Minimize exposure to the SARS-CoV-2, reduce the chances of cross-transmission between patients and healthcare personnel, and prevent the development of postoperative complications from the management of patients with eye diseases during the 2019 coronavirus disease pandemic (COVID-19).
MethodsCOVID-19 literature review and consensus establishment between different Spanish ophthalmology societies in order to provide guidelines and recommendations of maximum resources primarily conditioned by the state of alert, confinement and social distancing that occurs in Spain since March 16, 2020.
ResultsThe recommendations will promote the adoption of action and protection measures for eye care in outpatient clinics, surgical areas and hospitalization, for unconfirmed (asymptomatic and symptomatic) and confirmed COVID-19 patients. Measures must be adapted to the circumstances and availability of personal protective equipment in each of the centers and Autonomous Communities, which will be updated according to the pandemic phases and the measures adopted by the Spanish Government.
ConclusionsDuring the COVID-19 pandemic, attention to the potential health risks to the population caused by coronavirus should prevail over the possible progression of the common eye diseases. Ophthalmologists and other eye care professionals must assume a possible progression of these diseases due to the impossibility of adequate patient follow-up.
Minimizar la exposición al virus SARS-CoV-2, reducir las posibilidades de transmisión cruzada entre pacientes y personal sanitario, y evitar el desarrollo de complicaciones postoperatorias por la atención a pacientes con enfermedades oculares durante la pandemia de enfermedad por coronavirus 2019 (COVID-19).
MétodosElaboración de un documento de revisión del estado del conocimiento sobre COVID-19 y consenso entre diferentes sociedades oftalmológicas españolas y afines, al objeto de proporcionar guías y recomendaciones de máximos recursos primariamente condicionadas por el estado de alerta, confinamiento y distanciamiento social que acontece en España desde el 16 de marzo de 2020.
ResultadosLas recomendaciones promoverán la adopción de medidas de actuación y protección para el desarrollo de la actividad asistencial en consultas externas, área quirúrgica y hospitalización, tanto para pacientes no confirmados –asintomáticos y sintomáticos– como confirmados de COVID-19. Deberán ser adaptadas a las circunstancias y disponibilidad de equipos de protección individual en cada uno de los centros y Comunidades Autónomas, debiendo ser actualizadas en función de las fases de la pandemia y de las medidas que adopte el Gobierno de la nación.
ConclusionesDurante la pandemia COVID-19, la atención a los potenciales riesgos de salud para la población ocasionados por el coronavirus deberá prevalecer sobre la posible progresión de enfermedades oculares comunes. Tanto médicos oftalmólogos como restantes profesionales de la salud ocular deberán asumir una posible progresión de dichas enfermedades ante la imposibilidad de un seguimiento adecuado de los pacientes.
This document is the result of a review of the state of the art and the consensus between national ophthalmological societies. It summarizes recommendations for maximum care of ophthalmological patients, both positive or negative for COVID, during the pandemic caused by the coronavirus SARS-CoV-2. Said recommendations should be adapted to the circumstances and availability of equipment in each center and community and should be updated according to the development of the pandemic in the coming weeks. Once the state of alarm is canceled said recommendations will no longer be applicable.
During said period, priority should be given to the potential health risks for the general population caused by the COVID-19 pandemic over the possible progression of diseases such as chronic glaucoma, diabetic retinopathy, age-related macular degeneration and corneal and inflammatory diseases, among others. A possible progression of said diseases should be assumed due to the impossibility of providing adequate follow-up to our patients.
Reason for the update: scientific evidenceSymptomatic COVID-19 patients frequently exhibit respiratory symptoms including fever, cough and dyspnea. Other symptoms include rash, myalgia, gastrointestinal disorders and loss of taste and smell. Conjunctivitis has also been reported. Severe complications include pneumonia and acute respiratory distress syndrome. The incubation period of the disease can range between 2 and 24 days during which any infected patient may remain asymptomatic and contagious.1–4
In addition, SARS-CoV-2 is transmitted through aerosols or respiratory droplets on the conjunctiva,5–7 with conjunctivitis being a possible first expression of systemic infection.8 A study published in the Journal of Medical Virology comprising 30 patients hospitalized due to COVID-19 in China reported that only one exhibited conjunctivitis and the ocular secretions of this patient included SARS-CoV-2. Accordingly, SARS-CoV-2 can infect the conjunctiva and produce conjunctivitis, with virus particles remaining in ocular secretions. A larger study published in the New England Journal of Medicine, researchers documented “conjunctival congestion” in 9 out of 1099 patients (0.8%) with COVID-19, confirmed in laboratories of 30 hospitals in China.1,5–8
Due to the high transmissibility of SARS-CoV-2, it is necessary to maintain a safety distance over 1.5m to diminish contagion risk.9 Ophthalmological examinations conducted at a distance of 30cm with slit lamp, or even closer in some specific maneuvers such as intraocular pressure (IOP) measurements or direct ophthalmoscopy, should be considered as a risk and managed in accordance with the standard procedures for these situations.4,7,8,10,11
In the light of the above evidence, ophthalmologists and ophthalmological nurses and assistants as well as opticians and optometrists are extremely vulnerable during patient examinations. In fact, ophthalmology is one of the medical specialties that has been highly affected by the current pandemic.1 On the other hand, due to high performance consultations, patients frequently spend over 1.5h in the practice. In addition, the mean age of patients is high and it is precisely the elderly population that exhibits greater mortality and morbidity in SARS-CoV-2 infections. Lastly, health professionals could become transmission agents of the disease on a risk population and therefore should strictly adhere to preventive measures.
Patients at risk: proposed actionsAs stated above, COVID-19 is transmissible through conjunctival secretions and conjunctivitis can exhibit different symptoms. According to published series, between 0.9 and 31.6% of patients exhibit said disease and this increases the probability that ophthalmologists will be the first health professionals to examine patients potentially affected by SARS-CoV-2. Due to close contact with the patient through slit lamps, ophthalmoscopies, tonometers and similar equipment requiring a distance under 1m, the following measures must be adopted:
- 1.
