Presently there are many retrofilling materials in the market, nevertheless, little is known about their toxicity on gingival fibroblasts.
ObjectiveTo assess cytotoxicity of three materials to human gingival fibroblasts and L929 mouse fibroblasts cell line.
Material and methodsEndoSequence® BC RRMTM (ERRM; root repair material), white MTA Angelus® (MTA) and intermediate restoration material (IRM®) conditioned media were obtained when materials were freshly mixed, at setting time and after 1, 24 and 72hours of setting time. Cell morphology was assessed with light microscopy and cell viability was assessed through mitochondrial metabolic activity with 3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyl tetrazolium bromide (MTT). Statistical analysis was conducted with ANOVA.
ResultsWe found that ERRM material did not exhibit cytotoxic effects on used fibroblast, nevertheless, MTA and IRM® respectively exhibited moderate and severe cytotoxicity, thus indicating the materials were not fully harmless.
ConclusionBioceramic cements like ERRM could be considered the most compatible retrofilling-materials.
Existen diversos materiales de retroobturación, pero poco se sabe de su toxicidad sobre fibroblastos gingivales.
ObjetivoEvaluar la citotoxicidad de tres materiales de retroobturación sobre fibroblastos gingivales humanos y fibroblastos de la línea L929.
Material y métodosLos medios condicionados de los materiales de retroobturación EndoSequence® BC RRMTM (ERRM), trióxido mineral agregado MTA Angelus® blanco (MTA) y material de restauración intermedia (IRM®) se obtuvieron en fresco, al tiempo de fraguado, y después de 1, 24 y 72 horas del tiempo de fraguado. La morfología celular fue evaluada por microscopia de luz y la viabilidad celular fue evaluada a través de la actividad metabólica mitocondrial con 3-(4,5-dimetiltiazol-2-il)-2,5-difenil bromuro de tetrazolio (MTT). El análisis estadístico se realizó por ANOVA.
ResultadosEl material ERRM no mostró efectos citotóxicos sobre los fibroblastos. Sin embargo, el MTA y el IRM® mostraron citotoxicidad moderada y alta, respectivamente. Esto revela que el MTA y el IRM® no son completamente inocuos.
ConclusiónLos materiales biocerámicos como el ERRM pueden ser considerados los materiales de retroobturación más biocompatibles.
In the field of Endodontics, surgery is recommended in cases when canal treatment failed or when there is any contraindication to conducting conventional root canal treatment.1 In an event of periapical surgery, an incision is first performed in order to uncover the apical section of the infected tooth. After this, infected tissue in the affected area is removed with a curette. Retrofilling site is later prepared, retrofilling is performed and, finally, incision is sutured. In this procedure, retrofilling materials are used which ideally would provide apical seal and facilitate periapical tissue reparation.2 Microscope use, surgical techniques developments, improvement of lighting and availability of new retrofilling materials greatly contribute to the success of periapical surgery.3,4 These advantages have heralded a new era in endodontic surgery.5
An ideal retrofilling material must possess suitable adhesion to dentin, appropriate sealing, dimensional stability, insolubility to tissue fluids, fair compressibility, adequate working time and rapid setting. Moreover, it must be resorbable, radio-opaque, easy to manipulate and biocompatible with the host's tissues.6–8 Among materials used in retrofilling we can count the following: amalgam, resin materials, zinc oxide-eugenol cements, glass ionomer cements, polycarboxylate cements Cavit™ and gutta-percha.9
