To determine the pharmacological measures implemented for secondary prevention in patients with acute coronary syndrome in order to identify whether the implemented management corresponds to recommended clinical practice guidelines and to intervene in those cases where incomplete treatments are found.
MethodsA pre- and post-quasi-experimental study was performed in patients with acute coronary syndrome who were affiliated with the Colombian health system. The patients were monitored for one year from the occurrence of acute coronary syndrome, and all dispensed medications were reviewed. For those patients in whom a lack of a prescription (β-blockers+renin-angiotensin-aldosterone system inhibitors (RAASi)+dual antiaggregation+statin) was identified, an intervention was performed with their treating physicians, showing the analysis of each case, the missing medication, and the evidence supporting the recommendation. The results were measured three months later.
ResultsA total of 829 patients with acute coronary syndrome who underwent percutaneous coronary intervention (90.1%) or coronary bypass (9.9%) were identified. The mean age was 63.8±10.6 years and 73.1% were men. The recommended pharmacological therapy was completed in 729 patients (87.9% of cases). The intervention performed on the remaining 100 patients was able to add the missing drug in 23.0% of the cases. Statistical analysis showed no significant differences with the drug that should have been initiated nor with the success of the intervention.
ConclusionThe majority of patients with acute coronary syndrome are adequately treated after percutaneous intervention with medications recommended by the guidelines. Limited success in the adjustment of the management acute coronary syndrome was achieved following the recommendations given to the responsible physicians.
Determinar las medidas de prevención secundaria implementadas en pacientes que sufrieron un síndrome coronario agudo para identificar si corresponden con guías de práctica clínica e intervenir casos con tratamientos incompletos.
MétodosEstudio cuasiexperimental, antes y después, en todos los niveles de atención, en el que se incluyeron pacientes que sufrieron un síndrome coronario agudo, afiliados al sistema de salud de Colombia. Se hizo seguimiento un año (1 enero y 31 diciembre de 2014) a partir del episodio del síndrome coronario agudo y se revisó toda la medicación dispensada. En quienes se identificó falta de prescripción (β-bloqueadores+ISRAA+antiagregación dual+estatina) se realizó una intervención sobre sus médicos tratantes mostrando el análisis de cada caso, el medicamento faltante y la evidencia que avala la recomendación. Tres meses después se midieron los resultados.
ResultadosSe hallaron 829 pacientes con síndrome coronario agudo sometidos a intervención coronaria percutánea (90,1%) o baipás coronario (9,9%). La media de edad fue 63,8±10,6 años y 73,1% fueron hombres. La terapia farmacológica recomendada se cumplió en 729 pacientes (87,9% de casos). La intervención hecha sobre los 100 pacientes restantes logró que agregaran el fármaco faltante en 23,0% de casos. El análisis estadístico no mostró diferencias significativas con el fármaco que debía iniciarse ni con el éxito de la intervención.
ConclusiónLa mayoría de pacientes que sufrieron un síndrome coronario agudo están tratados de manera adecuada después de la intervención percutánea, con medicamentos recomendados por las guías. Se logró un limitado éxito en el ajuste del manejo tras las recomendaciones dadas a los médicos responsables.
