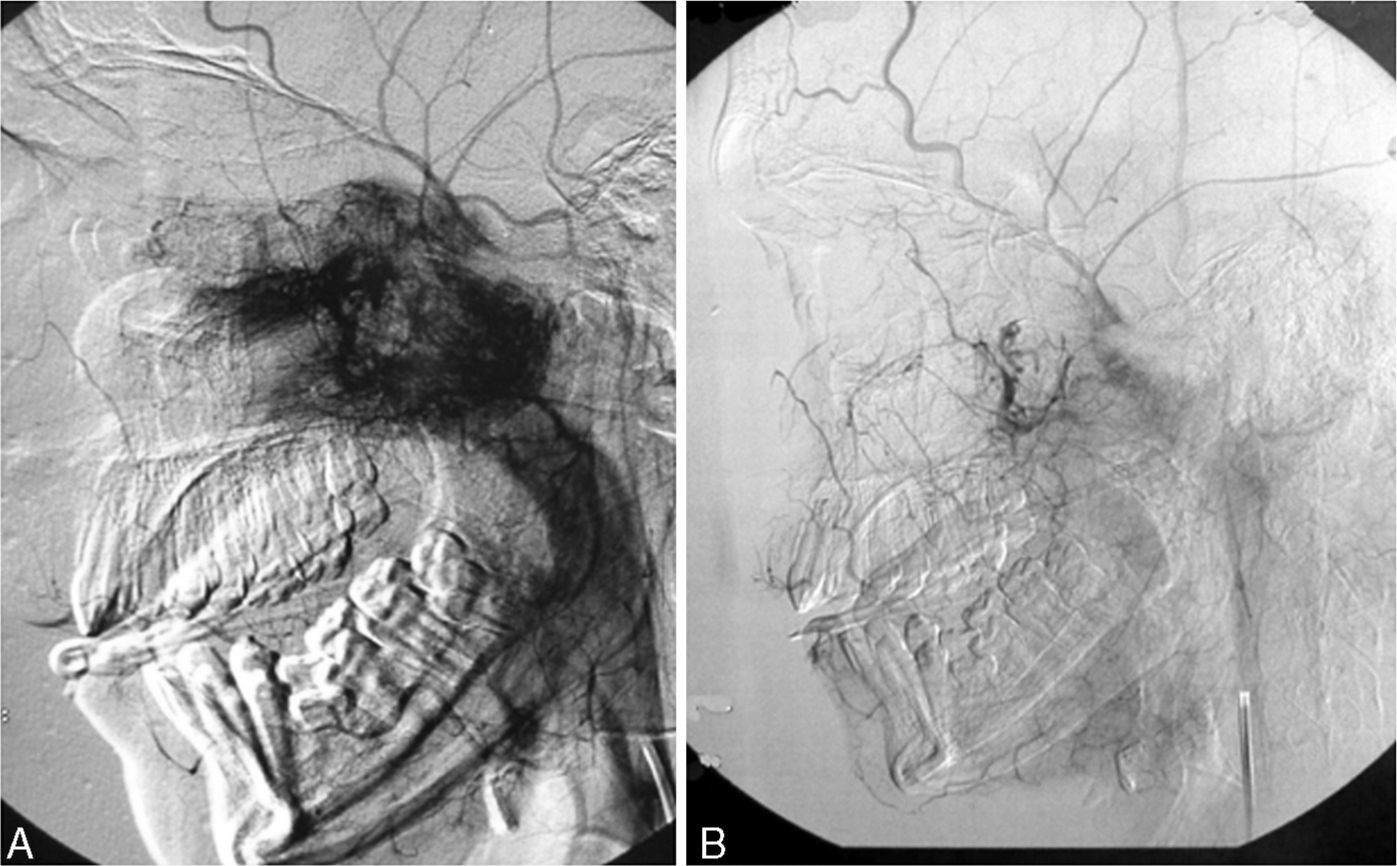
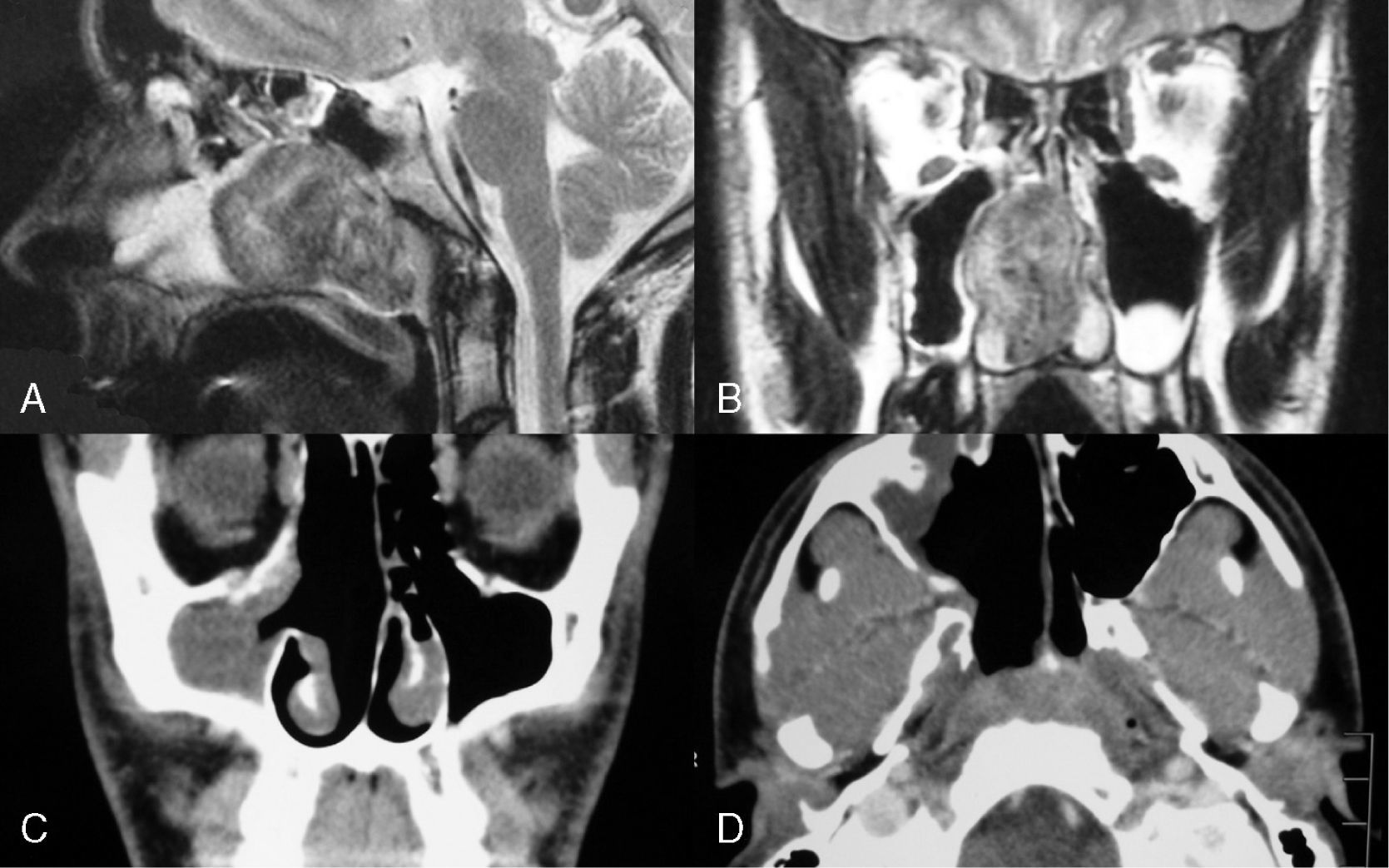
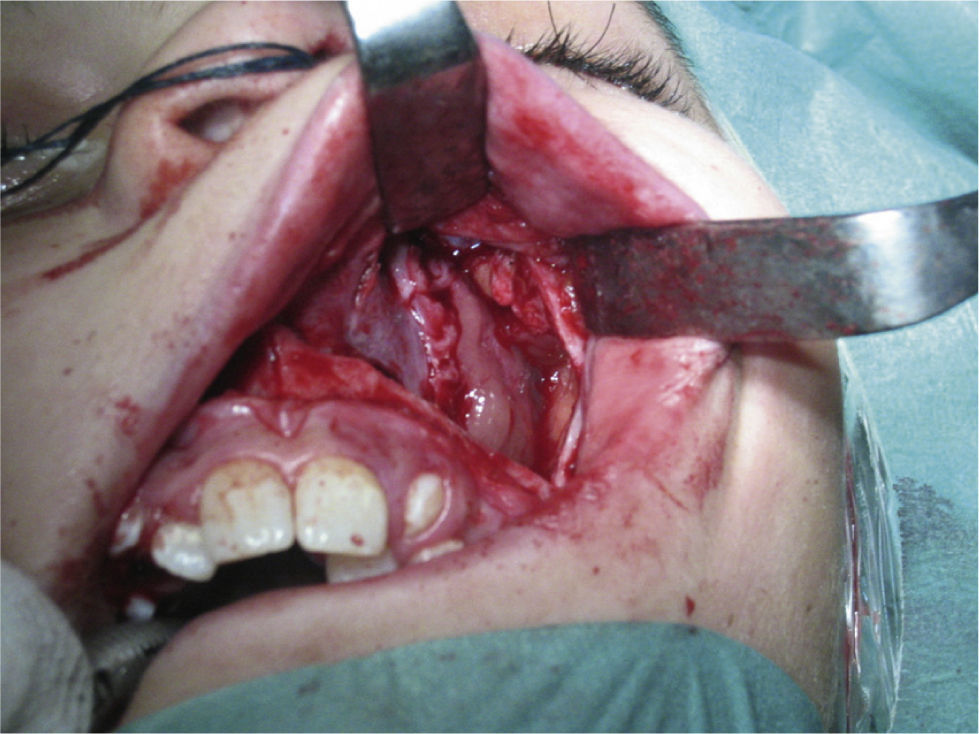
Juvenile nasopharyngeal angiofibroma (JNA) is a rare benign tumour in adolescent males. It may be associated with a significant morbidity because of its anatomical location and its locally destructive growth pattern. Severe haemorrhage constitutes a high risk in JNA and its surgical management could be complex.
Material and methodsWe retrospectively analysed the clinical data from 43 patients with JNA surgically treated in our Department from 1993 until 2010. Mean postoperative follow-up time was 85 months.
