Sunitinib (SUN) and Pazopanib (PAZ) are two oral tyrosine kinase inhibitors (TKIs) against vascular endothelial growth factor (VEGF). Their efficacy and safety in metastatic renal cell carcinoma (mRCC) has been proven with phase III studies. However, real world data is limited. The objective of this study is to assess the clinical benefit of SUN and PAZ in routine practice.
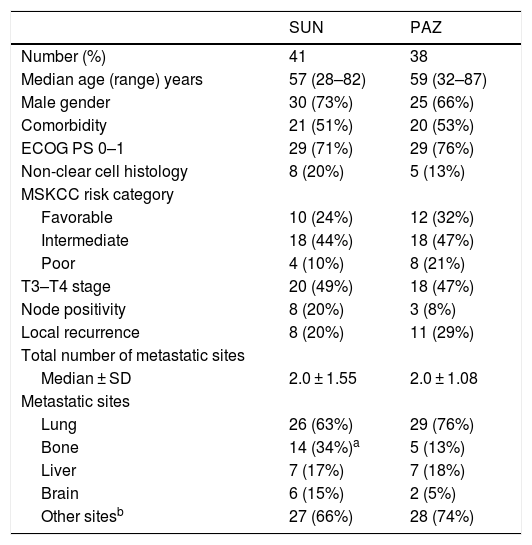
MethodsWe reviewed the medical records of seventy-nine mRCC patients treated with SUN (50 mg/day on 4/2-schedule) or PAZ (800 mg/day continuously). Patients were assessed retrospectively at two Turkish hospitals between 2006 and 2016.
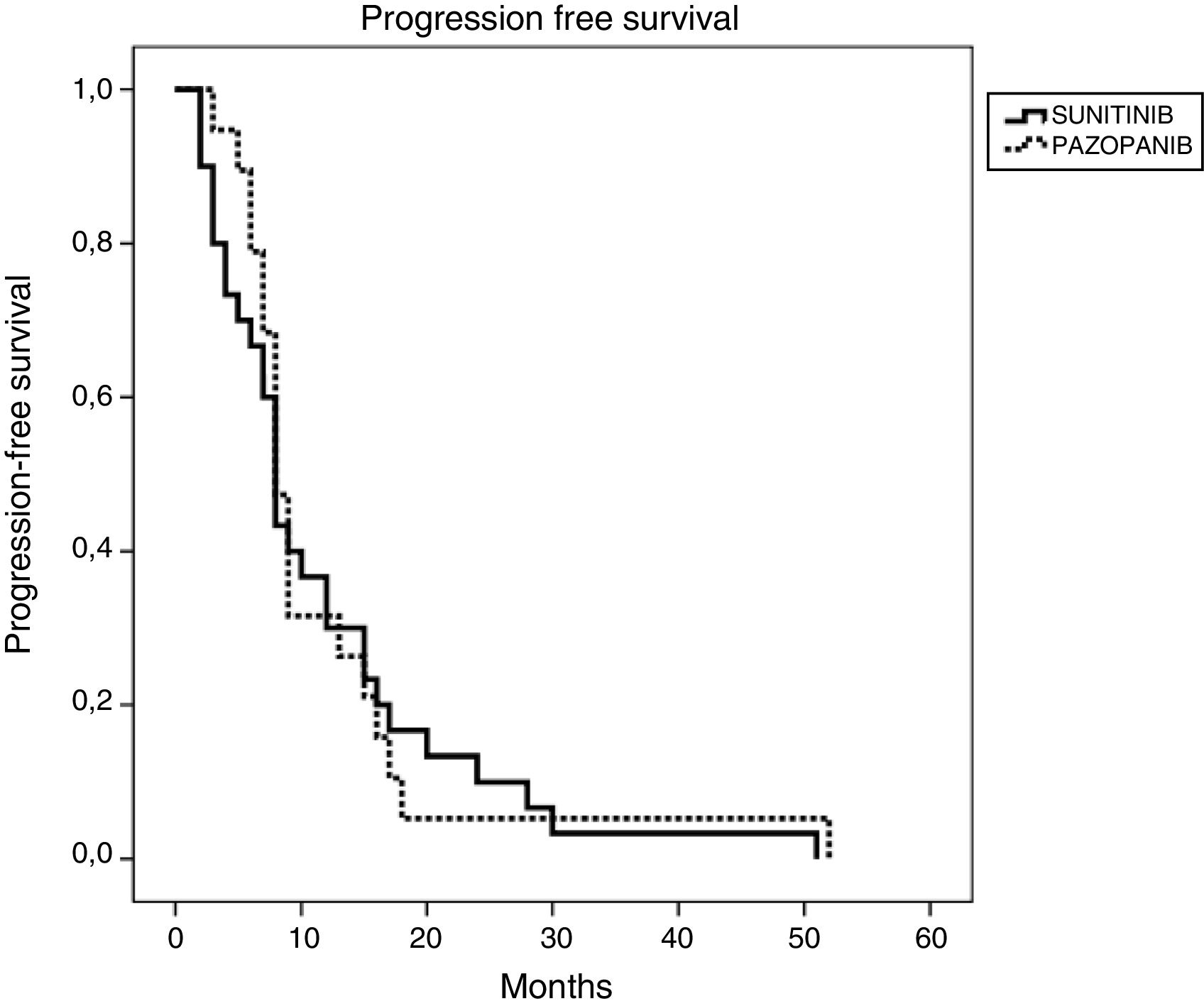
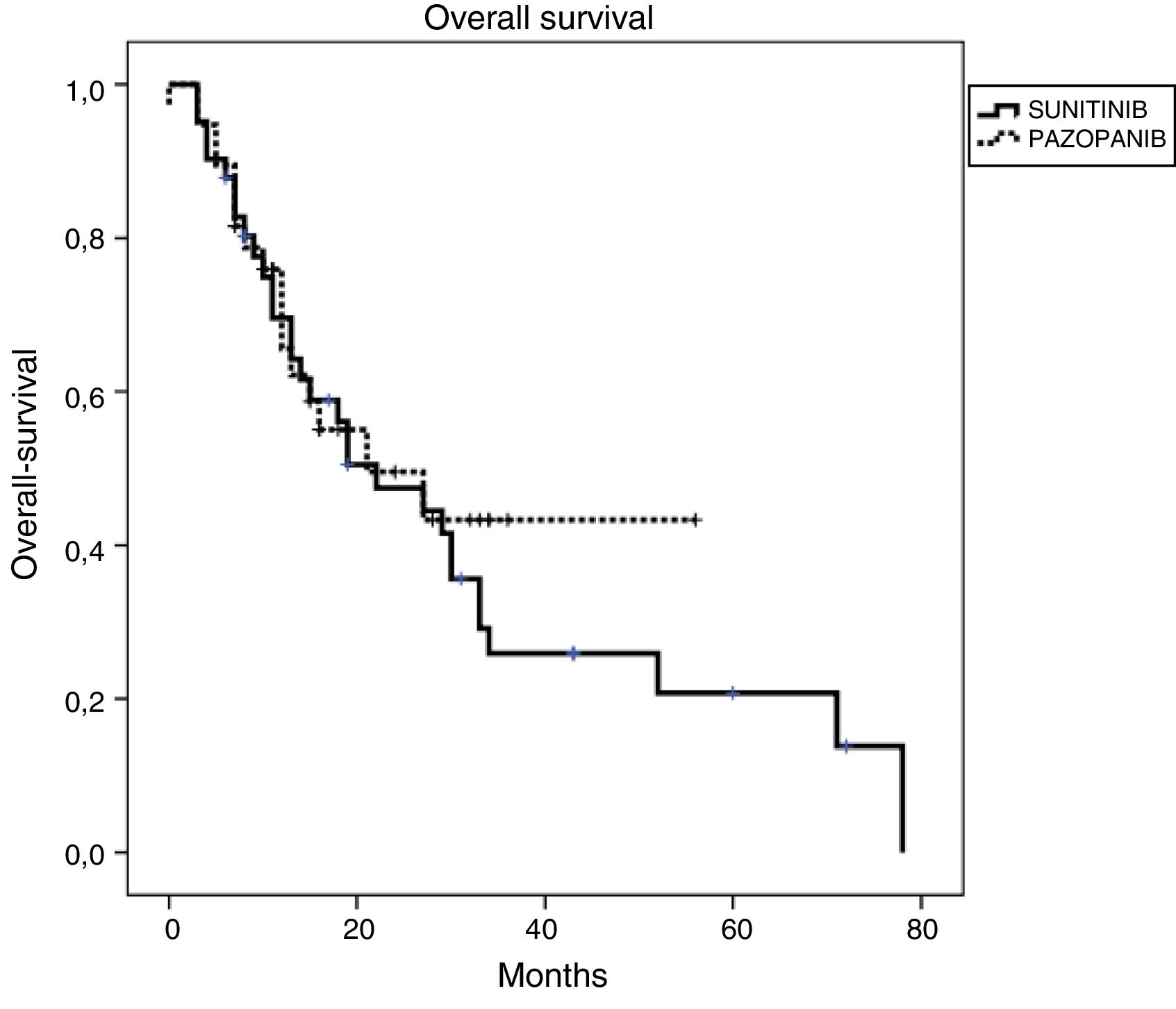
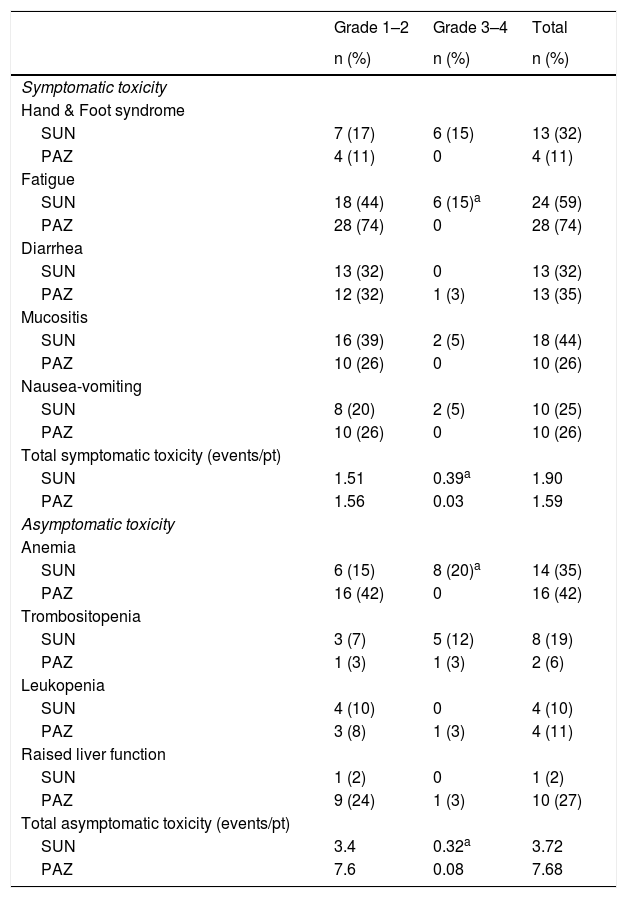
ResultsFor the entire cohort median age of patients was 60 (28–87) years and 70% of them were male. The objective response rate (ORR) and disease control rate (DCR) in SUN/PAZ groups were 34/37% (p = 0.96) and 78/87% (p = 0.046), respectively. With a median follow up duration of 15 months, median progression-free survival (PFS) and overall survival (OS) in SUN/PAZ groups were 8/8 months (p = 0.83) and 22/21 months (p = 0.53), respectively. The common all grade toxicities for SUN vs. PAZ were fatigue (59 vs. 74%), skin changes (44 vs. 44%), anemia (35 vs. 42%), hypothyroidism (37 vs. 19%; p = 0.02) and hypertension (33 vs. 50%). In patients treated with