Amalgam was the most frequently used material in periapical surgeries, nevertheless, it did not provide suitable clinical results.4 After amalgam, use of cements based on zinc oxide and eugenol was established. Some of the disadvantages of these cements are long setting time and high solubility. Incorporation of polymethylmethacrylate was an improvement in these cements. Intermediate restoration material (IRM®) is the outcome of this improvement. This cement is easy to handle, easy to mix, possesses better sealing capacity, requires lesser setting time4 and provides suitable postoperative results.10 Nevertheless, this cement is not biocompatible, since it elicits inflammatory response in tissues surrounding the tooth.3 Another cement used is mineral trioxide aggregate (MTA), composed mainly of calcium silicate. It was developed at Loma Linda University, California, in 1993, later available in the market from 1999 onwards. MTA possesses excellent sealing capacity and provides suitable long term results. Moreover, it is a tissue-biocompatible material since it does not cause inflammatory reactions around teeth.11 Furthermore, it possesses reparation capacity through hard tissue formation and counts with antibacterial properties.7 Although MTA as retrofilling material has won recognition from the time it was introduced in the endodontic market, it is a material difficult to handle12 and hard to place during treatment.13 An additional drawback of this material is its prolonged setting time.6,11 MTA has been improved with variations in its composition. MTA Angelus® (Angelus, Londrina Brazil) is one of these variations.12 White MTA Angelus® has shown to possess greater amounts of calcium carbonate, calcium silicate, and barium and zinc phosphates. These elements contribute to improve setting time and manipulation ease.6,14 MTA Angelus® is similar to original MTA with respect to biocompatibility and antimicrobial capacity as well as its physical and chemical characteristics.15 A new retrofilling material called EndoSequence® BM RRMTM (ERRM, root repair material) is composed of a combination of calcium silicate and calcium phosphate, and provides biomedical and dental use.16 It exhibits the following physical properties: it is hydrophilic and homogeneous, since it is presented in a pre-mixed paste, and thus possesses exceptional dimensional stability.17,18 Additionally, it is a highly radio-opaque material. This characteristic makes it easy to place during treatment and later to be identified in X-rays. Its 12.8 pH is partly responsible for its anti-bacterial nature, which constantly decreases during a seven day period rendering it thus highly biocompatible.6,19 The manufacturer of ERRM claims that it possesses physical and mechanical properties similar to MTA but with better fit and handling characteristics.1
BiocompatibilityMTA is a biocompatible material.8,14 Torabinejad and his team showed that MTA, both freshly mixed and fully set, is less cytotoxic than IRM®.8,20 Moreover, MTA does not interfere with cell adhesion.8,17 MTA and IRM® have been widely researched and are used as retrofilling materials.10 Prospective clinical studies have shown that both possess suitable ability for apical healing.3 The manufacturer of ERRM claims that this material possesses excellent physical characteristics as well as suitable biocompatibility, providing very favorable results when it is used as retrofilling material, although there is scant literature supporting this claim. Different retrofilling materials exist in the market, but few comparative reports about their cytotoxicity potential using fibroblasts are available 13. Due to the aforementioned facts the aim of the present research paper was to compare cytotoxicity of White MTA Angelus®, IRM® and recently launched in the Mexican market, EndoSequence® BC RRMTM (ERRM) retrofilling materials on human gingival fibroblast culture (FGH) and L929 mouse fibroblast cell line.
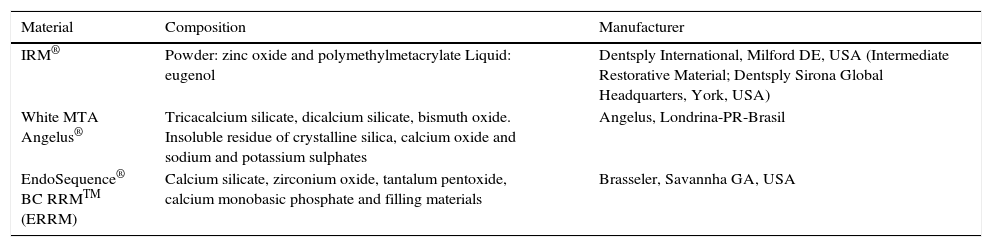
MATERIAL AND METHODSRetrofilling materialsThree retrofilling materials were used: intermediate restoration material (IRM®) which is a cement based on zinc oxide and eugenol, white MTA Angelus® (MTA) and EndoSequence® BC RRMTM root repair material (ERRM) which is a bioceramic material (Tables I and II). Materials were prepared as presented in table II.
Retrofilling material, composition and manufacturer.