Cardiovascular diseases are currently the leading cause of death in industrialized countries and are expected to occupy the first place in developing countries in 2020.1 Several factors have influenced their increase, such as increased life expectancy, smoking, sedentary lifestyle, and bad eating habits.2,3 The situation in Colombia is similar, with ischemic heart disease being the leading cause of death for both men and women older than 45 years, surpassing even violent deaths and cancer.3,4
The optimal management of acute coronary syndrome includes the immediate relief of the ischemia and the prevention of adverse outcomes, such as heart attack and death. In developed countries, mortality from acute myocardial infarction (AMI) has decreased by almost 30% over the last two decades.4,5
Secondary prevention is especially important because ischemic episodes continue to occur at a high rate after the acute phase. Data show that up to 20.0% of patients are readmitted to the hospital subsequent to the acute event and that 18.0% of men and 23.0% of women over 40 years of age die during the first year after the main ischemic episode.4,5 With the widespread use of coronary interventions, fibrinolytic agents, and antithrombotic treatment in addition to secondary prevention, the total mortality per month has been reduced to between 4.0% and 6.0%.4 This result demonstrates the importance of antithrombotic treatment in the long-term clinical outcome. Moreover, in light of the current evidence, all measures and treatments with proven efficacy should be offered to patients to favorably impact the course of the disease.5,6
Currently, high quality evidence supports the use of medications for secondary prevention, including acetylsalicylic acid (ASA), clopidogrel, β-blockers, angiotensin-converting enzyme inhibitors (ACEI), angiotensin-II receptor blocker (ARB-II), and statins.5,7–15 Additionally, there are proposals within the Screening Tool of Older Person's potentially inappropriate Prescriptions/Screening Tool to Alert doctors to Right Treatment (STOPP/START) that aim to identify patients with potentially inappropriate prescriptions or in whom a particular morbidity is being treated that requires certain medications that are recommended by the best evidence to be omitted.16
The health system of Colombia (SGSSS) offers universal coverage to the population through a payment regimen and another program that is subsidized by the state, which includes a series of health technologies such as interventions on coronary vessels and the medications necessary for acute coronary syndrome treatment through a benefits plan called the Compulsory Health Plan (Plan Obligatorio de Salud, POS). Because the appropriate programs, pharmacological treatments, and adherence to management are fundamental to the success of reducing cardiovascular mortality, we investigated the pharmacological measures implemented as part of secondary prevention in patients with acute coronary syndrome during 2014 to identify whether the management in place adhered to the current recommendations. We intervened in cases where errors, incomplete treatment, or inappropriate management were found to ensure that all pharmacological measures with proven benefit in such patients were put in place.
Materials and methodsStudy design and participantsA pre- and post-quasi-experimental study was conducted in patients who suffered from acute coronary syndrome and who were members of an insurance entity called Entidades Promotoras de Salud (EPS) of the SGSSS between January 1 and December 31, 2014. All patients of any age and gender who were diagnosed with acute coronary syndrome in the main cities of Colombia were enrolled.
Information on the date, type of acute coronary syndrome, and type of procedure performed on the patient (i.e., percutaneous coronary intervention with stent placement or cardiac surgery) was provided by the EPS. Information on the medications prescribed and dispensed to each patient was collected from the database of the entity responsible for dispensing the drugs to the patients (Audifarma S.A.).
Each research subject was monitored for one year from the time of the occurrence of acute coronary syndrome, and all medications dispensed over that period were reviewed.
The following variables were considered:
- 1.
Sociodemographic: age, gender, city, and level of education.
- 2.
Clinical: type of acute coronary syndrome discriminated by the date of presentation of the event and the type of intervention received by the patient (medicated or non-medicated stent placement, cardiac surgery, or other).
- 3.
Pharmacological: medications received in the post-discharge formulation as follows: 1-platelet antiaggregants (i.e., ASA, clopidogrel, prasugrel, and ticagrelor); 2-anticoagulants (i.e., heparin and warfarin); 3-β-blockers (cardioselective and nonselective); 4-renin-angiotensin-aldosterone system inhibitors (RAASIs: ACEI and ARA-II); and 5-hypolipidemics (statins and fibrates).
- 4.
Co-medication: other co-medications that the patients received were evaluated as follows: 1-nitrovasodilators (isosorbide dinitrate); 2-antihypertensive calcium antagonists; 3-other anti-ischemic medications; 4-inotropics (digoxin); 5-antiarrhythmics (amiodarone, others); 6-diuretics (hydrochlorothiazide, furosemide); and 7-antidiabetics (metformin, insulin, others).
A database was developed that considered each cohort of patients according to the type of intervention received for the management of acute coronary syndrome. The prescription relevance and the lack thereof were established in all cases. Based on scientific evidence, patients should be treated with dual antiaggregation therapy, β-blockers, RAASi, and statins. Therefore, patients who received these drugs were considered adequately treated.
In patients who did not receive the complete recommended prescriptions, the clinical history was reviewed to search for the prescriptions received, the type of intervention performed for the acute coronary syndrome, and the relevance of the formulated medications to purge patients with all appropriate drug indications and those who did not have all appropriate indications for all of their prescriptions.
For those cases in which a lack of a prescription for any of the medications was identified, an intervention was performed aimed at those responsible for the health care of the cases in the EPS. This intervention included sending emails, interviewing the physicians, and showing them the analysis of each case, the missing medication, and the evidence supporting the recommendation. Three months later, a measurement of the results of the intervention was performed for each patient to seek changes in the dispensation, specifically of the missing drugs.
The protocol received the approval of the Bioethics Committee of the Universidad Tecnológica de Pereira and respected the principles of patient confidentiality according to recommendations of the Declaration of Helsinki.