All patients visiting the practice should be adequately assessed by means of triage before entering the waiting room to identify the following conditions to determine possible exposure to SARS-CoV-2:
- •
Does the patient have fever, general discomfort, breathing symptoms (dry cough, dyspnea), ageusia, anosmia, diarrhea or headache?
- •
Does the patient exhibit symptoms of conjunctivitis such as red eye, secretion, itching, ocular irritation or acute photophobia?
- •
Does the patient live or has lived with a relative or close partners that have been diagnosed or are in quarantine due to COVID-19 in the past 14 days?
- •
- 2.
Only patients with urgent ocular pathology in the absence of coronavirus infection symptoms should be examined. Patients with suspected or confirmed positive symptoms of the coronavirus that visit due to genuine ocular emergencies such as ocular perforation or acute glaucoma shall be referred to the Emergency Departments of reference hospitals (section A2, a).
- 3.
Waiting rooms with only one companion if necessary and observing a distance of 2m between individuals. Avoids the accumulation of patients in waiting rooms or procedure rooms.
- 4.
Ophthalmological examination (seeing General Recommendations A1 and A2).
- 5.
Desinfection of all surfaces of units, lenses and devices (appendix A in additional material).
- 1.
All non-urgent activity must be postponed.1 Whenever possible, remote telematics consultations should be prioritized (telephone voice call or video conference) and patients must be rescheduled ensuring that personal consultations can be conducted safely after the first phase of the crisis.1,7,8 The ophthalmological and systemic risk/benefit ratio of patients should be carefully considered in all cases. The following link shows a stratification of ophthalmological risk according to pathology groups developed by the Moorfields Eye Hospital NHS Foundation Trust12 (https://www.eurotimes.org/wp-content/uploads/2020/03/Moorfields-Ophthalmology-Risk-Stratification-Version-2.pdf). It is recommended to obtain an informed consent for remote medical care, which can be previously sent to the patient, a relative or caregiver through email and returned signed or otherwise accepted by means of voice recordings. In the event of difficulties related to the reception of said informed consent, it could be provided verbally and registered in the patient record. Appendix B (in the additional material) shows the informed consent model developed by the Ophthalmological Society of Spain and validated by the Ethical Committee thereof. Telephone consultations for children are particularly relevant due to the anxiety that the current situation could produce in parents, and this type of consultation could avoid unnecessary visits to Emergency Departments. Personal consultations should be conducted only in the following cases:
- a.
Urgent consultations and reviews for the same reason.
- b.
First and second reviews of urgent surgical procedures.
- c.
Consultations for urgent or not deferrable intravitreal injections, checking patient profiles in the agendas, establishing telephone communications for an individual assessment of each case (i.e., single eyes, Treat & Extend age-related macular degeneration with 4-week intervals or patients in loading dose).
- d.
Patients with glaucoma: in cases with a tendency to verify progression or with severe damage, increasing anti-glaucoma treatments during this period could be ethically reasonable while awaiting an adequate ophthalmological examination. Remote e-prescription is a crucial tool for any therapeutic change. Anamnesis conducted over the telephone will enable the detection of possible intolerances to treatment and to implement timely changes.
- e.
In patients with noninfectious uveitis in treatment with systemic immunomodulating and immunosuppressant therapies, the following recommendations should be taken into account:
- i.
Telephone communication to determine the condition of the patient and, if required by the latter, scheduling a personal consultation to perform essential supplementary tests (including analytic control of possible medication side effects).
- ii.
Recommending the patient that if infection by SARS-CoV-2 is suspected or confirmed, medication must be interrupted and treating physicians must be advised about the type of medication being used. Consult the consensus document of the Ocular Inflammation Society of Spain to determine possible exceptions to this recommendation13 (https://seioc-uveitis.com/wp-content/uploads/2020/04/DOCUMENTO-DE-CONSENSO-SEIOC-PACIENTES-UVEÍTIS-NO-INFECCIOSAS-EN-TRATAMIENTO-TERAPIA-INMUNOMODULADORA-INMUNOSUPRESORA-EN-PADEMIA.pdf).
- iii.
If necessary, local treatments could be a good alternative for selected cases as described in the previous point.
- iv.
For patients without confirmed SARS-CoV-2 infection or symptoms suggesting said disease, it is recommended to maintain medication, verifying the drug, dosage, pathologies and concomitant treatments.
- v.
Use of tocilizumab (TCZ): TCZ is a therapeutic option that could be considered as a third line in patients with non-infectious uveitis after conventional immunosuppressants and anti-TNF biological agents. Following the recommendations of the Spanish Agency of Medicaments and Health Products (AEMPS)14 to reserve intravenous TCZ for patients with COVID-19, and in the specific case of patients with noninfectious uveitis in treatment with intravenous TCZ, the most reasonable alternative would be the administration of subcutaneous TCZ with a syringe or pre-loaded tip.
- 2.
Placing information panels on the walls of waiting rooms, explaining the hygiene-related recommendations of the Health Ministry And Labor Risk Prevention Services, as well as notices to refrain from staying in waiting rooms if exhibiting fever or respiratory difficulties. Screens and panels can be used for displaying this information.
- 3.
Apply extreme cleanliness in all consulting and waiting rooms, including doorknobs, administrative areas, receptions, telephones, computer keyboards and mouse, etc.
- 4.
Patients should enter the practice preferably on their own. In exceptional circumstances they may be accompanied by one person only.
- 5.
All patients must wear surgical masks and use hydroalcohol gel dispensers in the waiting rooms. As regards children, parents should be asked to maintain the mask on during the entire examination.
- 6.
Patients and companions must wash their hands with hydroalcohol gel before entering and leaving the practice.
- 7.
FFP2 (N-95) or FFP3 masks and protective gloves and goggles must be used during examinations.
- 8.
Hands must be washed with water and soap or hydroalcohol gel before and after seeing each patient.
- 9.
Head and chin supports on slit lamps and other examination equipment must be cleaned with the antiseptic solutions routinely used for disinfection, as well as control levers and buttons before and after seeing each patient.