SUN, total grade 3–4 toxicities (mean number of toxic events per patients) were 0.71, whereas in patients treated with PAZ, total grade 3–4 toxicities were 0.11 (p < 0.001). SUN was associated with an increased incidence of grade 3–4 fatigue (p = 0.007), anemia (p = 0.001) and hypothyroidism that needed therapy (p = 0.02). Dose reduction in 49% and 24% of patients (p = 0.02), and treatment cessation in 37% and 26% of patients (p = 0.37) were required in the SUN and PAZ groups, respectively.
ConclusionsIn our study, there was no difference in terms of survival outcomes between two agents. However, patients treated with SUN had more grade 3–4 adverse events which prompted dose reduction.
Sunitinib (SUN) y Pazopanib (PAZ) son dos inhibidores orales de la tirosina kinasa (ITK) que actúan contra el factor de crecimiento endotelial vascular (VEGF, por Vascuar Endothelial Growth Factor). Su eficacia y seguridad en el carcinoma de células renales metastásico (CCRm) se ha demostrado con estudios de fase III. Sin embargo, la evidencia real es escasa. El objetivo de este análisis es evaluar el beneficio clínico de SUN y PAZ en la práctica clínica habitual.
MétodoRevisamos los registros médicos de 79 pacientes con CCRm tratados con SUN (50 mg/día en el régimen 4/2) o PAZ (800 mg/día continuo). Los pacientes fueron evaluados retrospectivamente en dos hospitales turcos entre 2006 y 2016.