| Material | Composition | Manufacturer |
|---|---|---|
| IRM® | Powder: zinc oxide and polymethylmetacrylate Liquid: eugenol | Dentsply International, Milford DE, USA (Intermediate Restorative Material; Dentsply Sirona Global Headquarters, York, USA) |
| White MTA Angelus® | Tricacalcium silicate, dicalcium silicate, bismuth oxide. Insoluble residue of crystalline silica, calcium oxide and sodium and potassium sulphates | Angelus, Londrina-PR-Brasil |
| EndoSequence® BC RRMTM (ERRM) | Calcium silicate, zirconium oxide, tantalum pentoxide, calcium monobasic phosphate and filling materials | Brasseler, Savannha GA, USA |
Conditioned media and their respective dilutions were obtained following the norm ISO 10993-5:2009 (International Standardization Organization 2009).20 Briefly, sterile teflon discs were placed on top of sterile Mylar Teflon sheets squares (1.1 x 1.1 cm; both from Tetraflon de México S.A. de C.V. Toluca, Estado de México). Teflon discs were filled with fresh materials to be later placed in tissue culture 24-well plates (Corning Inc., Corning, NY). To this mix, 1.5mL of supplemented Dulbecco's modified Eagle medium were added (DMEM; GIBCO, Invitrogen, Grand Island, New York, USA). Media was supplemented with 10% bovine fetal serum. (ByProductos SA de CV, Guadalajara, Jalisco, México), 8 mM L-glutamine and antibiotics (10,000 units/mL penicillin and 25 μg/ mL Fungizone® (GIBCO, Invitrogen, Grand Island, NY, USA). Culture medium was left in contact with retrofilling materials at different times: fresh (0 h), setting time (Table II) and at 1, 24 and 72hours after setting. Conditioned media was collected in Eppendorf tubes and stored at -20°C, until used. Media, which were not exposed to any cement, were also collected as control conditioned media, during the same times. Three dilutions of each conditioned medium (1/10, 1/100 and 1/1,000) were prepared.
Cell linesL929 mouse fibroblast cell line, as well as human gingival fibroblasts (HGF) were used in the present study. HGF were obtained from gingival tissue donated by healthy patients. These procedures were approved by the corresponding Ethics Committee; all donors signed informed consent forms. Gingival fibroblasts were harvested as follows: gingival tissue was placed in DMEM under sterile conditions, human gingival tissue was washed and divided into small pieces (1mm3). Gingival tissue pieces were placed in 3mL DMEM in 50mL tubes. After this, the medium was removed and new medium with 5mL of 5 mg/ mL collagenase type IV was placed (Sigman-Aldrich Co., St Louis, MO, USA). The tube was placed in water at 37°C for 2hours, and it was shaken for 30seconds every 30minutes. After this, the tube was centrifuged at 377g for 2minutes, supernatant was then removed, and cells were re-suspended in 5mL supplemented; the medium was then transferred to a 25cm2 culture flask (Corning Inc., Corning NY, USA). The flask was incubated at 37°C with 5% CO2 for 24hours. Non-adhered cells were removed and human gingival fibroblasts were left to grow until reaching confluence.21
Cell cultureBoth fibroblast cell lines were cultured in supplemented DMEM. Cell cultures were kept in 75cm2 cell culture flasks (Corning Inc., Corning NY, USA) until reaching 80-90% confluence, at which point they were sub-cultured. To this effect, fibroblasts were detached with 0.05% trypsin/EDTA (GIBCO, Invitrogen, Grand Island, NY). Trypsin was neutralized with an equal volume of fresh culture medium. Cell suspension was centrifuged for three minutes at 1,500rpm. Supernatant was discarded and cell pellet was re-suspended in 5mL of fresh medium. One fourth of the re-suspension was placed in a new culture flask. All experiments were conducted between subcultures three and 10.
Cell morphologyIn order to assess cell morphology, 104 fibroblasts/ well were seeded in 96-well plates (Corning Inc., Corning, NY) in a final volume of 100 μL culture medium per well. Cultures were kept in an incubator for 24 h (Nuaire TM Plymouth, USA) at 37°C, 95% relative humidity and 5% CO2. After 24 h, the time required to allow fibroblasts to adhere to the wells, culture medium was removed and 100 μL of all corresponding conditioned media were placed for another 24 h. Morphology was then evaluated with a model IX70 Olympus microscope (Center Valley, PA, USA). Images were captured with an Evolution-VF Cooled Color camera (Media Cybernetics, Rockville MD, USA) and with the computer program Capture pro 6.0 QImaging (Surrey, British Columbia, Canada).