- 10.
Patient care: only urgent cases shall be received.1 Patients shall be classified into groups, i.e., unconfirmed patients (asymptomatic or symptomatic) or patients confirmed with positive test results for SARS-CoV-2.
All cases should be considered as potentially contagious, applying the above described sequence of actions and general recommendations. In addition, the following shall be taken into account:
- a.
Use of plastic or methacrylate protective visors in slit lamps, laser devices, OCT, etc. due to the persistence of the coronavirus on plastic surfaces, these must be cleaned on both sides with some disinfectant such as alcohol or sodium hypochloride after every use.
- b.
Explorations shall be limited to the minimum.
- c.
Explicit instructions for the patient as well as the ophthalmologist to refrain from speaking as much as possible during the procedure, particularly while using slit lamp. As much as possible, maintain the safety distance at all times.
- d.
Explicit instructions to the patient to avoid from touching any material in the practice unless unavoidable (slit lamp, etc.). If the patient is a child accompanied by parents, inform them of this restriction.
- e.
Do not use occluding devices or stenopeic holes for measuring visual acuity.
- f.
Tonometry: pneumotonometry must not be carried out because the intense force of air could produce aerosol of teardrops, expelling them into the environment. It is recommended to use devices that enable the use of disposable protective terminals such as Tonopen® (Reichert technologies, NY, USA), Icare® (Icare, Helsinki, Finland) or Goldmann tonometer. If disposable terminals are not available, apply the recommended disinfection methods for each device. Perkins-type applanation tonometers must not the utilized due to the extreme proximity to patients involved in its use, with the exception of cases listed in point 12 of the general recommendations below.
- g.
Avoid diagnostic procedures requiring contact with the cornea. In absolutely necessary cases, disinfection must be carried out following the instructions of the Preventive Medicine Dept.
- h.
Eyedrops shall be administered with gloves. Preferably, single-dose eyedrops shall be utilized. Multidose eyedrops shall be discarded after being administered to each patient.
- i.
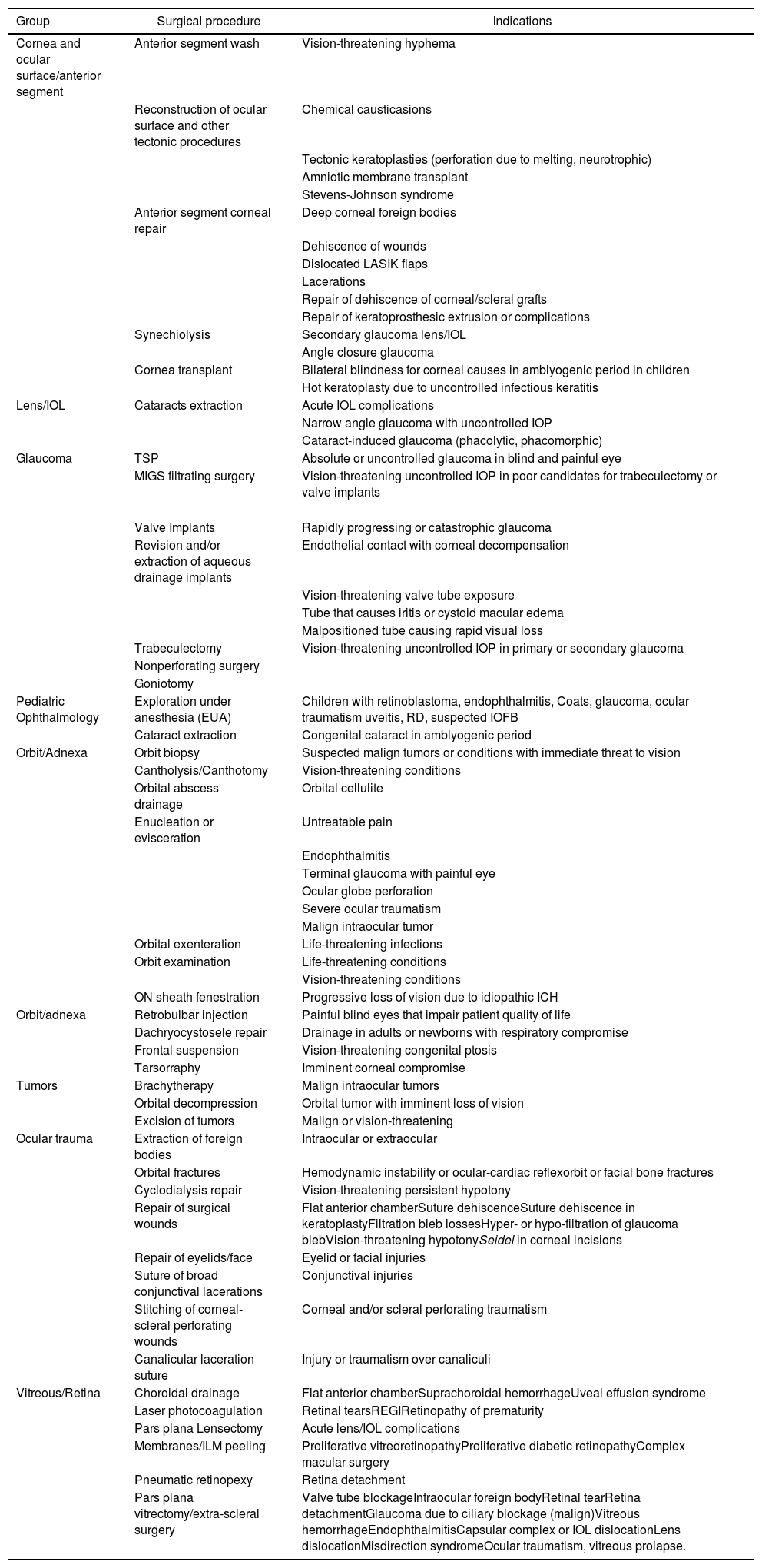
Surgery with general anesthesia shall be conducted only when strictly necessary1–4,8–11 on an urgency and preferential basis, i.e., retina detachments, pars plana vitrectomy in endophthalmitis, urgent glaucoma or ocular perforations. A complete list of urgent procedures is shown in Table 1.
