Recent molecular phylogenetic analysis of Lagenidium strains recovered from subcutaneous lesions in cats, dogs, and a human with lagenidiosis resolved into four clades; one of them was Lagenidium giganteum, but three others were novel.
AimsDue to the recent increase in L. giganteum infections from mammals, we studied 21 Lagenidium strains isolated from dogs and a human available in our collection.
MethodsMolecular phylogenetic studies and phenotypic characteristics were used to characterize the strains.
ResultsWe report the finding of three novel species, herein designated as Lagenidium ajelloi, sp. nov., Lagenidium albertoi sp. nov, and Lagenidium vilelae sp. nov. Their morphological and growth features are also presented.
ConclusionsOur study revealed the presence of three novel Lagenidium species infecting mammals.
Estudios recientes, basados en análisis filogenéticos, han revelado la existencia de cuatro clados de Lagenidium aislados en lesiones subcutáneas de gatos, perros y de un ser humano con lagenidiosis. Uno de los aislamientos se identificó como Lagenidium giganteum, pero los otros tres se consideraron especies nuevas.
ObjetivosDebido al incremento de las infecciones por L. giganteum en mamíferos, se han estudiado 21 aislamientos de Lagenidium procedentes de animales con lagenidiosis.
MétodosLos aislamientos se clasificaron fenotípicamente, además de llevar a cabo estudios de filogenia con ellos.
ResultadosSe proponen tres nuevas especies de Lagenidium: Lagenidium ajelloi, sp. nov., Lagenidium albertoi, sp. nov., y Lagenidium vilelae, sp. nov. Se tratan también sus características morfológicas.
ConclusionesNuestro estudio reveló la existencia de tres especies nuevas de Lagenidium responsables de infecciones en mamíferos.
For more than a century Pythium insidiosum was considered the only species in the Oomycota commonly causing infection in mammalian hosts.10,13 This paradigm was challenged by reports in the past decade indicating that strains of Lagenidium spp. also infected apparently healthy mammals, including humans, causing cutaneous and systemic infections.16,17,23,28 Recent phylogenetic analyses of several Lagenidium giganteum isolates recovered from mammals with lagenidiosis revealed that some isolates formed a monophyletic clade with other L. giganteum strains approved by the Environmental Protection Agency (EPA) as a mosquito control agent.34 Three other clades of Lagenidium isolates found during these investigations, and also associated with mammalian infection, displayed distinctive taxonomic and phylogenetic characteristics.34 The unique phenotypic attributes and phylogenetic affinities of these three novel Lagenidium clades represent previously undescribed species and are herein proposed as new.34
Materials and methodsLiving culturesThe complete list of the strains and genomic DNA samples used in this study is shown in Table 1. The following isolates were studied: two thermo-sensitive Lagenidium giganteum ATCC 36492 and ATCC 48336 (mosquito larvae pathogen, USA) strains; Lagenidium humanum=ATCC 76726 (dead skin of humans, USA); Lagenidium albertoi=MTLA13 (ATCCMYA-4932); Lagenidium ajelloi=MTLA06 (ATCCMYA-4936), MTLA07 (ATCCMYA-4937), and MTLA19 to 23; 12 thermo-tolerant L. giganteum=MTLA01 (ATCCMYA-4933), MTLA03, MTLA-04 (ATCCMYA-4934), MTLA-05 (ATCCMYA-4935), MTLA10, and MTLA12-18 recovered from dogs and a cat; and Lagenidium vilelae=MTLA24. L. albertoi was cultured by Dr. A. Chindamporn from a Thai man with keratitis28; L. ajelloi was first isolated from two dogs (MTLA06 and 07) with protracted subcutaneous swellings mimicking skin pythiosis in Alabama, USA by Dr. J. Newton. The other five isolates (MTLA19 to 23) also were recovered from dogs with protracted subcutaneous lesions (4) and with cutaneous and systemic infections involving the intestinal tract and arteries (1) by Florida, Georgia and North Carolina veterinarians in the USA. The 12 thermo-tolerant L. giganteum strains were isolated from dogs and a cat with acute cases of subcutaneous granulomas mimicking skin pythiosis as per Vilela et al.34 The only L. vilelae strain (MTLA24) in this study was isolated by Dr. J. Lewis from a North Carolina cat with a protracted mass at the base of the tail.23 The MTLA25 of L. vilelae was formalin fixed tissue from a Virginia cat with a protracted abdominal mass containing P. insidiosum-like hyphae and thus, studied only using its extracted genomic DNA.
The table shows the strains taxonomic names, their IDs, their acronyms and their ITS, CDC42, COXII, HSP90, and tubulin NCBI accession numbers.
| Taxonomic name | Strain ID | Acronym | ITS | CDC42 | COXII | HSP90 | Tubulin |
|---|---|---|---|---|---|---|---|
| L. giganteum | NPI01 | Lgm1 | EF016915 | 0 | 0 | 0 | 0 |
| L. giganteum | ATCC48336 | Lgm2 | JQ745259 | 0 | 0 | 0 | 0 |
| L. giganteum | ATCC36492 | Lgm3 | AY151183 | JX985758 | AF086697 | JX999093 | JX999124 |
| Lagenidium sp. | CBS127533 | Ls1 | HQ395647 | 0 | 0 | 0 | 0 |
| Lagenidium sp. A | CBS126881 | Lspa1 | HQ111460 | 0 | 0 | 0 | 0 |
| Lagenidium sp. A | CBS126885 | Lspa2 | HQ111452 | 0 | 0 | 0 | 0 |
| Lagenidium sp. A | CBS126878 | Lspa3 | HQ111464 | 0 | 0 | 0 | 0 |
| Lagenidium sp. A | CBS126880 | Lspa4 | HQ111459 | 0 | 0 | 0 | 0 |
| Lagenidium sp. B | CBS126882 | Lspb1 | HQ111461 | 0 | 0 | 0 | 0 |
| Lagenidium sp. B | CBS126879 | Lspb2 | HQ111463 | 0 | 0 | 0 | 0 |
| Lagenidium sp. B | CBS126884 | Lspb3 | HQ111450 | 0 | 0 | 0 | 0 |
| Lagenidium sp. B | CBS126883 | Lspb4 | HQ111462 | 0 | 0 | 0 | 0 |
| L. callinectes | NJM-0531 | Lc1 | AB285488 | 0 | 0 | 0 | 0 |
| L. callinectes | NJM-8989 | Lc2 | AB285487 | 0 | 0 | 0 | 0 |
| L. callinectes | ATCC24973 | Lc3 | AB285486 | 0 | 0 | 0 | 0 |