Metabolic activity assay (MTT reduction)Cytotoxic potential of retrofilling materials was assessed based on norm ISO 10993-5.2009) (International Standardization Organization) using the 3-(dimethylthiazol-2-il)-2 diphenyl tetrazolium bromide assay (MTT) (Sigma Aldrich St Louis, MO, USA).21 Briefly, the same cultures that were used to take fibroblast pictures were removed from their conditioned media and 50 μL of dissolution of 1 mg/ mL MTT were added into each well. Cultures were protected from the light and kept at 37°C with 5% CO2 for 2 h. After this time, MTT was removed and 100 μL of isopropanol were added (TECSIQUIM, TSQ, Iztacalco, Mexico City). Cultures were incubated for 30min at room temperature. After this time, dissolution absorbance was measured at 590nm in a plate reader model Synergy HT, Bio-Tek brand (Vermont USA). Absorbance values were normalized considering 100% as the absorbance obtained from untreated cultures.
StatisticsA one-way ANOVA variance analysis for equal samples was used to determine possible differences among cement groups and among different times. Tukey test was used as post-hoc test, using KaleidaGraph® computer program, Mac version 3.6.2 (Synergy Software, Reading, PA, USA). Conditions were considered statistically different when p value was ≤ 0.05.
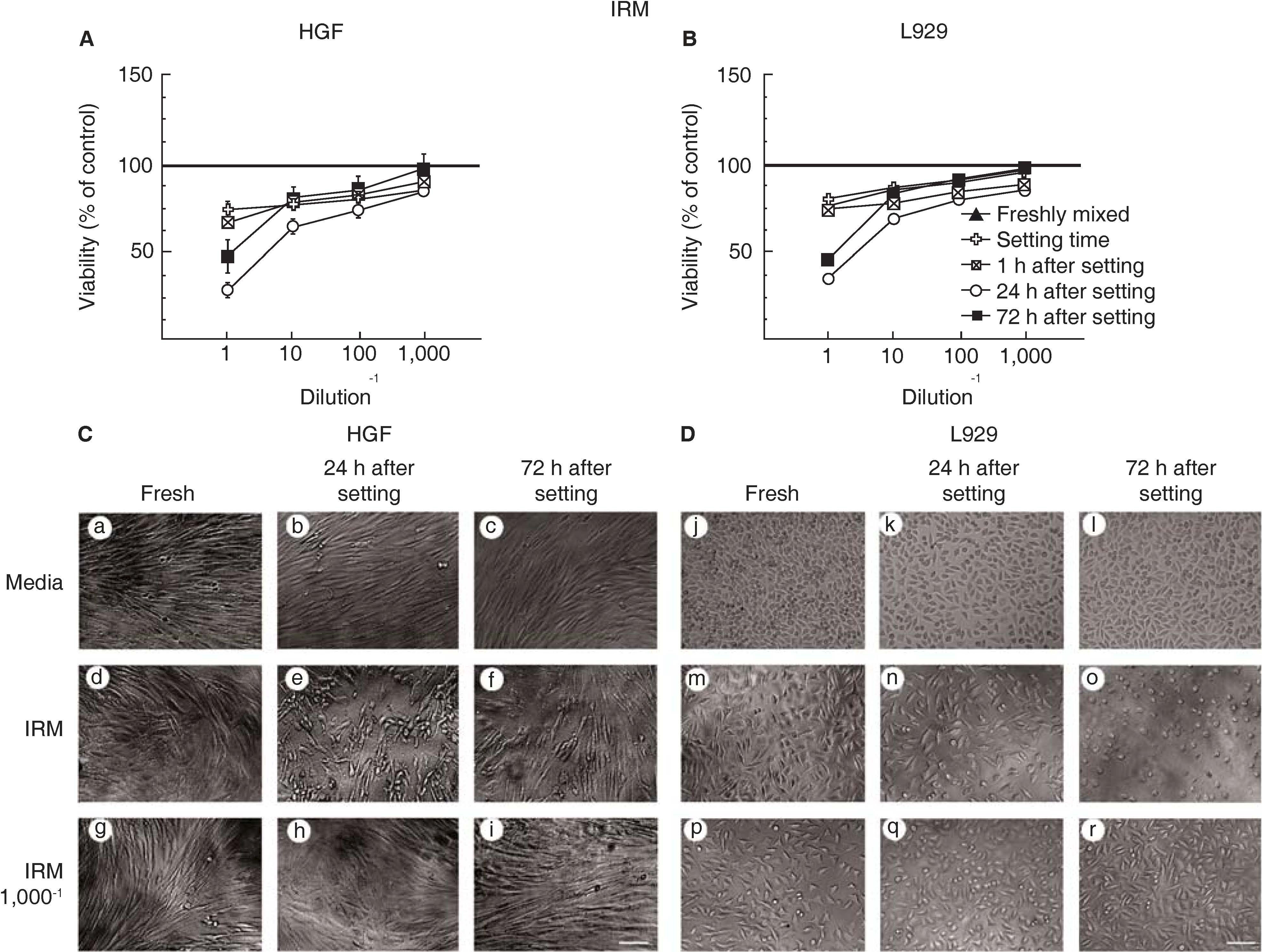
RESULTSCells treated with Intermediate restoration material (IRM®) were severely damaged at 24hours after material had set, but recovered their morphology after 72hours after the material had set.