Table 1.Urgent ophthalmological procedures during the COVID-19 pandemic.
Group Surgical procedure Indications Cornea and ocular surface/anterior segment Anterior segment wash Vision-threatening hyphema Reconstruction of ocular surface and other tectonic procedures Chemical causticasions Tectonic keratoplasties (perforation due to melting, neurotrophic) Amniotic membrane transplant Stevens-Johnson syndrome Anterior segment corneal repair Deep corneal foreign bodies Dehiscence of wounds Dislocated LASIK flaps Lacerations Repair of dehiscence of corneal/scleral grafts Repair of keratoprosthesic extrusion or complications Synechiolysis Secondary glaucoma lens/IOL Angle closure glaucoma Cornea transplant Bilateral blindness for corneal causes in amblyogenic period in children Hot keratoplasty due to uncontrolled infectious keratitis Lens/IOL Cataracts extraction Acute IOL complications Narrow angle glaucoma with uncontrolled IOP Cataract-induced glaucoma (phacolytic, phacomorphic) Glaucoma TSP Absolute or uncontrolled glaucoma in blind and painful eye MIGS filtrating surgery Vision-threatening uncontrolled IOP in poor candidates for trabeculectomy or valve implants Valve Implants Rapidly progressing or catastrophic glaucoma Revision and/or extraction of aqueous drainage implants Endothelial contact with corneal decompensation Vision-threatening valve tube exposure Tube that causes iritis or cystoid macular edema Malpositioned tube causing rapid visual loss Trabeculectomy Vision-threatening uncontrolled IOP in primary or secondary glaucoma Nonperforating surgery Goniotomy Pediatric Ophthalmology Exploration under anesthesia (EUA) Children with retinoblastoma, endophthalmitis, Coats, glaucoma, ocular traumatism uveitis, RD, suspected IOFB Cataract extraction Congenital cataract in amblyogenic period Orbit/Adnexa Orbit biopsy Suspected malign tumors or conditions with immediate threat to vision Cantholysis/Canthotomy Vision-threatening conditions Orbital abscess drainage Orbital cellulite Enucleation or evisceration Untreatable pain Endophthalmitis Terminal glaucoma with painful eye Ocular globe perforation Severe ocular traumatism Malign intraocular tumor Orbital exenteration Life-threatening infections Orbit examination Life-threatening conditions Vision-threatening conditions ON sheath fenestration Progressive loss of vision due to idiopathic ICH Orbit/adnexa Retrobulbar injection Painful blind eyes that impair patient quality of life Dachryocystosele repair Drainage in adults or newborns with respiratory compromise Frontal suspension Vision-threatening congenital ptosis Tarsorraphy Imminent corneal compromise Tumors Brachytherapy Malign intraocular tumors Orbital decompression Orbital tumor with imminent loss of vision Excision of tumors Malign or vision-threatening Ocular trauma Extraction of foreign bodies Intraocular or extraocular Orbital fractures Hemodynamic instability or ocular-cardiac reflexorbit or facial bone fractures Cyclodialysis repair Vision-threatening persistent hypotony Repair of surgical wounds Flat anterior chamberSuture dehiscenceSuture dehiscence in keratoplastyFiltration bleb lossesHyper- or hypo-filtration of glaucoma blebVision-threatening hypotonySeidel in corneal incisions Repair of eyelids/face Eyelid or facial injuries Suture of broad conjunctival lacerations Conjunctival injuries Stitching of corneal-scleral perforating wounds Corneal and/or scleral perforating traumatism Canalicular laceration suture Injury or traumatism over canaliculi Vitreous/Retina Choroidal drainage Flat anterior chamberSuprachoroidal hemorrhageUveal effusion syndrome Laser photocoagulation Retinal tearsREGIRetinopathy of prematurity Pars plana Lensectomy Acute lens/IOL complications Membranes/ILM peeling Proliferative vitreoretinopathyProliferative diabetic retinopathyComplex macular surgery Pneumatic retinopexy Retina detachment Pars plana vitrectomy/extra-scleral surgery Valve tube blockageIntraocular foreign bodyRetinal tearRetina detachmentGlaucoma due to ciliary blockage (malign)Vitreous hemorrhageEndophthalmitisCapsular complex or IOL dislocationLens dislocationMisdirection syndromeOcular traumatism, vitreous prolapse. IOFB: intraocular foreign body; TSP: trans-scleral cyclophotocoagulation; COVID-19: coronavirus disease 2019; RD: retina detachment; EUA: examination under anesthesia; ICH: intracranial hypertension; REGI: repeated expandable gas injections; LASIK: laser assisted in situ keratomileusis; IOL: intraocular lens; MIGS: minimally invasive glaucoma surgery; ILM: inner limiting membrane; ON: optic nerve; IOP: intraocular pressure.
Adapted from the American Academy of Ophthalmology.15
- j.
Avoid lacrimal pathway examinations unless strictly necessary. Should it be necessary to remove a silicone guide out of the nasal pathway, it shall be conducted with FFP2/FFP3 facial mask and visor.
- a.
These patients must not be seen by the ophthalmologist before being examined by the Emergency Department according to the protocol of each hospital.
- i.
Peripheral centers: these patients shall be referred to public reference hospitals and received at the specific areas equipped with all individual protection equipment (IPE) utilized by personnel trained in the use thereof.
- ii.
Reference hospitals: specific consultation rooms for Covid-19 positive patients must be set up7 in a different area separated from the rest of ophthalmological consultation rooms, applying the specific measures for full IPE (FFP2/FFP3 masks, gloves, shoe and head covers, impervious aprons and facial goggles or visors) as per the Preventive Medicine guidelines of each hospital.
- i.
- b.
Multidose eyedrops utilized for a patient must be discarded.
- c.
Special care must be taken with patients exhibiting conjunctivitis to avoid contact with the conjunctival mucosa and secretions.
- 11.