| L. vilelae | CBS127042 | Lv1 | HQ111455 | 0 | 0 | 0 | 0 |
| Lagenidium sp. C | CBS127284 | Lspc1 | HQ111472 | 0 | 0 | 0 | 0 |
| Lagenidium sp. C | CBS127285 | Lspc2 | HQ111470 | 0 | 0 | 0 | 0 |
| Lagenidium sp. C | CBS127283 | Lspd1 | HQ111471 | 0 | 0 | 0 | 0 |
| Lagenidium sp. C | CBS127276 | Lspc3 | HQ111451 | 0 | 0 | 0 | 0 |
| Lagenidium sp. C | CBS127281 | Lspd2 | HQ111473 | 0 | 0 | 0 | 0 |
| Lagenidium sp. D | CBS127277 | Lspd3 | HQ111454 | 0 | 0 | 0 | 0 |
| L. caudatum | CBS58485 | Lca1 | HQ643136 | 0 | AF290309 | 0 | 0 |
| L. myophilum | NJM-8403 | Lm1 | AB285497 | 0 | AF290311 | 0 | 0 |
| L. humanum | ATCC76726 | Lh1 | JX970867 | JX985757 | AF290310 | JX999094 | JX999125 |
| L. giganteum mammals | ATCCMYA4933 | Lgp1 | JF919611 | JF919613 | JX970853 | JX999097 | JX999127 |
| L. giganteum mammals | MTLA-03 | Lgp2 | JX999099 | JF919612 | JX970852 | JX999099 | JX999128 |
| L. giganteum mammals | ATCCMYA4934 | Lgp3 | JX999096 | JF919613 | JX970854 | JX999096 | JX999126 |
| L. giganteum mammals | ATCCMYA4935 | Lgp4 | JX999098 | JF919614 | JX970855 | JX999098 | JX999129 |
| L. giganteum mammals | MTLA-10 | Lgp5 | KJ506111 | KJ506076 | KJ506083 | KJ506097 | KJ506111 |
| L. giganteum mammals | MTLA-12 | Lgp6 | KJ506112 | KJ506078 | KJ506084 | KJ506098 | KJ506112 |
| L. giganteum mammals | MTLA-14 | Lgp7 | KJ506113 | KJ506077 | KJ506085 | KJ506099 | KJ506113 |
| L. giganteum mammals | MTLA-15 | Lgp8 | KJ506116 | KJ506081 | KJ506087 | KJ506103 | KJ506116 |
| L. giganteum mammals | MTLA-16 | Lgp9 | KJ506115 | KJ506080 | KJ506086 | KJ506102 | KJ506115 |
| L. giganteum mammals | MTLA-17 | Lgp10 | KJ506117 | KJ506082 | KJ506088 | KJ506101 | KJ506117 |
| L. giganteum mammals | MTLA-18 | Lgp11 | KJ506114 | JX970841 | KJ506089 | KJ506100 | KJ506114 |
| L. ajelloi | ATCCMYA4936 | La1 | JX970885 | JX970885 | JX970841 | JX999112 | JX999143 |
| L. ajelloi | ATCCMYA4937 | La2 | JX970884 | JX970884 | JX970842 | JX999113 | JX999144 |
| L. ajelloi | MTLA-19 | La3 | KJ506134 | KJ506070 | KJ506092 | KJ506106 | KJ506120 |
| L. ajelloi | MTLA-20 | La4 | KJ506135 | KJ506071 | KJ506094 | KJ506109 | KJ506124 |
| L. ajelloi | MTLA-21 | La5 | KJ506137 | KJ506073 | KJ506095 | KJ506108 | KJ506122 |
| L. ajelloi | MTLA-22 | La6 | KJ506138 | KJ506069 | KJ506096 | KJ506107 | KJ506123 |
| L. ajelloi | MTLA-23 | La7 | KJ506136 | KJ506072 | KJ506093 | KJ506110 | KJ506121 |
| L. albertoi | ATCCMYA4932 | Lal1 | JX970870 | JX970838 | JX970838 | JX999115 | JX999142 |
| L. vilelae | MTLA-24 | Lv2 | KJ506133 | KJ506075 | KJ506091 | KJ506105 | KJ506119 |
| L. vilelae | MTLA-25-(Tissue) | Lv3 | KJ506132 | KJ506090 | KJ506090 | KJ506104 | KJ506118 |
| Pythium insidiosum | MTPI-36 | Pi1 | AY486144 | 0 | 0 | 0 | 0 |
| Pythium grandisporangium | ATCC28295 | Pg1c | AY151182 | 0 | 0 | 0 | 0 |
| Pythium aphanidermatum | STR-135 | Pa1a | AY151180 | 0 | 0 | 0 | 0 |
| Pythium diliense | STR-66 | Pd1a | AY151181 | 0 | 0 | 0 | 0 |
| Pythium flevoense | STR-W041 | Pf1b | EU240168 | 0 | 0 | 0 | 0 |
| Pythium diclinum | STR-P7824 | Pdi1b | EF153675 | 0 | 0 | 0 | 0 |
| Pythium capillosum | CBS222-94 | Pc1b | AY598635 | 0 | 0 | 0 | 0 |
| Pythium sylvaticum | CBS453.67 | Ps1f | AY598657 | 0 | 0 | 0 | 0 |
| Pythium pleroticum | CBS776.81 | Pp1e | AY598642 | 0 | 0 | 0 | 0 |
| Pythium iwayamai | CBS156.64 | Piw1g | AY598648 | 0 | 0 | 0 | 0 |
| Pythium undulatum | CBS157.69 | Pu1j | AY598708 | 0 | 0 | 0 | 0 |
| Pythium ultimum | CBS398.51 | Pul1i | AY598642 | 0 | 0 | 0 | 0 |
| Pythium nunn | CBS808.96 | Pn1j | AY598709 | 0 | 0 | 0 | 0 |
0=no DNA sequences available.
MTLA and MTPI strains.
Biomedical Laboratory Diagnostics, Michigan State.
University culture collection.
The purity of the strains was determined by the induction of zoospores using Mendoza and Prendas24 protocol in culture and then selecting a single colony. The pure isolates obtained by this method were kept in our collection and later used in these analyses. With the exception of MTLA25 (genomic DNA sample extracted from biopsied tissue),30 genomic DNAs were extracted from cultures of isolates grown on 2% Sabouraud dextrose broth (SDB). Temperature studies at 25 and 37°C and the production of sexual and asexual structures were evaluated on brain heart infusion (BHI) (DIFCO, Detroit, MI USA), corn meal agar (CMA) (BBL, Sparks, MD, USA), and 2% Sabouraud dextrose agar (SDA). The different stages of sporangium formation, zoospore cleavage and release were assessed on colonized grass leaves in water cultures containing Ca2+ and other ions as described by Mendoza and Prendas.24 Briefly, the strains in Table 1 were subcultured on SDA plates at 37°C for 24h. After incubation 5mm×5mm blocks were cut from the advancing edges and placed on 2% water agar plates. Sterile 4mm×10mm grass blades were laid on top of the agar blocks and incubated at 37°C for 24–72h, depending upon the time required to parasitize the grass blades. The grass blades were then placed in beakers containing 50ml of sporulation mix. The sporulation mix was made of two solutions: solution number one consists of (NH4)2HPO4 (66.04g), KH2PO4 (68.05g), and K2HPO4 (87.09g) in 500ml of H2O; and the other consists of CaCl2·2H2O (18.38g), and MgCl2·6H2O (25.42g) in 250ml of H2O. The sporulation mix was obtained by mixing 0.5ml of the first solution plus 0.1ml of the second solution in 1.0L of sterile distilled water. Beakers containing 50ml sporulation mix plus parasitized grass blades were incubated at 37°C and the development of sporangia and zoospores was evaluated microscopically at 30min intervals for 6 or more hours. Some strains required additional overnight incubation at 25°C to develop zoospores.