Exposing human gingival fibroblasts (Figure 1A) or L929 fibroblasts (Figure 1B) to IRM® did not affect cell viability when the sealer was fresh, at the setting time or after 1 hour after material was set. However, cell viability was reduced to about 70%, when the conditioning media was from 24 hour after material had set. Cell viability recovered to 50% when fibroblasts were exposed to conditioning media from 72hours after sealer had set. Cell viability remained high 80 to 90% at all dilutions at all times of conditioning medium from all but 24hours after sealer had set (Figures 1A and 1B).
Viability of human gingival fibroblasts (HGF, panel A) and L929 mouse cell line fibroblasts (panel B) after exposition to IRM®. Symbols represent the following: fresh cement (black triangle) setting (white cross) at 1h (stricken square) at 24h (white circle) and at 72h after setting (black square); 100% cell viability represents the state without cement (black line in 100%). Data represent average percentage of cell viability in three independent experiments conducted in triplicate. Microphotographs of HGF (panel C) and L-929 (panel D) exposed to IRM® concentrated conditioning medium and at 1/1,000 diluted IRM® conditioning medium. Scale: 100μm.
Treated human gingival fibroblasts (Figure 1C) or L929 fibroblasts (Figure 1D) had similar morphology to untreated fibroblasts, when exposed to IRM®, freshly mixed, just set and 1 hour after setting. However, by 24hours after setting, fibroblasts had lost their capacity to maintain a homogenous monolayer (Figures 1C and 1D panel e and n, respectively). By 72hours after setting IRM® caused severe damage L929 cell line, cells had rounded morphology (Figure 1D panel o). Cell morphology was not affected with IRM® dilutions (Figures 1C and 1D panel g-i and p-r, respectively).