It is recommended to postpone all scheduled surgeries. Only urgent and not deferable procedures shall be conducted when associated to irreversible loss of vision in the short term. Some examples include retina detachment surgeries, oncological procedures, uncontrolled high pressure glaucoma, retinopathy of prematurity and genuine ocular emergencies (traumatism, perforating wounds, intraocular foreign bodies).7,8,11Table 1 illustrates the full range of urgent ophthalmological procedures.15General recommendations for all urgent operations to be carried out are:
- a.
A clinic checklist must be conducted prior to interventions to determine the possible existence of COVID-19 symptoms of positive epidemiological contacts.
- b.
A supplementary informed consent must be provided for surgery during the COVID-19 pandemic in addition to the specific consent for the procedure to be conducted.
- c.
Cardio-respiratory auscultation (anestesiologist) and body temperature must be taken at admission.
- d.
Preoperative analytics with COVID risk indication parameters. Electrocardiogram (EKG). Chest x-ray and thorax CAT to be assessed by the anesthesiologist.
- e.
It is recommended to carry out the polymerase chain reaction (PCR) for the SARS-CoV-2 virus on all patients that will undergo surgery. PCR must be included in the surgical checklist. In the emergency situation does not allow this, the patient must be considered as suspected positive.
- f.
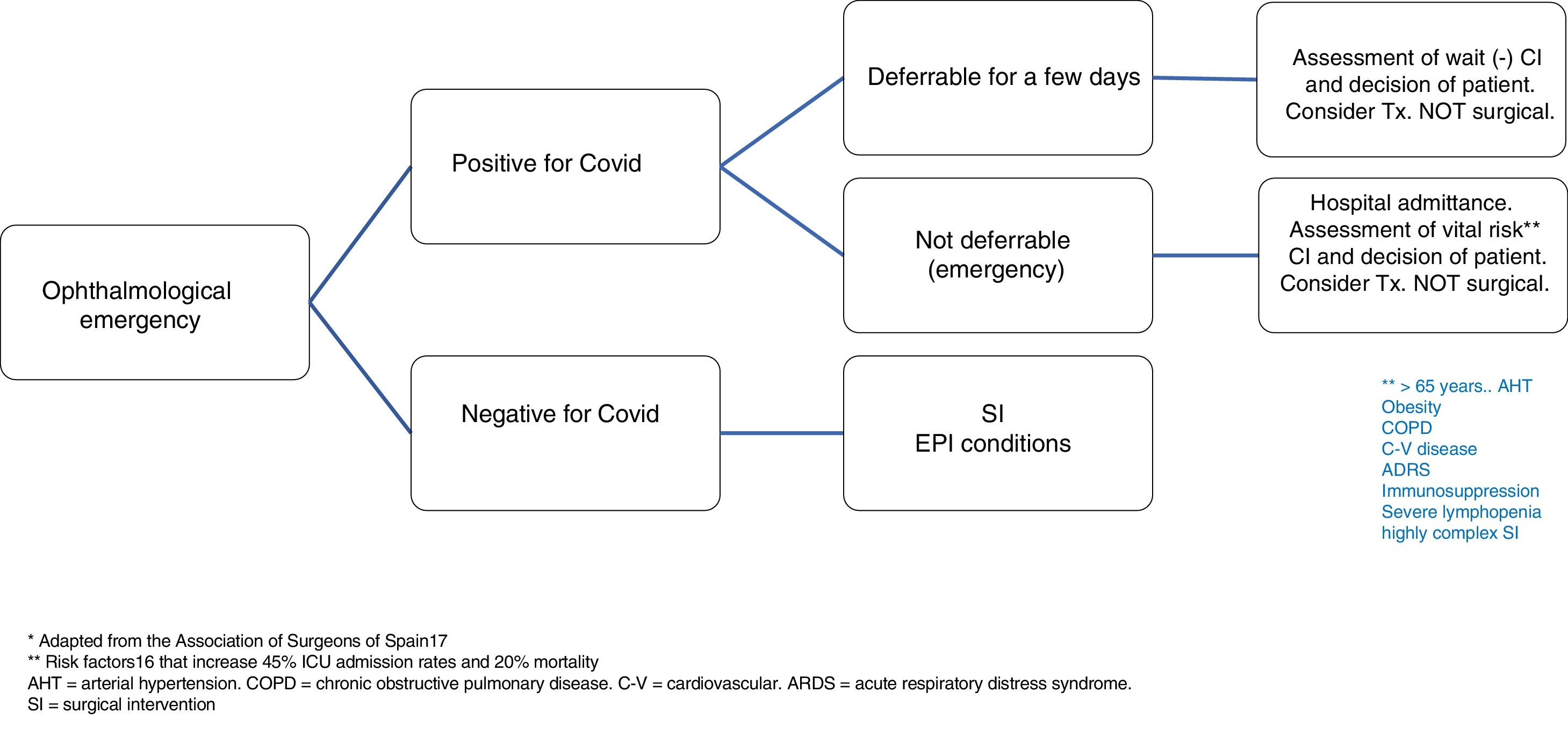
In the case of patients with confirmed COVID-19 diseases that require emergency surgery, the algorithm shown in Fig. 1 should be adopted.
- i.
If the emergency allows it, surgery should be deferred as much as possible, ideally until PCR is negative. Vital risk should always be evaluated against the ocular benefit together with the patient.
- ii.
If the emergency does not allow deferring surgery (for instance, ocular perforation), vital risk must be assessed above all in patients over 65 years of age or those who exhibit hypertension, obesity, chronic bronchial or lung pathology, cardiovascular disease, acute respiratory distress syndrome, immunosuppression, severe lymphopenia or if the surgery is highly complex. In these circumstances, a recent study conducted in China by Lei et al.16 reported very high ICU admission and mortality rates of approximately 45 and 20%, respectively.
- i.
- g.