Morphological description of hyphal structures, sporangia, zoospores, and other propagulesThe morphological features were evaluated after subculture on BHI, CMA, and SDA following 24–72h of incubation at both 25 and 37°C. Briefly, 4mm×4mm agar blocks were cut from the inoculated plates and mixed with one drop of lactophenol cotton blue (phenol 20ml, lactic acid 20ml, glycerol 40ml, and distilled water, 20ml) to assess the microscopic features of the hyphae and other structures. The development of sporangia and zoospores were evaluated in sporulation medium (see above). After incubation at the induction temperatures, beakers containing 50ml of sporulation medium and parasitized grass blades were inspected on inverted microscope. Grass blades with more than 10 vesicles that contained zoospores were removed and placed on glass slides containing 5μl of merthiolate (0.02%) to kill the zoospores and facilitate their measurements.
Molecular analysisThe strains (Table 1) were inoculated into 250ml flasks containing 100ml of SDB and incubated for 72h at 37°C on a shaker rotating at 150rpm as per Vilela et al.34 After incubation the cultures were killed with merthiolate (0.02%, wt/vol) and then filtrated to recover the hyphal cell mass. The hyphal cell mass was then transferred to a mortar and ground in the presence of liquid nitrogen. The DNA from the disrupted hyphae was treated with sodium dodecyl sulfate and proteinase K and then incubated at 60°C for 1h and their genomic DNA extracted with phenol, chloroform, isoamyl alcohol following Sambrook and Russell protocol.30 PCR was by hot start amplification of the complete ITS sequences and the partial coding DNA sequences of cytochrome oxidase II (COXII); heat shock protein 90 (HSP90); cell division cycle 42 (CDC42); and tubulin (TUB), as per Vilela et al.34 (Table 1). Briefly, the PCR temperature cycling parameters were as follows: initial DNA denaturation for 10min at 95°C followed by 40 cycles of DNA denaturation for 1min at 95°C, annealing for 1min at 60°C, and elongation at 72°C for 2min, concluded by a final elongation of 2min at 72°C. The amplicons were ligated into pCR™ 2.1-TOPO® vector (Invitrogen, Carlsbad, CA, USA), purified and then sequenced using BigDye Terminator chemistry in an ABI Prim 310 genetic analyzer (Perkin-Elmer, Foster City, CA, USA).
Phylogenetic analysisEach of the sequenced genes was analyzed independently in combination with sequences of Lagenidium spp. and Pythium spp. available at the National Center for Biotechnology Information (NCBI). Concatenated genomic DNA sequences from the studied strains containing the ITS sequences and the partial gene sequences of the selected four exons (CDC42, COXII, HSP90, and TUB) were aligned with Lagenidium and Pythium spp. groups a–c, e–g, i and j22 DNA sequences from NCBI using CLUSTAL W, v. 1.81 with default settings followed by a visual inspection (Table 1). Dashes (-) were used to annotate missing nucleotide data especially in Pythium spp. CDC42, COXII, HSP90, and TUB exons. The aligned sequences were exported for parsimony analysis using heuristic search with TBR branch swapping and distance analysis by Neighbor-Joining (MEGA-6, Tamura, Stecher, Peterson, Filipski, and Kumar). Large insertions were coded as one event by excluding all but one nucleotide per insertion and the resulting gaps were treated as missing data. Neighbor-Joining analysis used one of four approaches: uncorrected distances, maximum-likelihood estimates of distance with a time reversible model (6ST), empirical base frequencies with no rate variation among sites, or a gamma distribution with four rate categories and a shape parameter of 0.5. Branch support was estimated as the percentage of trees from resampled data sets containing the branch, using either parsimony (1000 resampling, heuristic, nni branch swapping) or