ResultadosLa mediana de edad de toda la cohorte fue 60 (28–87) años y el 70% de ellos eran hombres. La tasa de respuesta objetiva (TRO) y la tasa de control de la enfermedad (TCE) en los grupos SUN/PAZ fueron 34/37% (p = 0.96) y 78/87% (p = 0.046), respectivamente. Con una mediana de seguimiento de 15 meses, las medianas de supervivencia libre de progresión (SLP) y de supervivencia general (SG) en los grupos SUN/PAZ fueron de 8/8 meses (p = 0,83) y 22/21 meses (p = 0,53), respectivamente. La toxicidad común entre SUN vs. PAZ incluía fatiga (59 vs. 74%), cambios en la piel (44 vs. 44%), anemia (35 vs. 42%), hipotiroidismo (37 vs. 19%; p = 0.02) e hipertensión (33 vs. 50%). En los pacientes tratados con SUN, la toxicidad total de grado 3–4 (número medio de eventos tóxicos por paciente) fue de 0,71, mientras que, en los pacientes tratados con PAZ, la toxicidad total de grado 3–4 fue de 0,11 (p < 0,001). SUN se asoció con una mayor incidencia de fatiga de grado 3–4 (p = 0.007), anemia (p = 0.001) e hipotiroidismo requiriendo tratamiento (p = 0.02). Fue necesario reducir la dosis en los grupos SUN y PAZ en el 49% y el 24% de los pacientes (p = 0,02), y el cese del tratamiento en el 37% y el 26% de los pacientes (p = 0,37), respectivamente.
Conclusionesen nuestro estudio no hubo diferencias en términos de supervivencia entre los dos agentes. Sin embargo, en los pacientes tratados con SUN se dieron más eventos adversos de grado 3–4, siendo necesaria la reducción de la dosis.