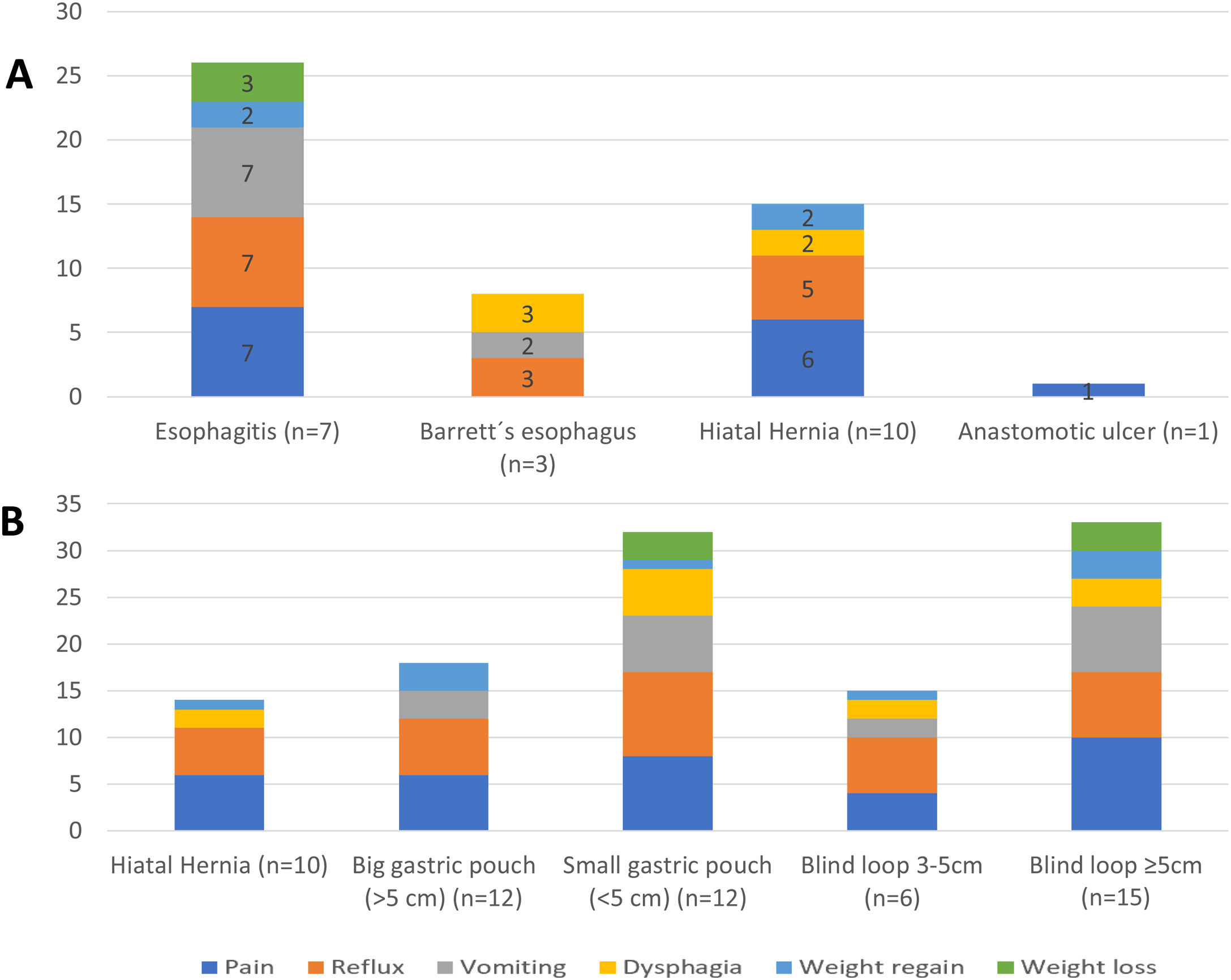
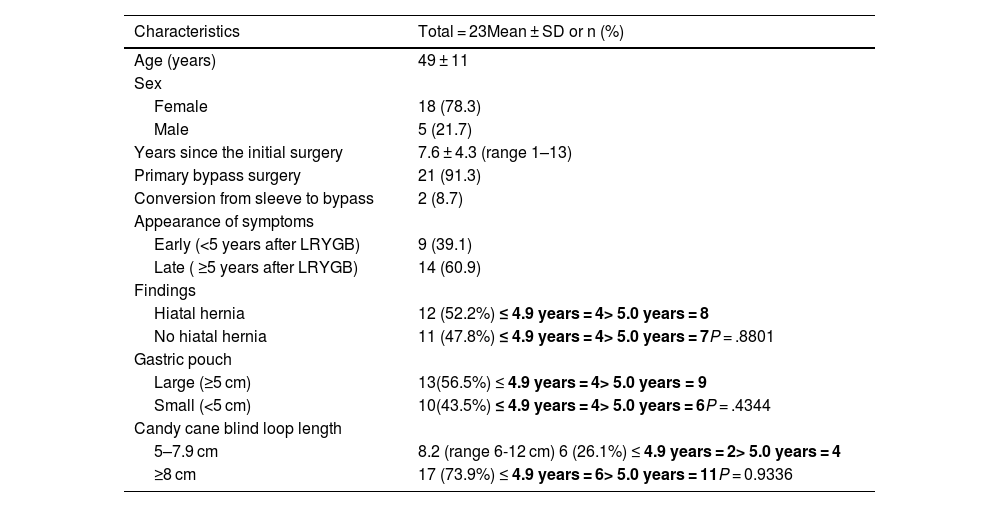
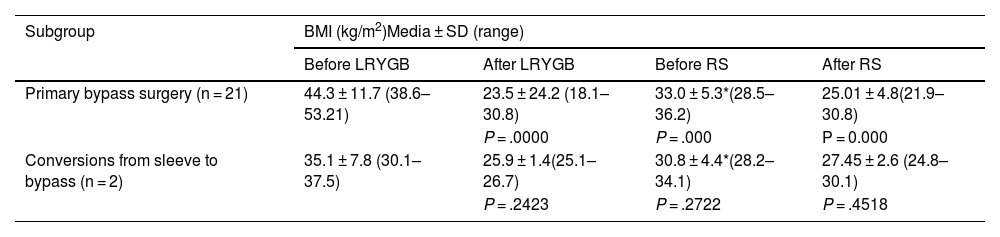
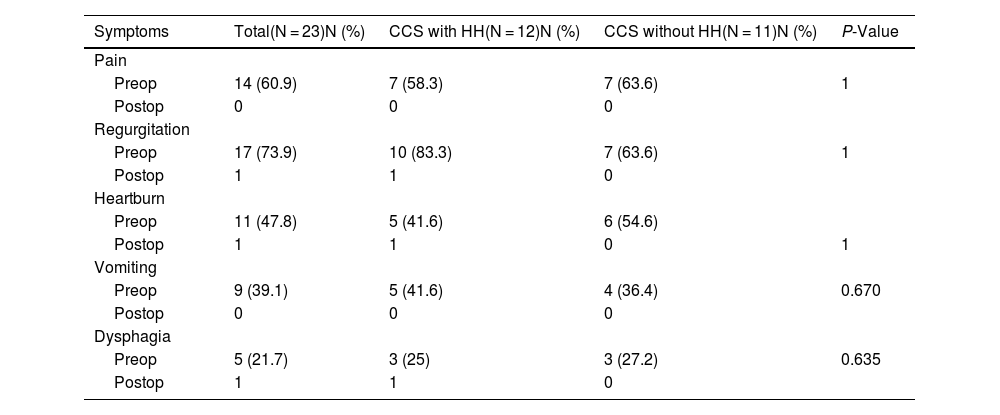
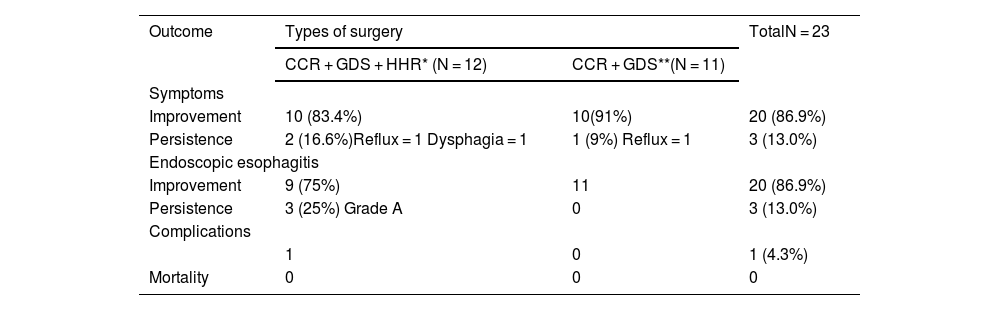
ResultsThe study included 23 patients, with a mean age of 49 ± 11 years. Twenty-one patients underwent primary LRYGB, and 2 were converted to this technique after sleeve gastrectomy (SG). The mean time from LRYGB to symptom onset was 7.6 ± 4.3 years. Pain and reflux symptoms were the most frequent, with no differences between patients with or without HH (P < .05). CCS coexisted with a large gastric pouch in 56.5% and HH in 52.2% of cases. A defective lower esophageal sphincter, abnormal esophageal motility, and pathological acid reflux test were observed. After surgery, improvement was observed in 86.9%.
ConclusionCCS can lead to gastrointestinal symptoms following LRYGB, regardless of the presence of HH. Complete examinations are crucial for diagnosis and to determine the surgical intervention, which is the best option for treatment.