Neighbor-Joining trees (1000 resampling, maximum-likelihood distances). Concatenated DNA sequences of the five investigated loci were used also in Bayesian analysis as per Vilela et al.34 Briefly, the test was conducted in MrBayes v3.2.1 ×64 using the GTR+I+gamma model, with two chains (one heated), two runs, sampling every 100th generation for 1×106 generations, and exclusion of the first 2.5×105 samples (representing the burn-in) prior to analysis. Support for branches was estimated as the percentage of parsimony trees (1000 resampling, heuristic, nni branch swapping) or Neighbor-Joining trees (1000 resampling, maximum likelihood distances) containing the branch as well as by determining the Bayesian probability estimated as the percentage of Bayesian trees possessing the branch after discarding the burn-in sample.
ResultsBased on the above studies, three novel Lagenidium clades are herein described as containing new species.
Lagenidium ajelloi, Newton, Vilela, Taylor, Walker, Mendoza sp. nov.Holotype=ATCCMYA 4936 (MTLA-07) isolated from a dog with lagenidiosis by Dr. J. Newton, Alabama, USA.=Lagenidium karlingii, sensu. Grooters, Proceedings, 25th Annual American College of Veterinary Internal Medicine Forum, 2007.=Paralagenidium karlingii, Grooters, Grooters, Spies, Chen, Glockling, Lévesque, de Cock. Nomenclatural novelties. Index Fungorum No. 34, 2013. Nomen nudum (http://www.bgbm.org/iapt/nomenclature/code/saintlouis/0001ICSLContents.htm; article 36)Etymology=In recognition of the numerous medical and veterinary mycological contributions of our friend and mentor (LM) Dr. Libero Ajello.
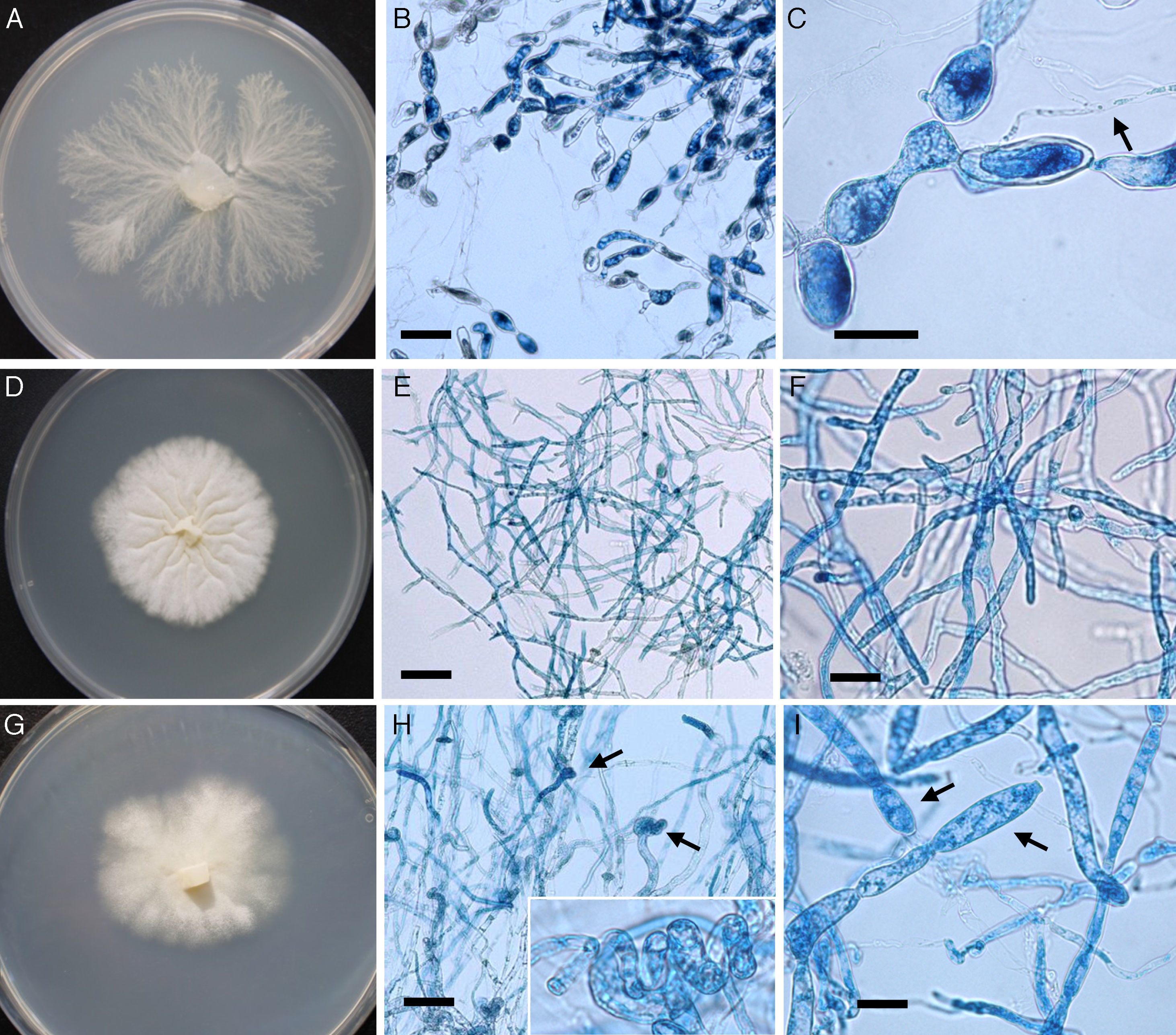
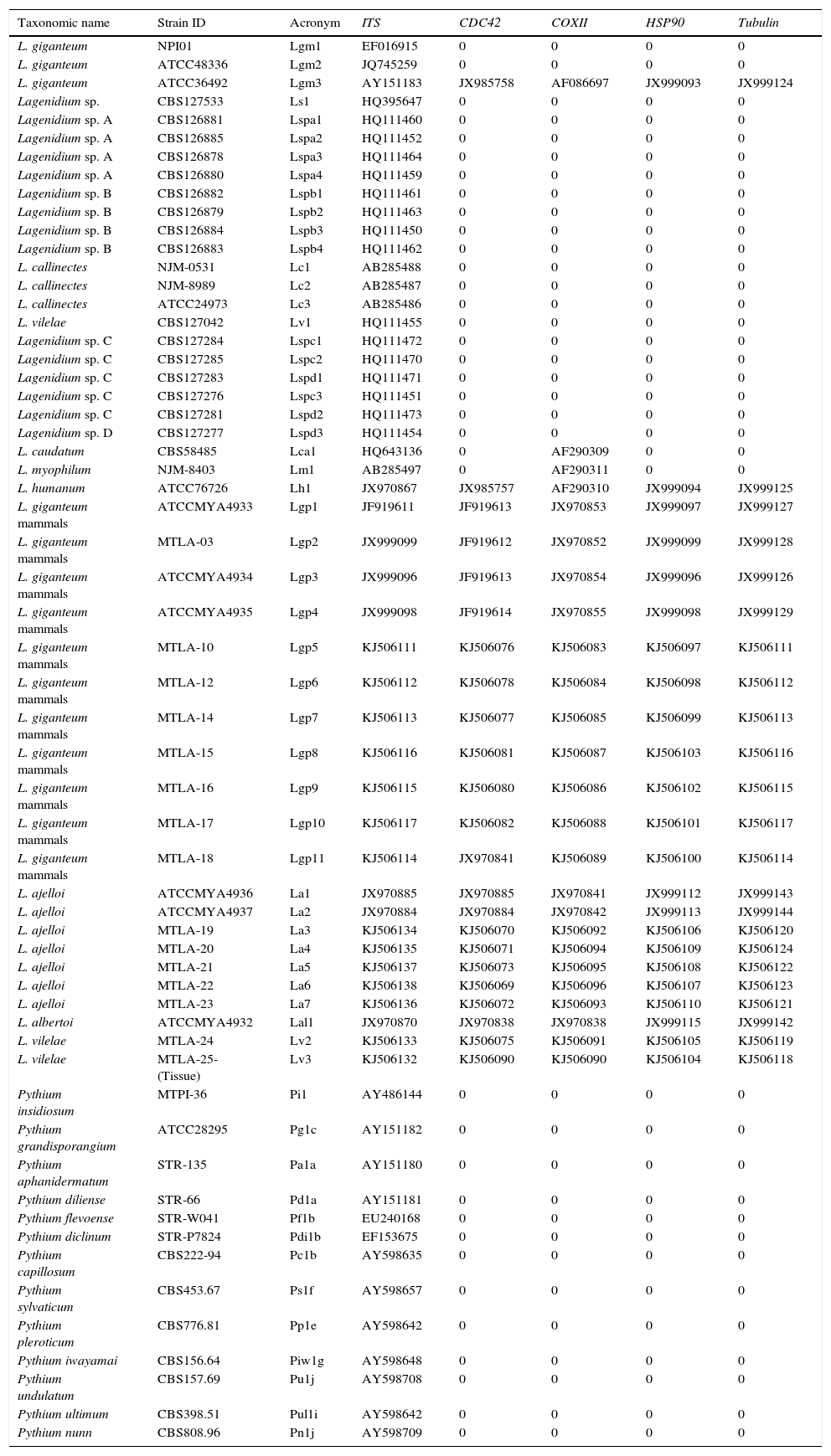
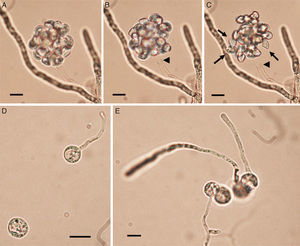
/ L. ajelloi (MTLA07) developed similar colonies on all media and showed slower growth rate at 37°C (∼2 to 4mm/day, diameter of the entire colony) and 25°C (0–1mm/day) compared to that of L. albertoi or L. vilelae \). Macroscopic and microscopic features were based on L. ajelloi strain MTLA07 subcultured on SDA. On this medium L. ajelloi developed yellowish, submerged, radiate colonies with few aerial hyphae. Microscopically, L. ajelloi developed a filamentous thallus strongly constricted at septa, resulting in the formation of long, branched chains of 60–600μm, with chains of cylindrical to ellipsoid detached segments (Fig. 1B,C). However, this feature was lost after several subcultures. In addition, L. ajelloi isolates displayed the presence of hyaline sparsely septate mycelioid hyphae (6–16μm) resembling fungal mycelia (Fig. 1C arrow). The strain did not form oogonia.
The figure depicts the macroscopic and microscopic features of the three novel mammalian Lagenidium species. Lagenidium ajelloi (Panels A–C) is characterized by the formation of submerged colonies with a branching radiate pattern on SDA (A). Microscopically L. ajelloi displays branching mycelioid structures strongly constrict at septa resulting in the formation of elongated smooth and ovoid segments (Panel B, Bar=80μm and Panel C, Bar=100μm). The arrow in Panel C shows a coenocytic hypha of L. ajelloi commonly found along with ovoid segments. The L. albertoi colony macroscopic features are shown in Panel D. Microscopically, this species developed mostly hyaline branching hyphae (E, Bar=60μm, F, Bar=20). The culture macroscopic feature of L. vilelae is shown in Panel G. Panels H and I depict the development of branched hyphae strongly constricted at the septa sometimes forming disarticulate elongate segments. The presence of numerous coiled hyphae was also found (Panel H arrows). Lower Panel H shows a close-up of the coiled hyphal structures. Long elongate hypha-like structures constricted at septa, as those depicted in Panel I were more common in liquid cultures (Panel I, arrows) (H, Bar=13μm and Panel I, Bar=15μm).