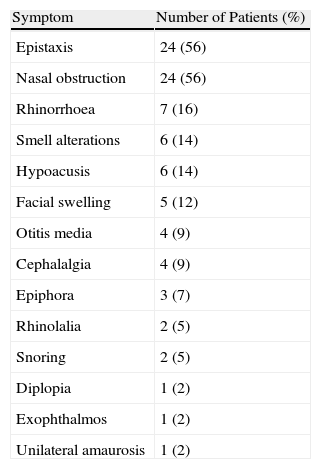
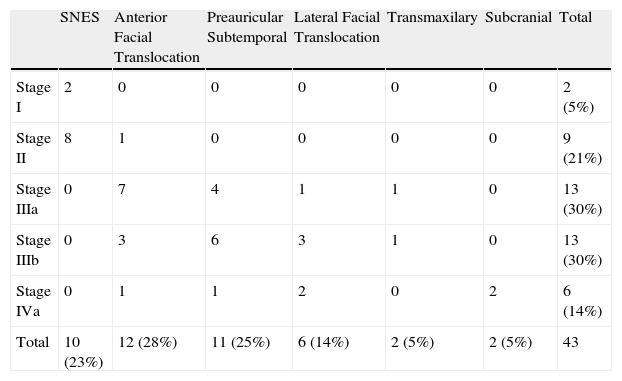
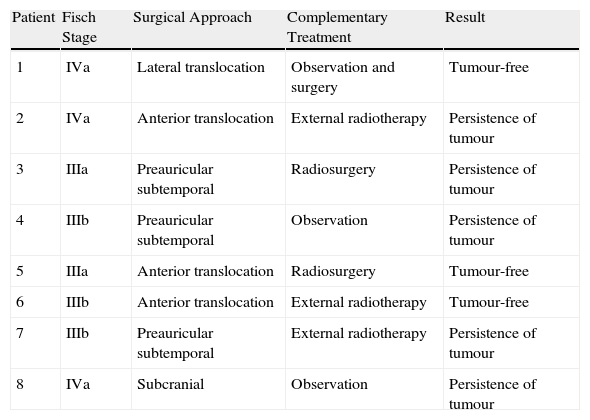
ResultsAnalysis was performed on 42 males and one female. Mean patient age was 16years old. The most common presenting symptoms were unilateral epistaxis (56%) and nasal obstruction (56%). Using the Fisch staging scale, tumours were classified as stage I in 2 patients, stage II in 9, stage III-a in 13, stage III-b in 13 and stage IV-a in 6. Preoperative selective embolisation was performed on 32 patients (74%). Thirty-three patients (77%) underwent an open surgical approach and 10 (23%) were treated by endoscopic approach. Complete resection of the lesion was achieved in 35 patients (81%) and tumour recurrence was observed in 2 (5%). All lesions treated via transnasal endoscopic approach were stage I and stage II lesions.
ConclusionSurgery is the treatment of choice for JNA. An endoscopic approach is feasible for early-stage lesions (Fisch I and II) and conservative external approaches are still useful in advanced stages (Fisch III and IV). The open approaches proved helpful with respect to exposure, safety, cosmetic outcome and low morbidity. Preoperative embolisation, if possible, is mandatory.
Los angiofibromas nasofaríngeos juveniles (ANJ) son tumores benignos raros, típicos de adolescentes varones y cuyo tratamiento de elección es la cirugía. Dada su localización y su comportamiento expansivo pueden generar una elevada morbilidad. Tienen gran tendencia al sangrado y su abordaje quirúrgico es complejo.
Material y métodoSe presenta una serie retrospectiva de 43 casos de ANJ tratados en nuestro servicio entre los años 1993 y 2010. El tiempo medio de seguimiento fue de 85 meses.
ResultadosLa muestra se componía de 42 varones y una mujer. Los síntomas de presentación más frecuentes fueron la epistaxis unilateral (56%) y la obstrucción nasal (56%). Dos ANJ pertenecían al estadio I de Fisch, 9 al II, 13 al IIIa, 13 al IIIb y 6 al IVa. En 32 pacientes (74%) se realizó embolización preoperatoria. En 33 pacientes (77%) se realizó un abordaje quirúrgico abierto y en 10 (23%) se intervino mediante cirugía endoscópica nasosinusal (CENS). Se consiguió la resección completa en 35 pacientes (81%) y 2 (5%) presentaron una recidiva tumoral. Todos los pacientes intervenidos mediante CENS pertenecían a los estadios I y II de Fisch no observándose persistencia ni recidiva tumoral.
ConclusionesEl tratamiento de elección de los ANJ es la cirugía. El abordaje mediante CENS es electivo para estadios precoces (I y II de Fisch). En estadios avanzados (III y IV de Fisch) los abordajes externos conservadores siguen teniendo su indicación por su seguridad con una escasa morbilidad y secuelas estéticas. La embolización preoperatoria, si es posible, es preceptiva.