Statistical analysisThe IBM SPSS Statistics version 23 for Windows (IBM, USA) statistical package was used for the data analysis. The description of the variables in frequencies and proportions and measures of the central tendency and dispersion were performed. The X2 test for categorical variables was employed. The level of statistical significance was determined as p<0.05.
ResultsA total of 829 patients with acute coronary syndrome who underwent percutaneous coronary intervention (90.1%) or coronary bypass (9.9%) during 2014 in 16 different cities across the country were enrolled. There was a clear predominance of males with 73.1% (n=605) of cases and a mean age of 63.8±10.6 years. In order of frequency, Bogotá was the city with the largest number of patients undergoing coronary intervention (n=264, 31.8%), followed by Barranquilla (n=82, 9.9%), Medellin (n=81, 9.8%), Bucaramanga (n=76, 9.2%), Pereira (n=58, 7.0%), Cartagena (n=57, 6.9%), Ibagué (n=6, 5.5%), and Cali (n=38, 4.6%), accounting for 84.7% of the evaluated population. The remaining patients were distributed in 8 other cities. The diagnosis was AMI with ST segment elevation in 79.0% of cases.
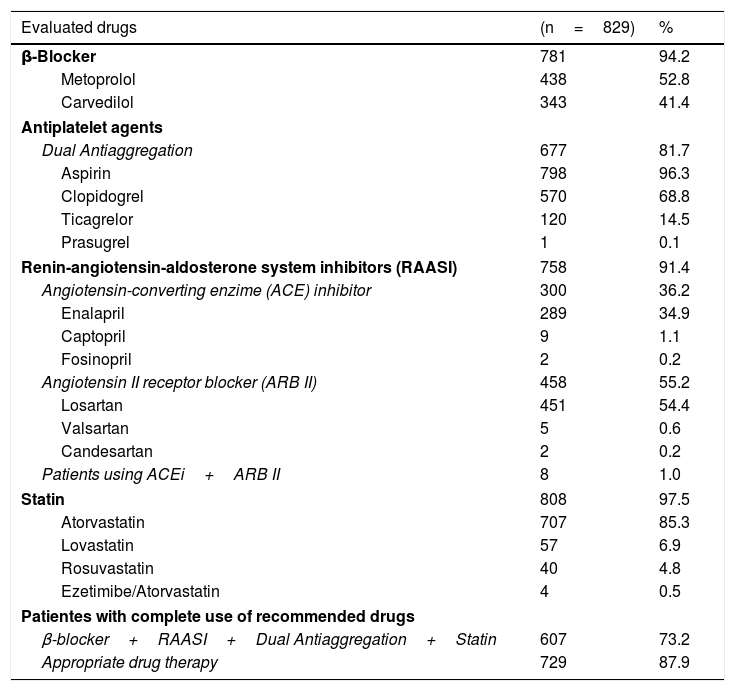
According to the database, the pharmacological therapy recommended by the international guidelines was fulfilled in 73.2% of the cases (β-blockers+RAASi+dual antiaggregation+statin). However, subsequent to the evaluation of all clinical histories, we found that 87.9% of the patients had adequate therapy adjusted for their comorbidities (e.g., anticoagulation in cases of atrial fibrillation) or the type of intervention received (e.g., only a platelet antiaggregant in patients with coronary bypass). Table 1 shows the distribution of the pharmacological therapies of the evaluated subjects.
Medications used in 829 patients diagnosed with acute coronary syndrome following percutaneous intervention or coronary bypass, Colombia 2014-2015.
| Evaluated drugs | (n=829) | % |
|---|---|---|
| β-Blocker | 781 | 94.2 |
| Metoprolol | 438 | 52.8 |
| Carvedilol | 343 | 41.4 |
| Antiplatelet agents | ||
| Dual Antiaggregation | 677 | 81.7 |
| Aspirin | 798 | 96.3 |
| Clopidogrel | 570 | 68.8 |
| Ticagrelor | 120 | 14.5 |
| Prasugrel | 1 | 0.1 |
| Renin-angiotensin-aldosterone system inhibitors (RAASI) | 758 | 91.4 |
| Angiotensin-converting enzime (ACE) inhibitor | 300 | 36.2 |
| Enalapril | 289 | 34.9 |
| Captopril | 9 | 1.1 |
| Fosinopril | 2 | 0.2 |
| Angiotensin II receptor blocker (ARB II) | 458 | 55.2 |
| Losartan | 451 | 54.4 |
| Valsartan | 5 | 0.6 |
| Candesartan | 2 | 0.2 |
| Patients using ACEi+ARB II | 8 | 1.0 |
| Statin | 808 | 97.5 |
| Atorvastatin | 707 | 85.3 |
| Lovastatin | 57 | 6.9 |
| Rosuvastatin | 40 | 4.8 |
| Ezetimibe/Atorvastatin | 4 | 0.5 |
| Patientes with complete use of recommended drugs | ||
| β-blocker+RAASI+Dual Antiaggregation+Statin | 607 | 73.2 |
| Appropriate drug therapy | 729 | 87.9 |
The prescribed co-medications included the use of isosorbide dinitrate (n=137, 16.5% of patients), trimetazidine (n=28, 3.4%), ivabradine (n=6, 0.7%), and other antihypertensives such as amlodipine (n=227, 27.4%), nifedipine (n=28, 3.4%), and verapamil (n=21, 2.5%).