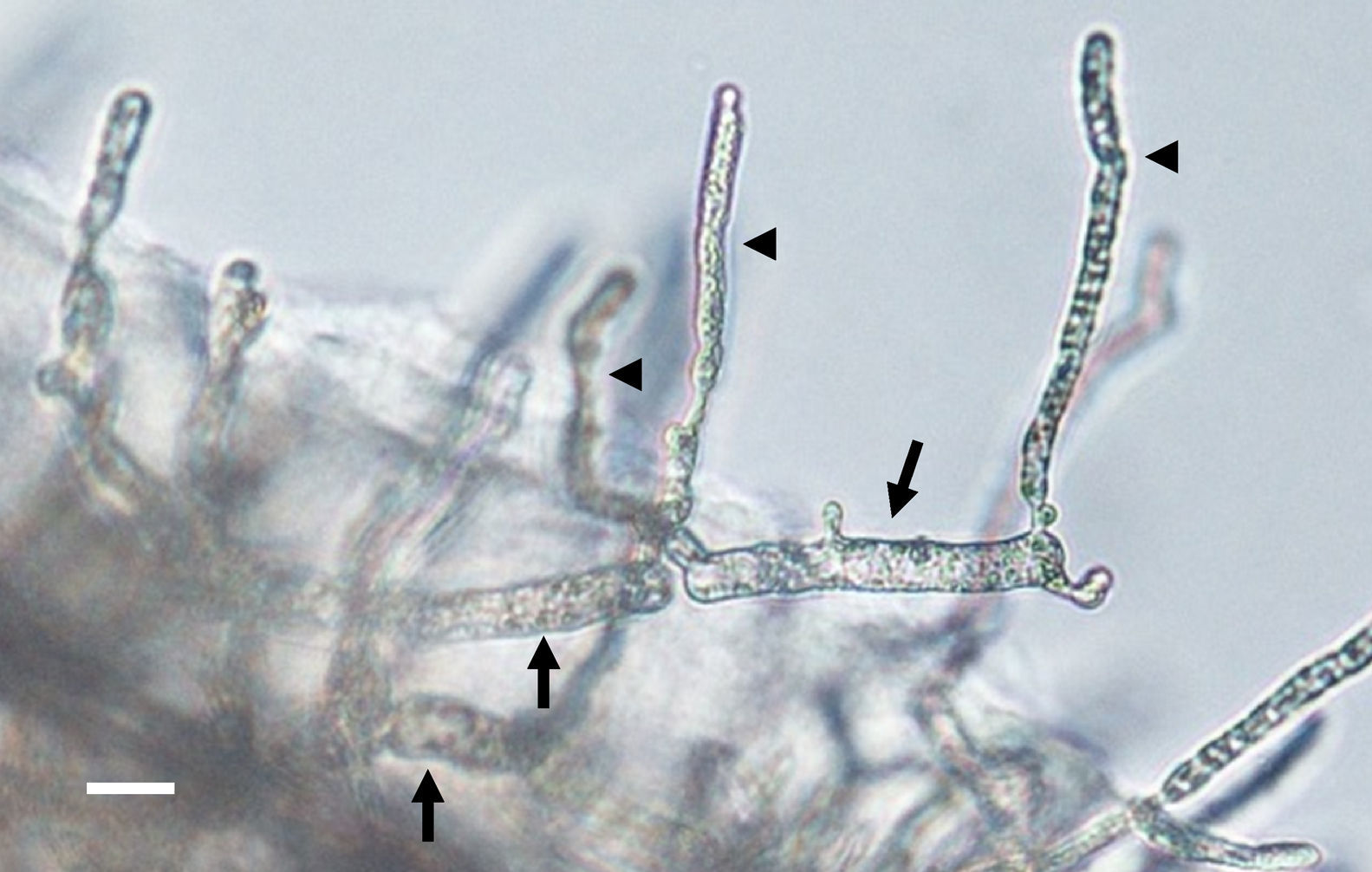
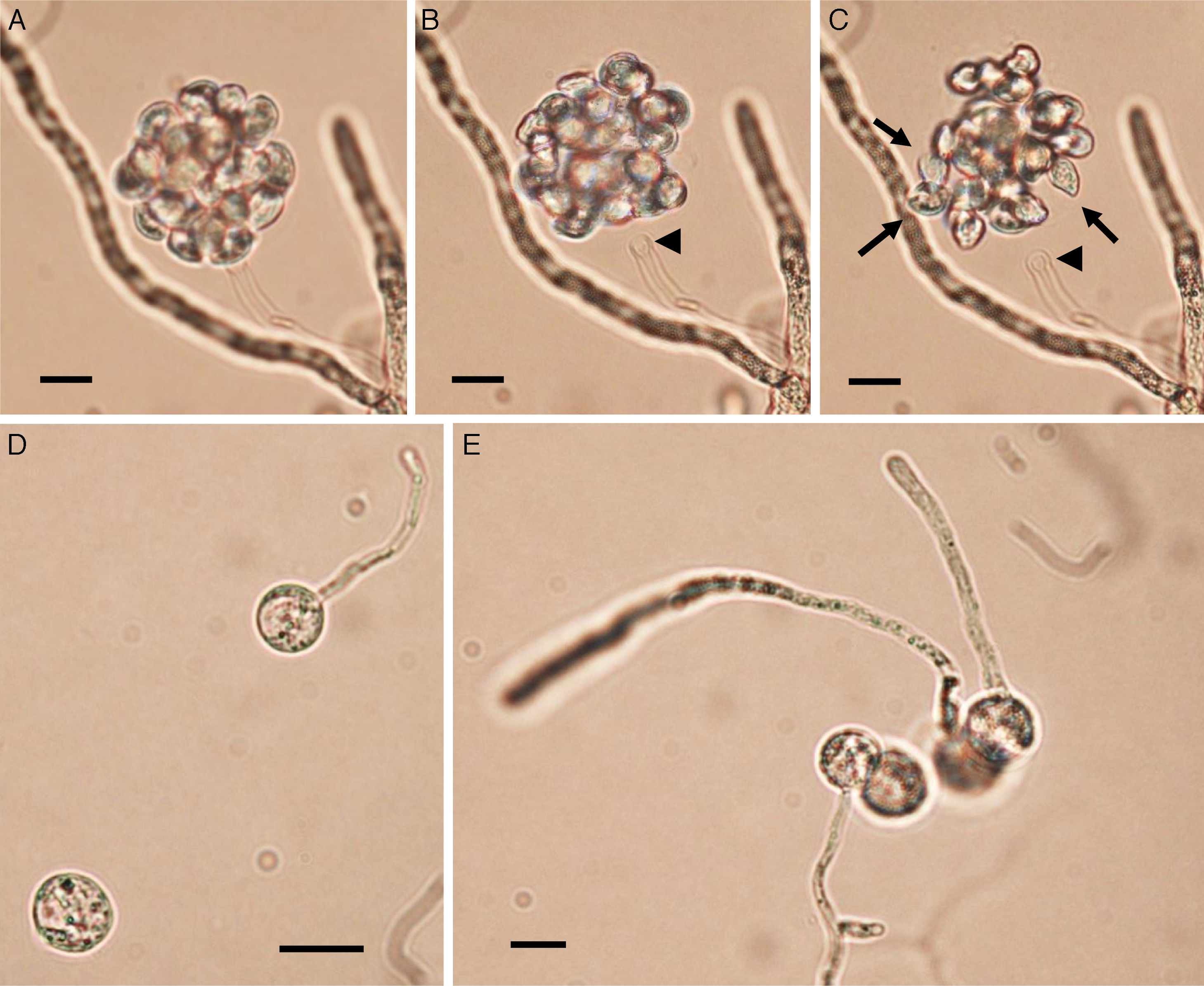
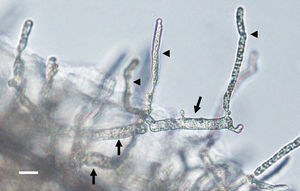
Zoospore formation of L. ajelloi in induction medium at 37°C took more than 24h. The development of one or more exit tubes from branched or detached ∼40μm×120μm in diameter sporangia was the first step (Fig. 2). First, the nuclei and cytoplasm flowed from the sporangia (Fig. 2, arrows) through exit tubes (Fig. 2, arrow heads) to form vesicles 35–65μm in diameter (Fig. 3A–C). Vesicles which formed on the grass blades harbored 30–45 reniform (11–19μm×8–10μm) biflagellate zoospores (Fig. 3A–C arrow). After vesicle formation, zoospores developed fully within 20min. The detachment of the entire vesicle from the tip of the hyphae was sometimes observed (Fig. 3B,C). The zoospores were released after disruption of the vesicle's cell wall by active zoospores. After release they swam for the next 25min before the formation of spherical cysts (11–16μm in diameter), which germinated by forming hyphal tubes (Fig. 3D,E) (Table 2).