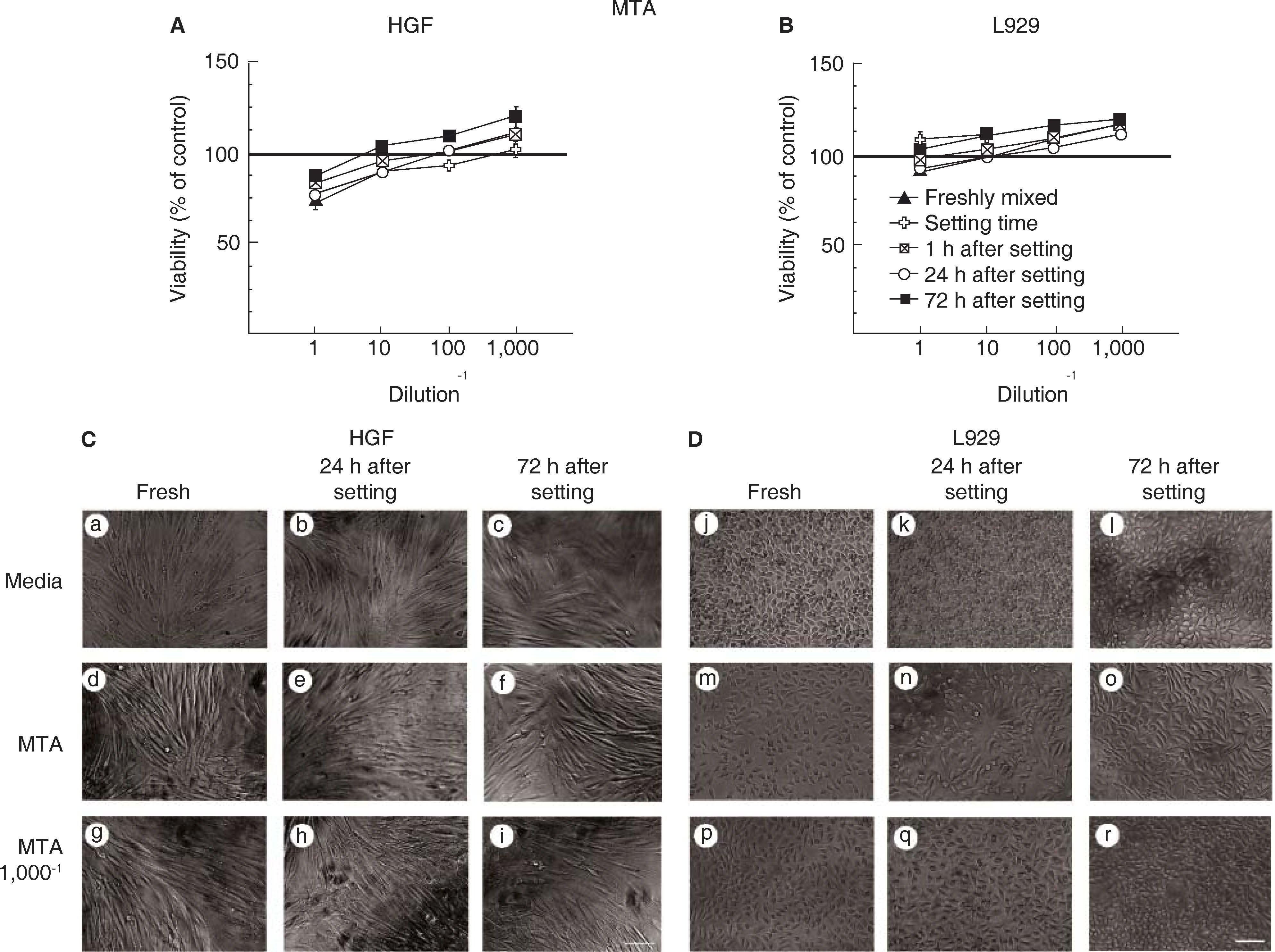
Cells treated with White MTA Angelus® (MTA) remained viable and exhibited typical fibroblasts morphology.
Exposing fibroblasts to MTA did not affect cell viability of either human gingival fibroblast (Figure 2A) or L929 cells (Figure 2B). Moreover, fibroblasts exposed to serial dilutions of the MTA conditioned medium showed an increased in metabolic activity. Accordingly, metabolic activity was higher than untreated fibroblast after 72hours after setting at 1/100 and 1/1,000 dilutions (Figures 2A and 2B). In agreement, human gingival fibroblasts had a typical bipolar elongated shape and remained attached to the culture dish at all times (Figure 2C). Similarly, L929 fibroblasts had a typical triangular elongated shape and remained viable (Figure 2D).
Viability of human gingival fibroblasts (HGF panel A) and mouse fibroblasts of L929 cell line (panel B) after exposition to MTA. Symbols represent the following: fresh cement (black triangle) setting (white cross), after 1h (stricken square), after 24h (white circle) and after 72h (black circle); 100% cell viability represents the state without cement (100% black line). Data represent cell viability percentage average of three independent experiments, conducted in triplicate. HGF microphotographs (panel C) and L929 (panel D) exposed to medium, concentrated MTA conditioned medium and to a 1/1,000 dilution of MTA conditioned medium. Scale 100μm.