An intervention was performed for the remaining 100 patients who were considered to be missing a prescription for some of the indicated medication. The intervention consisted of sending 19 emails to those responsible for the health care of these patients containing the analysis of each case and explanatory information. Additionally, two meetings were held with 66 medical coordinators of the chronic non-communicable disease services, medical leaders, and the quality auditors of the cities where each subject lived.
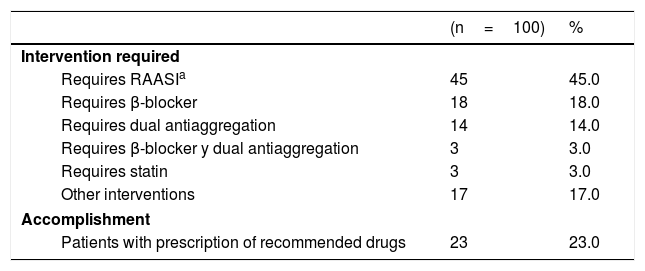
Table 2 shows the principal recommendations for each of the patients who were intervened with this strategy. The most frequent recommendation was the need for the RAASi drug (45.0%). Three months after the intervention, the recommendation was effective in 23.0% of the patients who required a missing medication, leaving the remaining patients without a benefit that could provide them with each of the suggested drugs.
Medications necessary to adjust the management of 100 patients who had an incomplete prescription after suffering from acute coronary syndrome following percutaneous intervention or coronary bypass, Colombia 2014-2015.
| (n=100) | % | |
|---|---|---|
| Intervention required | ||
| Requires RAASIa | 45 | 45.0 |
| Requires β-blocker | 18 | 18.0 |
| Requires dual antiaggregation | 14 | 14.0 |
| Requires β-blocker y dual antiaggregation | 3 | 3.0 |
| Requires statin | 3 | 3.0 |
| Other interventions | 17 | 17.0 |
| Accomplishment | ||
| Patients with prescription of recommended drugs | 23 | 23.0 |
The bivariate analysis using compliance with the proposed recommendation for each patient included in the intervention as the dependent variable showed no significant difference between the pharmacological groups that should have been initiated (RAASi, p=0.867; β-blocker, p=0.186; and dual antiaggregation, p=0.335) and between the cities in which the patients received care. The statistical analysis did not permit the establishment of variables associated with success or failure for the modification of the therapy by the treating physicians.
DiscussionAMI and coronary heart disease are the leading causes of mortality in Colombia and worldwide. However, the morbidity and mortality profile has changed progressively, improving the survival rate and the quality of life of patients due to new and better pharmacological, medical, and interventional therapies for the acute treatment and the subsequent prevention of new events.4–6,16 Therefore, the results of this study achieved the objective of showing an approach for the management of coronary events with secondary prevention in Colombian patients, with this being the first description of this type performed in the region.
Despite the strong evidence and recommendations made by the clinical practice guidelines for the proper prevention of new events after acute coronary syndrome in Colombian patients, there is still room for improvement to achieve the full benefit in all cases.5,16 Similar studies have shown that the problem in these patients resides in the decrease of adherence to treatment during the subsequent 12 months and not to the lack of a prescription for the recommended medications.17,18 This finding underscores the lack of knowledge on the part of the patients and of some clinicians of the importance and benefits of complete and adequate therapy, which opens opportunities for a new topic of interest to study in this population.