The figure depicts the development of articulate and disarticulated sporangia (arrows) of Lagenidium ajelloi on a grass blade in induction medium showing the formation of slender discharge tubes (arrow heads). The development of exit tubes from elongate sporangia (arrow heads) gives rise to vesicles at the tip of the each exit tube, as shown in Fig. 3 (Bar=40μm). L. albertoi and L. vilelae developed also similar sporangia structures in induction medium before zoospore formation.
Panels A–C show Lagenidium ajelloi vesicles containing zoospores on grass blades (see Fig. 2) after incubation in sporulation medium at 37°C (Bar=45μm). Fully develop zoospores before release are shown in Panel C (arrows). The detachment of the whole vesicle from the tip of the exit tube (Panels B and C arrow heads) just before zoospore release was also observed in some of the strains. Note the zoospore reniform shape (Panel C, arrows) before breaking out from vesicle. Panels D and E show a group of encysted spores one of them developing long germ tubes (Bar=15μm). Panel E shows the encysted zoospores developing long hyaline coenocytic hyphae with some branches after several hours of encystment. L. albertoi and L. vilelae also developed their zoospores and cysts in the same fashion.
Microscopic features of the mammalian pathogenic Lagenidium spp. including their appearances and structure on 2% Sabouraud dextrose agar and in induction zoospore medium.
| Strains | Hyphae | Structures | Vesicle | Zoospore | Encysted zoospore | Oospore |
|---|---|---|---|---|---|---|
| L. ajelloi sp. nov. ATCCMYA 4936 type strain | 6–12μm | Elongate chains of oval segments 60–600μm | Spherical/oval 35–65μm | 11–19μm by 8–10μm | 11–16μm | Not observed |
| L. albertoi sp. nov. ATCCMYA-4932 type strain | 8–15μm | Mainly branched hyphal structures | Spherical 27–65μm | 12–18μm by 8–10μm | 11–18μm | Not observed |
| L. vilelae sp. nov. (MTLA-24) type strain | 7–16μm | Few 17 to 300 μm elongate hyphal structures | Spherical 25–65μm | 12–17μm by 8–10μm | 11–18μm | Not observed |
Holotype=ATCCMYA-4932 (MTLA13) strain isolated from a Thai man with ocular lagenidiosis by Dr. Ariya Chindamporn. Reinprayoon U, Permpalung N, Kasetsuwan N, Plongla R, Mendoza L, Chindamporn A, Lagenidium sp. ocular infection mimicking ocular pythiosis. J. Clin. Microbiol. 2013;51:2778–2780.=Lagenidium sp. sensu Reinprayoon U, Permpalung N, Kasetsuwan N, Plongla R, Mendoza L, Chindamporn A. Lagenidium sp. ocular infection mimicking ocular pythiosis. J Clin Microbiol. 51:2778–2780, 2013.Etymology: Dedicated to an author's relative (LM).
L. albertoi (MTLA13) developed similar colonies on all media and grew comparatively faster at 37°C (8mm/day) and 25°C (3mm/day) (Table 2) than L. ajelloi but similar to L. vilelae. On SDA at 37°C L. albertoi developed white to yellowish submerged colonies with few aerial hyphae and folds radiating from the center of the colony (Fig. 1D). The main microscopic features were hyaline, 10–18μm filamentous, branched mycelioid structures with few septa (Fig. 1D–F). In liquid medium (SDB), the mycelioid structures developed constrictions at the septa with the occasional formation of long segments. Coiled hyphae were also detected on CMA and SDA. Oogonia were not found.
In induction medium L. albertoi developed 45–95μm detached segments (sporangia) with exit tubes very similar to L. ajelloi (Fig. 2). Vesicles 25–65μm in diameter contained biflagellated zoospores after 6h of incubation (37°C) near the end of the exit tubes. The number of vesicles harboring zoospores on grass blades was greater than those found using the same methodology in L. ajelloi or L. vilelae. Some vesicles detached from the opening of the exit tube just before zoospore release as described in L. ajelloi (Fig. 3C). The zoospores were similar in shape and size (12–18μm×8–10μm) to L. ajelloi and L. vilelae (see below). Upon release, zoospores swam 20–40min and then formed spherical cysts (11–18μm in diameter) and developed coenocytic germ tubes as described in L. ajelloi (Fig. 3).
Lagenidium vilelae, Lewis, Vilela, Taylor, Walker, Mendoza sp. nov.Holotype=MTLA24 isolated from a cat with lagenidiosis, North Carolina, USA submitted to us by Dr. John Lewis.=L. deciduum, Chi Y. Chen, Grooters, Spies, de Cock, Lévesque. Nomenclatural novelties. Index Fungorum No34, 2013. Nomen nodum (http://www.bgbm.org/iapt/nomenclature/code/saintlouis/0001ICSLContents.htm; article 36)Etymology: Dedicated to an author's family member (RV).
L. vilelae strain MTLA24 had a moderate growth rate at 37°C (9mm/day) and more slowly at 25°C (4mm/day) on all media. This strain developed submerged colonies with a white to yellowish radiate pattern with few aerial hyphae (Fig. 1G). In a rich medium such as BHI the colony morphology showed similar patterns, but the presence of aerial hyphae was more apparent. Microscopically, L. vilelae showed hyaline hyphae (7–18μm) strongly constricted near septa (Fig 1H,I arrows) with few lateral branches and the presence of occasional cylindrical, smooth (17–300μm long) hyphal segments on fresh samples. The finding of short- and long-coiled-hyphae was also detected in subcultured strains, especially on CMA and occasionally on the other media (Fig. 1E arrows, lower section). When L. vilelae cultures on grass blades were placed in induction medium the hyphae developed detached or connected sporangial segments containing one or more exit tubes from which 35–65μm in diameter vesicles were formed. The vesicles contained numerous reniform, 12–17μm×8–10μm biflagellate zoospores, similar to that depicted in Fig. 2. Oogonia were not found.