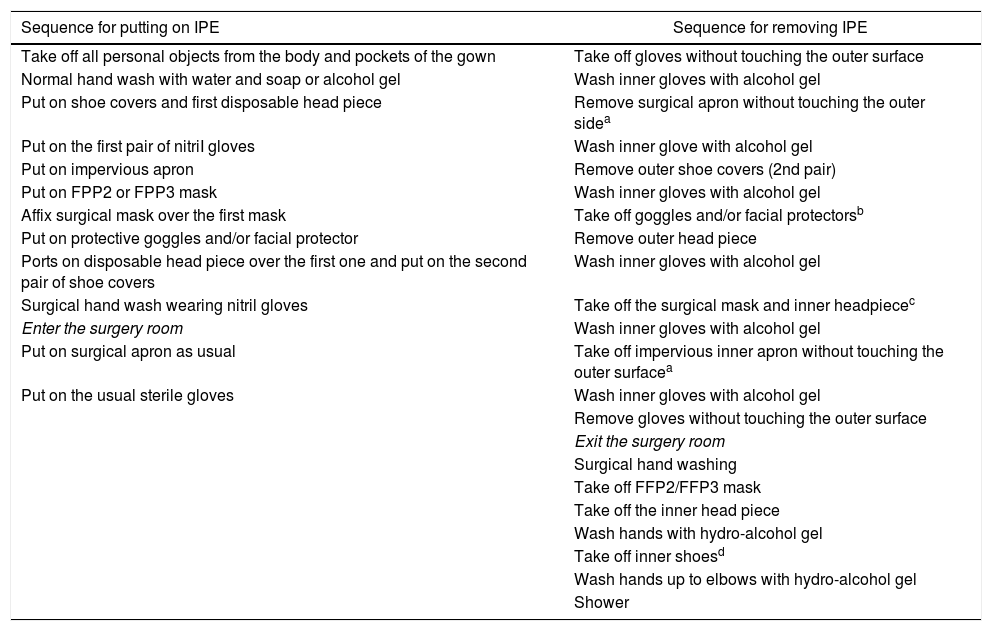
Adequate protection measures must be utilized, above all in A1 symptomatic and A2 patients, comprising impervious aprons, disposable head and shoe covers, facial protection with goggles or disposable plastic visors, protection mask (FFP2 or equivalent to N-95 or FFP3) and double gloves. Table 2 describes the instructions for using and withdrawing IPE17.
Table 2.Instructions for putting on and removing IPE in the surgery room.
Sequence for putting on IPE Sequence for removing IPE Take off all personal objects from the body and pockets of the gown Take off gloves without touching the outer surface Normal hand wash with water and soap or alcohol gel Wash inner gloves with alcohol gel Put on shoe covers and first disposable head piece Remove surgical apron without touching the outer sidea Put on the first pair of nitriI gloves Wash inner glove with alcohol gel Put on impervious apron Remove outer shoe covers (2nd pair) Put on FPP2 or FPP3 mask Wash inner gloves with alcohol gel Affix surgical mask over the first mask Take off goggles and/or facial protectorsb Put on protective goggles and/or facial protector Remove outer head piece Ports on disposable head piece over the first one and put on the second pair of shoe covers Wash inner gloves with alcohol gel Surgical hand wash wearing nitril gloves Take off the surgical mask and inner headpiecec Enter the surgery room Wash inner gloves with alcohol gel Put on surgical apron as usual Take off impervious inner apron without touching the outer surfacea Put on the usual sterile gloves Wash inner gloves with alcohol gel Remove gloves without touching the outer surface Exit the surgery room Surgical hand washing Take off FFP2/FFP3 mask Take off the inner head piece Wash hands with hydro-alcohol gel Take off inner shoesd Wash hands up to elbows with hydro-alcohol gel Shower Adapted from Balibrea et al.17
- h.
Only indispensable personnel must remain in the operating room.
- i.
Circulation of personnel in the operating room must be diminished as much as possible.
- j.
The surgical technique must be performed by highly experienced surgeons utilizing the shortest possible execution time.
- k.
Whenever possible, avoid general anesthesia.
- l.
Avoid surgical maneuvers that could disseminate airborne particles such as drills, electrical cutting and coagulation systems, ocular surface irrigation with serum, which should be substituted by viscoelastic.
- m.
Utilize trocars equipped with valves in vitreoretinal surgery and apply viscoelastic in the valves to minimize the dissemination of particles and aerosols in air exchanges.
- n.
As far as possible and only for COVID-negative patients, surgical procedures must be ambulatory to avoid hospital stays. Otherwise, the hospital must be equipped with non-COVID areas to avoid nosocomial infections.
- 12.
Interconsultations related to hospitalized patients must be limited to urgent cases only. The requesting department shall be contacted over the phone to verify said urgency. Non-urgent cases shall be given appointments at least 14 days after release. If consultation is necessary, it shall be conducted in the patient room with portable slit lamp, Perkins-type tonometer, Icare® (Icare, Helsinki, Finland) or Tonopen® (Reichert technologies, NY, USA) and indirect binocular ophthalmoscopy. Due to the excessive proximity required with Perkins-type applanation tonometry, it should be used only in exceptional circumstances that unavoidably require IOP control and in the absence of other portable contact tonometer types. For the same reason, direct ophthalmoscopy shall not be utilized for visualizing the ocular fundus. Said materials shall be cleaned by personnel after each use.7
- 13.
Trasplants: transplants deserve special consideration. It is recommended to perform only extremely urgent transplant. It would be crucial for donors to follow the recommendations of the BV-ES-20200122-5 document dated March 13, section 2 on “Donation of Tissue”: “in what concerns tissue donation, at the present time any confirmed COVID-19 case should be discarded. In the rest of cases and in the absence of a negative result 24 hours prior to the donation, tissue establishments are recommended to screen for SARS-CoV-2. In order to facilitate said screening at the tissue bank, the samples obtained for organ donor screening must be obtained (if possible, samples of the upper airway pathway). Positive or inconclusive results involve the cancellation of tissue donation”.
To date there is no evidence that healthy individuals should avoid the use of contact lenses or that contact lens users are at higher risk of coronavirus infection in comparison with spectacles users. Similarly, there is no evidence suggesting a correlation between the use of contact lenses and the propagation of COVID-19 or coronavirus infection related to the use of contact lenses. However, it is known that the SARS-CoV-2 virus can isolate in tears and conjunctiva which act as a transmission medium.