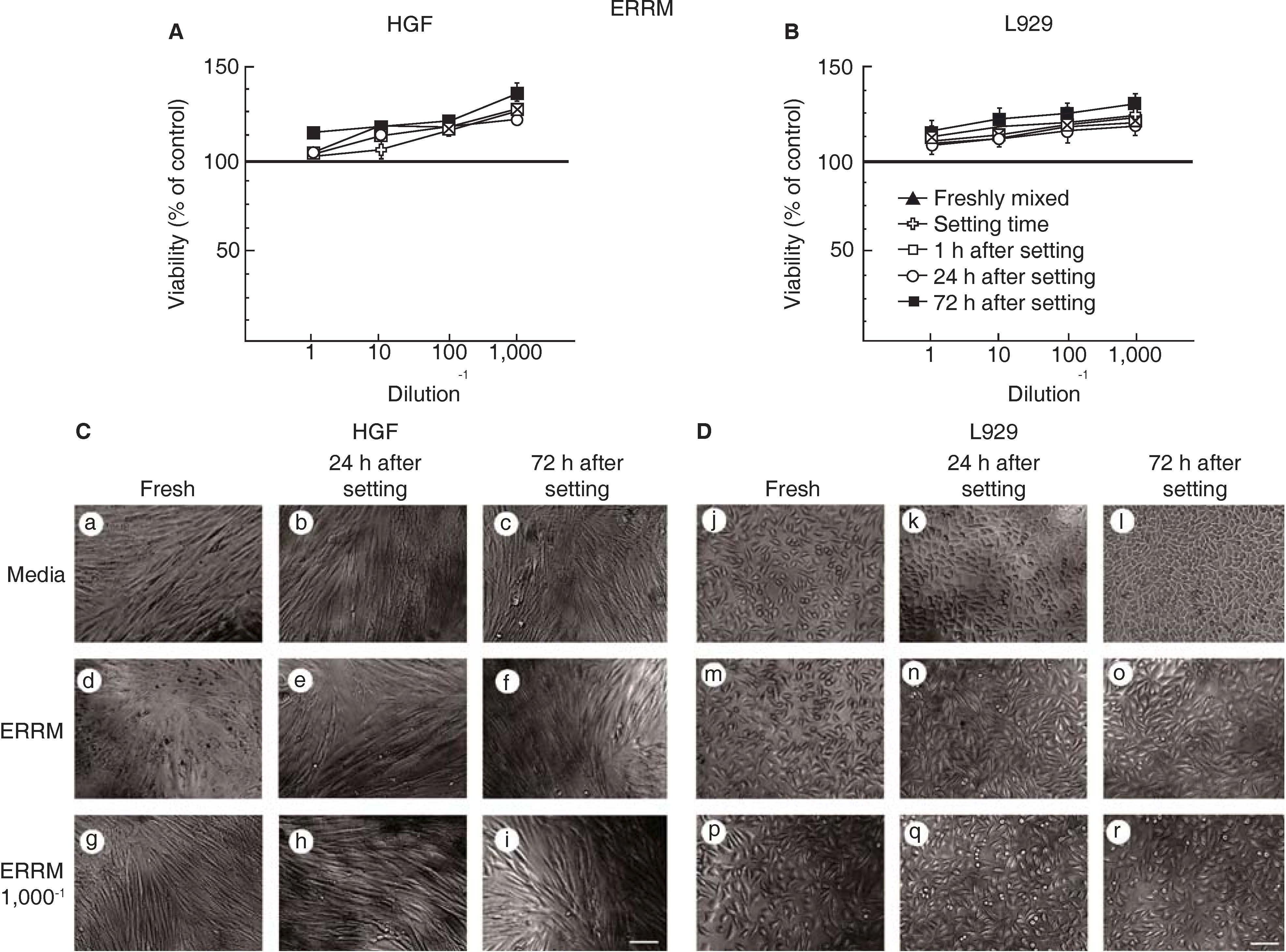
Cells treated with EndoSequence® BC RRMTM root repair material (ERRM) remained viable, exhibited typical fibroblasts morphology and displayed higher metabolic activity.
Similarly to MTA, exposing fibroblasts to ERRM did not affect cell viability of either human gingival fibroblast (Figure 3A) or L929 cells (Figure 3B). Interestingly, fibroblasts exposed to serial dilutions of the ERRM conditioned medium showed higher metabolic activity and greater viability than untreated fibroblasts (Figures 3A and B). In agreement, both human gingival fibroblasts and L929 fibroblast had a typical morphology and remained attached to the culture dish at all times (Figures 3C and 3D, respectively).
Human gingival fibroblast viability (HGF panel A) and fibroblasts of mouse cell line L929 (panel B) after exposition to ERRM. Symbols represent the following: fresh cement (black triangle), set (white cross), after 1h (stricken square) after 24h (white circle) and 72h setting (black square); 100% cell viability represents the state without cement (black line 100%). Data represent cell viability percentage average of three independent experiments, conducted in triplicate-HGF microphotographs (panel C) and L929 (panel D) exposed to medium, to ERRM concentrated conditioned medium and to a 1/1,000 dilution of ERRM conditioned medium. Scale 100μm.