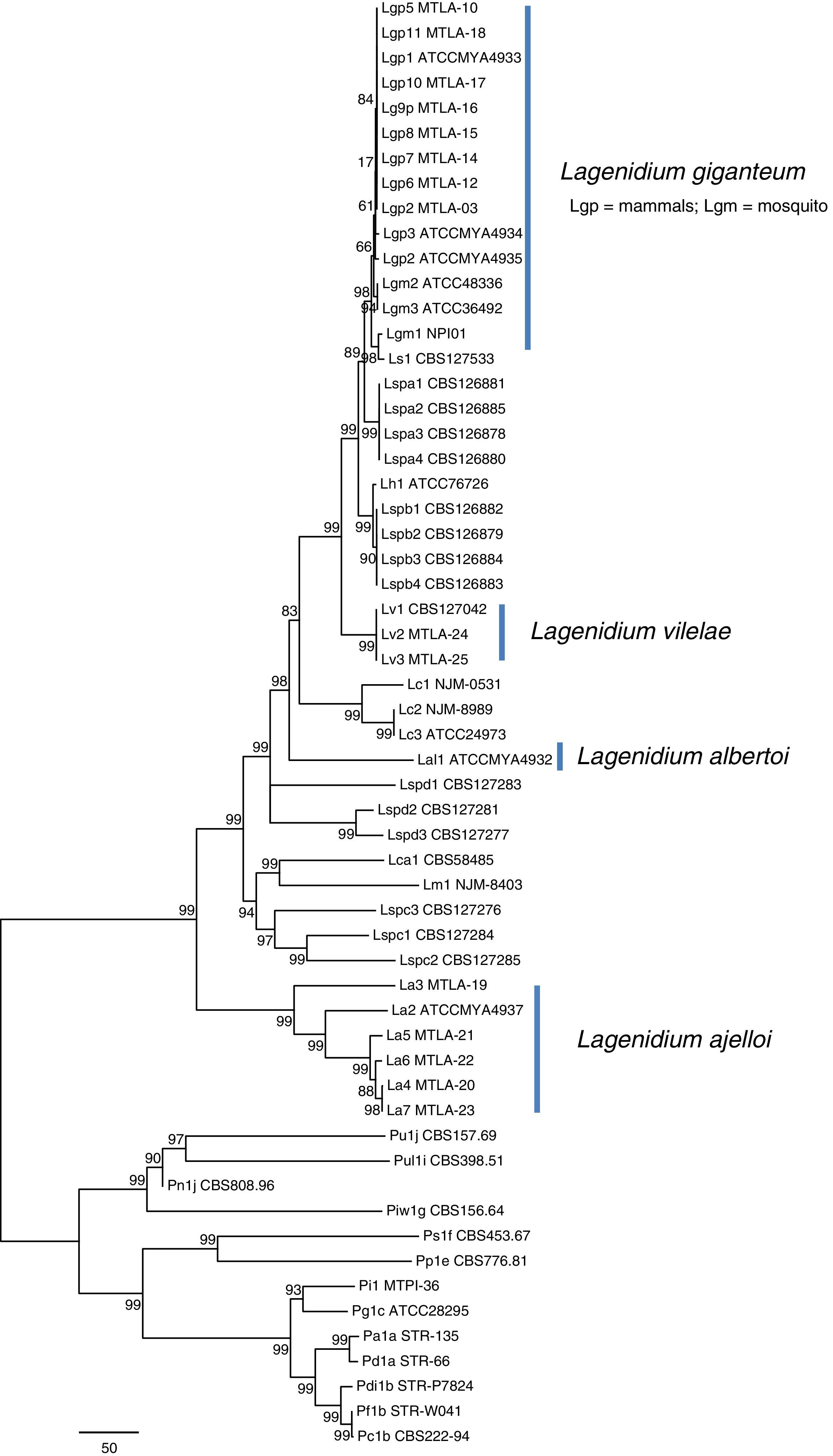
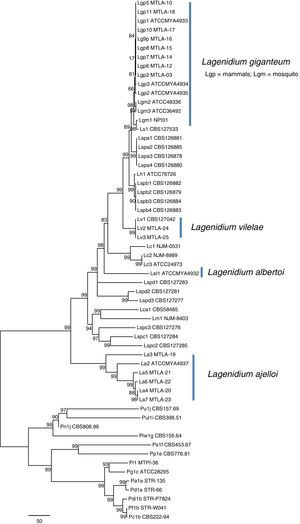
Phylogenetic features of the mammalian-pathogenic Lagenidium speciesPhylogenetic analysis by either parsimony or neighbor-joining of the ITS sequences or each the four exons showed no topological conflicts. Bayesian analysis of the ITS sequences and the four exons consistently placed the Lagenidium strains into four clades as described by Vilela et al.34 In Maximum Parsimony analyses, the thermo-tolerant L. giganteum (mammals) and thermo-sensitive L. giganteum (mosquito) form a monophyletic clade located at the crown of the genus (Fig. 4). These taxa, plus L. humanum (Lh1), and two unnamed entomophagous clades (Lspa and Lspb) were the sister taxon to L. vilelae (Lv). These clades plus L. vilelae form a larger strongly supported clade, sister to the cluster formed by L. albertoi (Lal1), L. callinectes (Lc 1 to 3), L. caudatum (Lca), L. myophilum (Lm) and three entomophagous Lagenidium isolates (Lspc1-3). L. ajelloi (La) formed a strongly supported taxon sister to the described clades (Fig. 4). An entomophagous Lagenidium sp. strain accession number HQ111455=CBS 127042 (Lv1, Table 1) clustered with strong bootstrap support with the two DNA sequences of L. vilelae (Fig. 4). In Bayesian and Parsimony analyses L. albertoi (Lal1) always grouped on a single clade (data not shown, Fig. 4).
Unrooted Maximum Parsimony phylogenetic tree based on sequence analysis of the complete rDNA ITS and 4 exons: CDC42, COXII, HSP90, and TUB concatenated data set showing the relationship among isolates of L. ajelloi, L. albertoi, L. vilelae with the data available in NCBI on other Lagenidium and Pythium species, as listed in Table 1. The numbers on the internal branches are percentage of trees based on 1000 bootstrapped data sets possessing the branch. The branch lengths correspond to corrected distances. In the tree, L. ajelloi, L. albertoi, and L. vilelae clustered in their own clades with strong bootstrap support. Several clusters of unnamed Lagenidium entomophagus species (Lspa, Lspb, Lspc, and Lspd) appeared also in their own clades. In addition, L. giganteum (Lgm and Lgp), L. callinectes (Lc), L. humanum (Lh), L. caudatum (Lca) and L. myophilum (Lm) all aligned within the Lagenidium monophyletic taxon with varied support. The scale bar represents number of substitutions per nucleotide.