For the above reason and due to the exceptional nature of the current situation, it is advisable to refrain from accepting any type of contact lens excepting cases with irregular corneas where other correction means cannot be applied as well as for some users of specific contact lens types for controlling the evolution of myopia. If the patient is already a contact lens user and exhibits ocular symptoms (conjunctival congestion, secretion, tearing) on general symptoms such as fever, dry cough, general discomfort, use of contact lenses should be suspended and replaced by spectacles. In any doubt, said patients should consult with their contact lens specialist.
Contact lenses shall be withdrawn from children unless exceptional circumstances make their use essential, such as aphakia or keratocone. In said cases, contact lenses shall be utilized with the indicated hygienic and disinfection measures unless exhibiting signs or symptoms indicating an ocular process that renders their use incompatible. Measures for adequate contact lens use would be the following:
- a.
Disinfection of surfaces in order to avoid contamination of that anything related to the use and maintenance of contact lenses, such as lens cases and solutions due to contact with previously contaminated surfaces.
- b.
According to the WHO recommendations18 (https://www.who.int/gpsc/information_centre/gpsc_lavarse_manos_poster_es.pdf?ua=1) hands must be washed before and after inserting and extracting contact lenses as well as the manipulation thereof and lens cases in order to avoid two-way contamination.
- c.
In what concerns soft daily contact lenses, handwashing precautions must be observed and used lenses discarded in isolated containers to avoid microbiological contamination, avoiding disposal in waste bins for plastics.
- d.
When utilizing non-disposable contact lenses such as those made of hydrogel, silicone hydrogel, rigid, hybrid, scleral or semi-scleral, in daily use or every time they are withdrawn from the eye, the following maintenance protocol must be carried out.
- 1.
Wash hands.
- 2.
Remove the contact lens.
- 3.
Clean with the adequate cleaner recommended by the ophthalmologist, applying mechanical friction.
- 4.
Rinse with sterile saline solution.
- 5.
Disinfect with 3% hydrogen peroxide, the only solution with scientific evidence19 on efficacy against coronavirus (https://www.cdc.gov/coronavirus/2019-ncov/faq.html#How-to-Protect-Yourself).
- 6.
Single-step hydrogen peroxide system for contact lenses shall be used, following the usage recommendations of the manufacturer.
- 7.
Contact lenses can be utilized after completing the disinfection procedure within the period of time established by the manufacturer, checking in each case the indications of the utilized the disinfection product.
- 8.
Wash hands before taking the lens to insert it onto the eye.
- 9.
Rinse with saline solution before placing the lens over the eye surface.
- 10.
Wash hands after inserting the lenses.
Specifications include the preparation of autologous serum and plasma-rich growth factors (PRGF). The preparation of hematic derivatives is recommended only in clinic situations that exclude any other options, such as neurotrophic ulcers stages II and III, and this treatment is necessary for the follow-up and clinic improvement of patients.
The precautions to be taken in the preparation of hematic derivatives shall be those established by the AEMPS report20 dated March 23, 2013, on the use of platelet-rich plasma (PRP) and taking into account the exclusion criteria of Appendix II of Royal Decree21 1088/2005 which specifies that patients with infections must be excluded during at least 2 weeks after complete clinical recovery from an infectious disease and after the disappearance of symptoms, including fever above 38°C and feverish symptoms in which we could classify the infection by SARS-CoV-2. In addition to the recommended serologies (VHB, VHC, VIH, HTLV-I, syphilis), the current health condition of the patient must be determined to take decisions on the basis of the above specifications.
As much as possible and in order to exempt the patient from frequent hospital visits it is recommended to prepare the highest number of eyedrops to cover their maximum established stability period as per the applied procedures.
Use of chloroquine/hydroxychloroquine in COVID-19 patients and macular retinal toxicityChloroquine and hydroxychloroquine are anti-malaria medicaments broadly used for treating a range of self-immune diseases. At present, there are no published clinic studies for both medicaments discussing their role in the management of SARS-CoV-2 infection,22 although there is a systematic review on the safety and efficacy of chloroquine for treating COVID-1923 based on its in vitro effectiveness to limit the replication of SARS-CoV-2.24 The results of the French cohort have been published, comparing the efficacy of treatment with hydroxychloroquine on its own (n=14) or associated to azithromycin (n=6) with 16 untreated patients, with positive results for the use of anti-malaria medicaments, although said study involved significant limitations. At present, the efficacy of said medicaments is being assessed in over 30 clinic trials, mainly conducted in China.22,23
In the light of the above, the Ministry of Health of Spain has designed a protocol for the management and treatment of patients infected by SARS-CoV-2 which is continuously updated. The protocol or recommends the use of base chloroquine for adults and hydroxychloroquine sulfate for adults and pediatric patients during 5 days with a loading dose of the first day. Reversible ocular disorders have been described after early treatment interruption22 but obviously there is no information of long-term ocular adverse events due to the short period of use of said recommended dose. Macular toxicity has been demonstrated in patients that use said medicaments for extended periods of time, with daily use being the most critical factor for said risk, which is correlated to actual body weight. In addition, patients in treatment with tamoxifen and those exhibiting renal insufficiency are at greater risk.25 Accordingly, treatments with a dosage of ≤5mg/kg of weight exhibit less than 1% of maculopathy risk after 5 years of treatment and less than 2% after 10 years of treatment.26
On the basis of the above data, the following recommendations are made:
- •
If possible, the patients must be informed before initiating treatment about the absence of scientific evidence on potential macular toxicity.
- •
The systemic benefit of the anti-malaria treatment must prevail over possible adverse ocular effects, and for this reason the presence of any type of retinopathy should not the considered as an absolute contraindication for the use of said medication for treating infections by COVID-19.
- •
The risk of retinopathy due to chloroquine or hydroxychloroquine at the dosage and duration of the treatment applied against COVID-19 is extremely low and does not justified screening tests for identifying maculopathies in these patients.27
None declared.