El síndrome del bastón de caramelo (CCS) es una complicación poco común del bypass gástrico laparoscópico en Y de Roux (LRYGB). Ocurre debido a asa ciega redundante en la anastomosis gastroyeyunal.
ObjetivoEvaluar el tipo de síntomas, hallazgos anatómicos, funcionales y evolución post tratamiento.
Material y métodosEstudio prospectivo de serie de casos realizado entre 2010 y 2022, que incluyó pacientes sintomáticos con CCS tras LRYGB. Los síntomas se correlacionaron con los hallazgos anatómicos y funcionales. Se definió bolsa gástrica grande si su tamaño era >5 cm y se diagnosticó CCS si su longitud era >5 cm. Debido al fracaso del tratamiento médico, se indicó cirugía de revisión (RS) para resección del asa yeyunal ciega elongada, eventualmente redimensionamiento de la bolsa gástrica redundante y reparación de la hernia de hiato si corresponde.
ResultadosSe incluyeron 23 pacientes, edad media de 49 ± 11 años. Veintiún pacientes se sometieron a LRYGB primario y dos se convirtieron a esta técnica después de una gastrectomía en manga (SG). El tiempo medio desde LRYGB hasta la aparición de los síntomas fue de 7,6 ± 4,3 años. Los síntomas de dolor y reflujo fueron los más frecuentes, sin diferencias entre pacientes con o sin HH (p < 0,05). El CCS en concomitancia con bolsa gástrica grande ocurrió en el 56,5% y la HH en el 52,2% de los casos. Se observó un esfínter esofágico inferior hipotensivo, motilidad esofágica inefectiva y reflujo ácido patológico. Después de la cirugía se observó mejoría en el 86,9%.
ConclusiónEl CC puede provocar síntomas gastrointestinales después de LRYGB independiente de la existencia o no de HH. Los exámenes completos son cruciales para diagnosticar y seleccionar las intervenciones quirúrgicas, la mejor opción de tratamiento.