Our phylogenetic analyses and that of Vilela et al.34 support the placement of the three new mammalian Lagenidium species basal to L. giganteum types recovered from mammals and mosquitoes. Thus, we propose three novel Lagenidium species based on the following: (a) except for the mammalian pathogen L. giganteum,34 there is no a formal description of other Lagenidium species pathogenic for mammals and (b) our phylogenetic data placed these unique Lagendium species in three well supported taxonomic clades. Thus, they represent previously undescribed species. Concatenate Bayesian and Maximum Parsimony analyses of the 25 Lagenidium gene sequences available at the NCBI and the 21 gene sequences from the strains in this study placed the pathogen of blue crabs L. callinectes (Lc) (placed by Dick11 in the genus Salilagenidium), the saprotrophic L. humanum (Lh1) (placed by Dick11 in the genus Myzocytiopsis), and eight yet to be named entomopathogenic Lagenidium spp. (Lspa and Lspb) from Taiwan basal to L. giganteum types (Lgm and Lgp). The mammalian pathogen L. vilelae (Lv) (including an entomophagous strain from Taiwan,15 (Lv1) formed a strongly supported taxon basal to the Lspa and Lspb entomophagous clades. L. albertoi (Lal1) was strongly supported in its own clade, suggesting this strain is unique among mammalian Lagenidium species. Similar results were recently reported using Bayesian analysis.34 The clade formed by three entomopathogenic Lagenidium spp. from Taiwan (Lspc1-3), the pathogens of nematodes L. caudatum (Lca) (placed by Dick11 in the genus Pythium), and shrimps L. myophilum (Lm) (placed by Dick11 in the genus Salilagenidium) were sister to the seven L. ajelloi (La) species recovered from mammals. The finding of Lagenidium spp. forming a monophyletic cluster to Pythium spp. challenges Dick11 decision to remove a number of Lagenidium species to other straminipilan groups.3 Surprisingly, of the 46 Lagenidium spp. in the study, only one has not yet been reported as an animal pathogen, indicating that the capacity to infect animals, including mammals, is deeply rooted in the genus.14,34 This and other studies suggest members of the genus Lagenidium are well equipped with virulence factors to infect animals including mammalian hosts.
Recently Grooters et al.15 proposed the genus Paralagenidium based on a single strain isolated from an USA dog. We had used their ITS DNA sequence in phylogenetic analysis (data not shown) and found that their KF915306 is analogous to the DNA sequences of L. ajelloi in this study. Grooters et al.15 proposal of the genus Paralagenidium was based on a single strain and two genes (COXI, ITS). But, the finding in this study and that of Vilela et al.34 indicate that the genus Lagenidium is monophyletic, thus the proposed genus Paralagenidium is not supported in these analyses and thus, considered a nomen nudum. Likewise, the Taiwan nematode strain L. deciduum (HQ111455) described by the same authors15 clustered with the two of our L. vilelae DNA sequences from mammals (Lv2, Lv3). Vilela et al.34 called the attention on two populations of L. giganteum: a thermo-sensitive (mosquito pathogen) and thermo-tolerant (mammalian pathogen). Although L. deciduum (HQ111455) clustered with L. vilelae in our analysis (data not shown), and the fact this strain was recovered from nematodes suggests that our L. vilelae strain from mammals may be a different type as is the case with L. giganteum and therefore, its true relationship with the mammalian L. vilelae strains is dubious at best. Thus, the legitimacy of these epithets is challenged under “The International Code of Botanical Nomenclature” Chapter V article 52 and 58.1. http://www.bgbm.org/iapt/nomenclature/code/saintlouis/0001ICSLContents.htm; and by the “International Code of Nomenclature for algae, fungi, and plants (Melbourne Code)” Chapter V article 36 http://www.iapt-taxon.org/nomen/main.php?page=title.
The newly-described Lagenidium species can be separated from other closely related Oomycota genera. For instance, with the exception of P. insidiosum and P. aphanidermatum,13 none of the other Pythium, Phytophthora, and Phytopythium species have been found to cause infections in mammals.11,12,19 Also, no other species in the Oomycota have been found to infect mammals except P. insidiosum, P. aphanidermatum,13 the thermo-tolerant species of L. giganteum,32 and the three novel species in this study. Furthermore, besides its pathogenic capabilities, our mammalian Lagenidium species in liquid cultures develop mycelioid structures strongly constricted at septa that soon became sporangia. Although Pythium and some other species can also develop this feature, in Lagenidium spp. these segmented structures become sporangia. The genera Myzocytium and Myzocytiopsis differ from our strains in that they have not been reported to cause disease in mammals.5,11,12,14,19 In addition, these species develop very short, endobiotic, and articulate or sometimes disarticulate mycelioid bodies (unicellular sporangia) with sexual oogonia within their infected hosts.5,20,25 None of our strains developed oogonia in their infected hosts. Likewise, our strains can be also differentiated from the genus Lagenocystis (Lagena) (Lagena nomen nudum, Foraminifera; see Williamson35) by their lack of sexual reproduction in the infected hosts and in culture.6 Additionally, the unique endobiotic holocarpic thalli of Pythiella7,26 (placed in the genus Pleocystidium by Dick11) set this genus apart from the three Lagenidium species in this study.6,18,25
Lagenidium species are well known pathogens of animals such as insects, crab, fish, nematodes and others.1,8,9,11,12,19,29 With the exception of L. humanum recovered from dead skin of humans and snakes,19 the other members of the genus typically parasitize invertebrates and, as recently discovered, mammals.16,17,23,28,34 Did the species pathogenic for mammals evolve from zoopathogenic ancestors infecting invertebrates to become mammalian pathogens? This kind of host switching across kingdoms has been postulated for the Oomycota and indeed there is strong scientific evidence supporting these views.9,18P. insidiosum for instance, was recently recovered from an infected mosquito in India.31 Otherwise, P. insidiosum infection in mosquitoes has rarely been observed, compared with the increasing numbers of new cases of pythiosis diagnosed every year in mammals inhabiting tropical and subtropical areas of the world.13 It is then quite possible that the mammalian pathogenic Lagenidium species, like other Oomycota species, evolved from marine ancestors to later acquire traits to infect plants, and those capacities were adapted later to infect lower animals and more recently evolved to affect mammals including humans.3,4,32 Vilela et al.34, based on the phylogenetic finding of several Lagenidium clades, concluded that the “trait of infecting mammals has arisen several times, independently, in the genus Lagenidium”, a finding supported also by our phylogenetic data.
Gene transfer has been implicated in the gaining of new virulence factors and several mechanisms of disease in the Oomycota.2,33 A recent study of L. giganteum transcriptome showed the presence of virulence factors from plant pathogenic Oomycota species that were found in other peronosporeans such as Phytophthora and Pythium species.21,27 However, the source of virulence factors related to mammalian infectivity from within the Oomycota, but outside of the genus Pythium, is unknown. Based on our data and that of Vilela et al.34 two putative scenarios to explain the sudden emergence of thermo-tolerant L. giganteum as a pathogen of mammals can be postulated: (1) the release of thermo-sensitive L. giganteum during field studies increased the population of this organism in the target ecological niches. The elevated numbers of the biocontrol organisms increased the likelihood of interacting with mammals, and perhaps allowed the mosquito pathogen also interacts with other Oomycota species and fungi, thus facilitating the acquisition of novel traits by horizontal gene transfer, or (2) the thermo-tolerant (mammals) and the thermo-sensitive (mosquito-larvae) strains are simple individuals with different phenotypes in one or more populations of L. giganteum in nature. It is then quite possible that L. giganteum (thermo-tolerant) wild-type strains may have been accidentally selected and used for field studies by mosquito control investigators. Although these hypotheses could explain in part the emergence of this species as a pathogen of mammals, the true ecological pressures leading the ancestor of the mammalian pathogenic Lagenidium species to gain new features infecting mammals have yet to be investigated.
Conflict of interestThe authors declare no conflict of interest.
This research was support in part by the Department of Microbiology and Molecular Genetics, Michigan State University and by Solid Tech Animal Health, Inc., Newcastle, Oklahoma, USA.