Antonio Piñero-Bustamante, Clínica Piñero, Sociedad Española de Oftalmología (SEO), Sevilla.
José Luis Encinas-Martín, Clínica Dr. Encinas, Sociedad Española de Oftalmología (SEO), Madrid.
José Manuel Benítez-del-Castillo Sánchez, Hospital Clínico San Carlos, Sociedad Española de Oftalmología (SEO), Madrid.
Julián García-Feijoo, Hospital Clínico San Carlos, Sociedad Española de Oftalmología (SEO), Madrid.
Marta Suárez-de-Figueroa Díez, Hospital Ramón y Cajal, Sociedad Española de Oftalmología (SEO), Madrid.
Pedro Arriola-Villalobos, Hospital Clínico San Carlos, Sociedad Española de Oftalmología (SEO).
Fernando Llovet-Osuna, Clínica Baviera, Sociedad Española de Oftalmología (SEO), Valencia.
Carmelina Brito-Muguerza, Hospital Vithas, Sociedad Española de Oftalmología (SEO), Santa Cruz de Tenerife.
José García Arumí, Instituto de Microcirugía Ocular, Sociedad Española de Retina y Vítreo (SERV), Barcelona.
Alfredo García-Layana, Clínica Universitaria de Navarra, Sociedad Española de Retina y Vítreo (SERV), Pamplona.
Juan Donate López, Hospital Clínico San Carlos, Sociedad Española de Retina y Vítreo (SERV), Madrid.
María Gómez-Resa, Hospital Universitario Son Espases, Sociedad Española de Retina y Vítreo (SERV), Palma de Mallorca.
Maximino Abraldes López-Veiga, Complexo Hospitalario Universitario de Santiago de Compostela, Sociedad Española de Retina y Vítreo (SERV), Santiago de Compostela.
Elena Rodríguez-Neila, Hospital Juan Ramón Jiménez, Sociedad Española de Retina y Vítreo (SERV), Huelva.
José Juan Escobar-Barranco, Hospital Dos de Maig, Sociedad Española de Retina y Vítreo (SERV), Barcelona
Ramón Lorente-Moore, Complexo Hospitalario Universitario de Ourense, Sociedad Española de Cirugía Ocular Implanto-Refractiva (SECOIR), Orense.
Javier Aritz Urcola, Hospital Universitario Araba, Sociedad Española de Glaucoma (SEG), Vitoria-Gasteiz.
Carlos Brito-Suárez, Sociedad Española de Contactología (SEC), Zaragoza.
Gonzalo Carracedo Rodríguez, Facultad de Óptica-Optometría, Universidad Complutense de Madrid (UCM), Madrid.
Alfredo Adán-Civera, Hospital Clinic, Sociedad Española de Inflamación Ocular (SEIOC), Barcelona.
Alejandro Fonollosa-Carduch, Hospital Universitario de Cruces, Sociedad Española de Inflamación Ocular (SEIOC), Bilbao.
David Díaz-Valle, Hospital Clínico San Carlos, Sociedad Española de Inflamación Ocular (SEIOC), Madrid.
Maite Sainz de la Maza, Hospital Clinic, Sociedad Española de Inflamación Ocular (SEIOC), Barcelona.
Jesús Merayo-Lloves, Instituto Oftalmológico Fernández-Vega, Sociedad Española de Superficie Ocular y Córnea (SESOC), Oviedo.
Pilar Gómez de Liaño Sánchez, Hospital Universitario Gregorio Marañón, Sociedad Española de Estrabismo y Oftalmología Pediátrica (SEEOP), Madrid.
Rosario Gómez de Liaño Sánchez, Hospital Clínico San Carlos, Sociedad Española de Estrabismo y Oftalmología Pediátrica (SEEOP), Madrid.
Alfonso Arias Puente, Hospital Universitario Fundación Alcorcón, Sociedad Oftalmológica de Madrid (SOM), Madrid.
Miguel Harto-Castaño, Hospital Universitario la Fe, Sociedad Española de Oftalmología Pediátrica (SEDOP), Valencia.
Alicia Serra Castanera, Hospital San Joan de Déu, Sociedad Española de Oftalmología Pediátrica (SEDOP), Barcelona.
Marta Galdós Iztueta, Centro Oftalmológico Astarloa, Sociedad Española de Oftalmología Pediátrica (SEDOP), Bilbao.
Sandra de Fernando Aisa, Instituto Oftalmológico Bilbao, Sociedad Española de Oftalmología Pediátrica (SEDOP), Bilbao.
Pilar Tejada-Palacios, Hospital Universitario 12 de Octubre, Sociedad Española de Oftalmología Pediátrica (SEDOP), Madrid.
Gorka Martínez-Grau, Clínica Barraquer, Sociedad Española de Cirugía Plástica Ocular y Orbitaria (SECPOO), Barcelona.
Marco Sales-Sanz, Hospital Ramón y Cajal, Sociedad Española de Cirugía Plástica Ocular y Orbitaria (SECPOO), Madrid.
María Teresa Dapena-Crespo, Clínica Oftalmológica Dapena-Lavín, Sociedad Ergoftalmológica Española (ERGO), Madrid.
José María Aguilar Ortiz, Clínica Aguilar, Sociedad Ergoftalmológica Española (ERGO), Madrid.
José María Alonso Herreros, Hospital General Universitario Los Arcos del Mar Menor, Sociedad Española de Farmacia Hospitalaria (SEFH), San Javier, Murcia.
Silvia Berisa Prado, Instituto Oftalmológico Fernández Vega, Sociedad Española de Farmacia Hospitalaria (SEFH), Oviedo.
Please cite this article as: Gegúndez-Fernández JA, Zarranz-Ventura J, Garay-Aramburu G, Muñoz-Negrete FJ, Mendicute del Barrio J, Pablo-Júlvez L, et al. Recomendaciones para la atención oftalmológica durante el estado de alarma por la pandemia de enfermedad por coronavirus COVID-19. Arch Soc Esp Oftalmol. 2020;95:300–310.