A study conducted in Israel estimated that the lack of adherence to the use of RAASi in patients who suffered from acute coronary syndrome could increase mortality by up to 57.0%, whereas failure to use ASA increased mortality by 28.0% and not using statins increased it by 36.0%. The sum of the non-adherent patients compared with those who did adhere to the complete secondary prevention therapy presented an increase in mortality of 38.0%.19 This evidence reinforces the importance of monitoring and compliance to the guideline recommendations as well as the justification for notifying those responsible for the care of these patients to adjust the required therapy.
An important finding was that almost all patients used ASA. The problem with this group of antiaggregants was the lack of combination with another antiplatelet (19.3% of cases) during the first year after the onset of acute coronary syndrome, leaving patients without the possibility of the additional benefits offered by the use of these drugs in conjunction.7,12,14,20–23 RAASi was the group with the second highest lack of prescriptions (8.6%) despite being widely used. There is growing concern about the increased frequency of the use of ARBs above the ACEIs because the latter are the recommended first line medications that have demonstrated their superiority in the prevention of coronary events and mortality by cardiovascular causes. Therefore, ARB-II should be used only as alternatives in patients who cannot tolerate the first drugs.4,5,24,25
The importance of this recommendation is supported by a large amount of evidence on the use of ACEIs in this indication, in addition to better outcomes of interest sought in this type of patient.10,11,15,24–28 Therefore, we expected that these drugs would be used more frequently, which was in contrast to our findings in this study. This discrepancy can be explained by the change in the usage patterns of this group of medications. The use of the ARB-II (especially losartan) progressively grew after their incorporation into the POS until they became the most prescribed drugs in Colombia. This change was driven by physicians who believed that these drugs had the same effectiveness and benefits as the ACEIs but with fewer adverse reactions; this assumption was questioned by several authors.24,25,29
Despite the existing recommendations regarding therapy and the suggestions put forward and made commonplace among the physicians responsible for health care, therapy adjustment was achieved in only 23.0% of the cases. Therefore, the number of patients who do not receive proper pharmacological therapy continues to be troubling because this situation subjects these patients to an increased risk of complications and death.6,10,12,13
Some possible causes for the lack of response to the recommended adjustment may be related to a lack of updating by the clinicians responsible for patient care, the lack of continuing medical education programs focused on cardiovascular issues, and failures in the adherence to clinical practice guidelines that include the joint use of this therapy as the pillar of cardiovascular secondary prevention. However, the results of this intervention do not greatly differ from those found in similar studies with cardiovascular medications for the treatment of arterial hypertension.30,31
This study has some limitations because the information comes from a database of dispensed medications and clinical history records. Therefore, it is not possible to know whether the medication was taken by the patient him/herself and if he/she did so at the recommended doses. Moreover, it is not possible to quantify the frequency of purchase of these cardiovascular medications without a medical formula by some of the patients. No information was collected that indicated the reasons why the patient was not receiving the recommended medication such as intolerance, ejection fraction or functional class. Regarding the results of the intervention, it is not possible to ensure that the medication adjustment was due to the intervention performed by this team in all cases, and we could not determine what other factors might have had an influence. Neither was information obtained about which medical specialty the treating physicians had, nor whether the patients were in a cardiovascular rehabilitation program. Non-experimental studies have some inherent biases due to the lack of randomization in the selection of the patients. However, in a publication of the Cochrane Collaboration, Anglemeyer et al. showed that the findings of non-experimental studies could be as valid as those of clinical trials with no significant differences in the outcomes.32
The findings of this study, combined with the failure to adopt the recommendations, suggest the need to strengthen the training of professionals through a greater number of interventions and continuing education in the appropriate use of medications of interest, as well as the need to continuously update the most prevalent pathologies. It is essential that those responsible for the health care of patients with acute coronary syndrome review the clinical practice guidelines and be audited by the scientific directors of the institutions on compliance and adherence to them.
ConclusionsIt can be concluded that the patients with acute coronary syndrome in the 16 cities in this study were, for the most part, treated properly after percutaneous intervention or coronary bypass with the medications recommended by the clinical practice guidelines, particularly with ASA, clopidogrel, metoprolol, losartan, atorvastatin, and enalapril. Limited success was achieved in the adjustment of therapy following the recommendations given to these particular physicians. Nevertheless, there are still patients undergoing an incomplete therapy who continue to be exposed to risks of complications and death, creating unnecessary costs for the health system, which is invited to improve the quality of care and adherence to the guidelines.
FundingThis study received funding from the Universidad Tecnológica de Pereira, Audifarma S.A. Pereira, Colombia.
Conflict of interestNone.
To the Universidad Tecnológica de Pereira, Audifarma S.A.