A yearly estimation of orthodontic procedures mentions that approximately 5.5% of these procedures involve periapical surgery.13 In this case, it was necessary to use biocompatible retrofilling materials. Different retrofilling materials exist in the market, but few comparative reports about their cytotoxicity potential using human gingival fibroblast are available.13 For the aforementioned reason, in the present study, we assessed in vitro biocompatibility of three retrofilling materials used in periapical surgery: one of them, EndoSequence® BM RRMTM (ERRM, Root Repair Material), recently launched in the Mexican market and two well know materials: IRM® and white MTA Angelus®.
Cytotoxicity was evaluated following the international standard ISO 10993-5 (ISO 10993. 2009), which describes the tests for assessing cytotoxicity in vitro. Cytotoxicity was assessed using human gingival fibroblasts (HGF) and L929 mouse fibroblast cell line. Human gingival fibroblasts were selected to imitate cell response induced by cements in periapical tissue after endodontic retrofilling. L929 mouse cell line fibroblasts have to their high sensitivity to toxic products.22,23 Cytotoxicity was evaluated with the use of two parameters: morphology and cell viability. These parameters were assessed using cements at different circumstances: fresh, set, 12, 24 and 72hours after setting time.
Most of the studies with cell cultures have focused on the cytotoxicity of materials used in endodontics, especially when the material is set.2 When materials are in a fresh state, they release great amounts of chemical sub-products which are toxic to cell cultures. Nevertheless, in virtue of their clinical conditions (in vivo), these sub-products dilute in fluids of interstitial tissues and are eliminated through the vasculature. Therefore, cytotoxic effect of diluting the materials has also being studied.2,24
In the present study we found that IRM®, a material composed of zinc oxide and eugenol, showed to be the most cytotoxic 24 and 72hours after set. This fact concurs with other recent studies.4,8,25,26 One explanation for this high cytotoxicity might be illustrated by the fact that both eugenol and zinc are cytotoxic.18,27 It is thought that released zinc is partly responsible for its prolonged cytotoxic effect.28 Another explanation could be that variations in composition of reinforced materials based on zinc oxide and eugenol could affect their dissolution rate, causing variations in cytotoxicity.4,29 Moreover, in IRM®, eugenol might exhibit affinity to polymethylmethacrylate, limiting the release of this material and rendering it less cytotoxic.4 Another explanation could be that when IRM® is used as retrofilling material, it is recommended to use greater proportion of powder than liquid, since this provides great handling advantages, short setting time and decrease of toxicity and solubility.30
Nevertheless, this strong cytotoxicity was not seen if IRM® was diluted. In concordance, Trope et al31 in a histological study confirmed the favorable response of tissues to IRM®.4 The advantages of IRM® are the following: low cost, ease of mixing and manipulation. Several studies have reported positive results when using this material in periradicular surgery.3,10,11,31
Due to the fact that materials used in Endodontics do not possess all these ideal characteristics, MTA was initially developed as a retrofilling material, and was later used for other procedures such as pulp capping, pulpotomy, apexogenesis, apexification, root perforation reparations, as well as a filling material for root canals. MTA has been recognized as a bioactive, osteoconductor and biocompatible material. Several reviews on MTA’ s chemical properties, biocompatibility and clinical applications have been published.7,13,14 The present study revealed that with mineral trioxide aggregate (MTA) cytotoxicity was almost nil in its fresh state, this concurs with other recent published studies,1,6,8,16,32 where MTA has been deemed as a highly biocompatible retrofilling material. Likewise, Kim and Kratchman5 mention in their article that MTA is the most biocompatible retrofilling material, and that it can be used with predictable results in endodontic surgery procedures.33 In our study, results concurred with others which showed MTA's high biocompatibility in comparison to IRM®.8,17
The recently launched in the Mexican market retrofilling material ERRM did not show any degree of cytotoxicity indeed, it increased fibroblast metabolic activity; making it the most biocompatible material of this study. Nevertheless ERRM has shown some cytotoxicity, possessing a degree of cytotoxicity similar to MTA's.6,16,27
CONCLUSIONEndoSequence® BC RRMTM root repair material (ERRM) is the most biocompatible retrofilling material and IRM® is more cytotoxic than MTA.
Research in the authors’ laboratory was supported by grant PAPIIT IA202013 (to EUQ) from Dirección General de Asuntos del Personal Académico, Universidad Nacional Autónoma de México, Mexico. To Nancy Mora Pérez, Master in Basic Biomedical Research, for his technical support.
This article can be read in its full version in the following page: http://www.medigraphic.com/facultadodontologiaunam