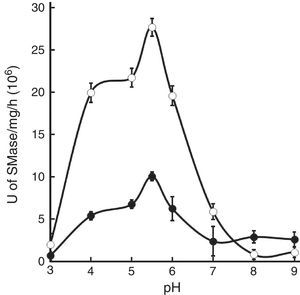
Sphingomyelinases (SMases) catalyze the hydrolysis of sphingomyelin to ceramide and phosphorylcholine. Sphingolipids are recognized as diverse and dynamic regulators of a multitude of cellular processes mediating cell cycle control, differentiation, stress response, cell migration, adhesion, and apoptosis. Bacterial SMases are virulence factors for several species of pathogens. Whole cell extracts of Mycobacterium tuberculosis strains H37Rv and CDC1551 were assayed using [N-methyl-14C]-sphingomyelin as substrate. Acidic Zn2+-dependent SMase activity was identified in both strains. Peak SMase activity was observed at pH 5.5. Interestingly, overall SMase activity levels from CDC1551 extracts are approximately 1/3 of those of H37Rv. The presence of exogenous SMase produced by M. tuberculosis during infection may interfere with the normal host inflammatory response thus allowing the establishment of infection and disease development. This Type C activity is different from previously identified M. tuberculosis SMases. Defining the biochemical characteristics of M. tuberculosis SMases helps to elucidate the roles that these enzymes play during infection and disease.
Las esfingomielinasas (SMasas) catalizan la hidrólisis de esfingomielina a ceramida y fosforilcolina. Los esfingolípidos son reconocidos como reguladores diversos y dinámicos de una multitud de procesos celulares que median en el control del ciclo celular, la diferenciación, la respuesta al estrés, la migración celular, la adhesión y la apoptosis. Las esfingomielinasas bacterianas son factores de virulencia reconocidos en varias especies de patógenos. En este trabajo se analizaron los extractos de células enteras de las cepas de Mycobacterium tuberculosis H37Rv y CDC1551 utilizando [N-metil-14C]-esfingomielina como sustrato. Se identificó actividad de SMasa-ácida dependiente de zinc en ambas cepas. La actividad máxima se observó a pH 5.5. Curiosamente, los niveles de actividad de SMasa generados a partir de extractos de la cepa CDC1551 son aproximadamente un tercio de los de la cepa H37Rv. La presencia de una SMasa exógena producida por M. tuberculosis durante la infección puede interferir con la respuesta inflamatoria del huésped, permitiendo así el establecimiento de la infección y el desarrollo de la enfermedad. Esta actividad tipo C es distinta de las actividades previamente reportadas para M. tuberculosis. Definir las características bioquímicas de las esfingomielinasas de M. tuberculosis ayudará a dilucidar el papel que desempeñan estas enzimas durante la infección y la enfermedad.
Sphingomyelinases (EC 3.1.4.12) (SMases) are enzymes that catalyze the hydrolysis of sphingomyelin to ceramide and phosphorylcholine5. Sphingolipids are ubiquitous molecules of eukaryotic cell membranes and constitute a significant portion of membrane lipids8. Sphingolipids have been shown to be involved in a variety of physiological functions including regulation of cell growth, differentiation, cell migration, adhesion, inflammation and apoptosis2,22,24.
SMases are classified into sub-type C and D based on the site of sphingomyelin hydrolysis and on the resulting products. Products from SMase C are ceramide and phosphorylcholine while SMase D degrades sphingomyelin to phosphoryl ceramide and choline11,19. Furthermore, SMases are classified by their optimum pH activity and localization into (a) acid SMase (A-SMase), (b) secretory Zn2+-dependent SMase (A-sSMase), (c) neutral Mg2+-dependent SMase (N-SMase), (d) neutral Mg2+-independent SMase (N-iSMase), (e) alkaline SMase, and (f) bacterial SMase-phospholipase C5. Several species of bacteria, viruses, and even parasites infect host cells by exploiting either the A-SMase or the N-SMase-ceramide system, or both3,6,14. Alternatively, it has been shown that cellular stress produces plasma secretion of A-SMases resulting in the formation of membrane-embedded, ceramide-enriched lipid rafts and the reorganization of receptor complexes leading to signaling events and regulation of a cellular phenotype that protect mice in a sepsis model9.
Moreover, several pathogenic bacteria produce SMases with direct activity on target host cells. These virulence factors include Clostridium perfringens alpha-toxin, possessing phospholipase C-SMase and biological activities causing hemolysis, dermonecrosis and potentially death of the host20. Inhibition of Bacillus cereus SMase prevented mortality in mice15. Phospholipase C/SMase from the opportunistic pathogen Pseudomonas aeruginosa has hemolytic activity and is selectively cytotoxic to mammalian endothelial cells23.
Staphylococcus aureus invades non-professional phagocytes, eventually resides in acidified phago-endosomes and subsequently escapes from this compartment due to a synergistic activity of the cytolytic peptide, staphylococcal δ-toxin and the SMase β-toxin4. A similar effect has been reported for the C. perfringens SMase activity25.
We previously reported the identification of alkaline, neutral, and acidic SMase activities from Mycobacterium tuberculosis26. In the present study, we report a novel acidic mycobacterial Zn2+-dependent SMase activity.
Materials and methodsMycobacterial whole cell extractsM. tuberculosis strains H37Rv and CDC1551 were grown in Middlebroook 7H9 medium supplemented with OADC for 10 days at 37°C. Cultures were harvested by centrifugation at 3000×g for 15min. An approximate mass of 800mg (wet weight) in 1ml of PBS plus 0.5% Triton X100 was gamma-irradiated for 48h in a Gammacell 3000 equipment (Elan, Nordion International Inc, Kanata, Ontario, Canada). The gamma-irradiated bacteria were washed twice with PBS and resuspended in 1ml of sodium acetate-buffer solution 100mM pH 5.5 (ABS) with 0.5% Triton X-100 (Sigma Chemical Co., St. Louis, MO) and Boehringer Manheim complete EDTA-free protease inhibitor cocktail (cat. No. 1836-170). Bacterial suspensions were disrupted with the Fast Prep System (MP Biomedicals, Santa Ana, CA) by applying 5 cycles of 1min at setting 6, with 2min resting on ice in between runs. The whole cell extracts were centrifuged twice at 16000×g for 8min at room temperature, to remove intact bacterial cells. The preparations were divided into 200μl aliquots and stored at −70°C until used. Protein quantification was done by the method proposed by Lowry et al.13.
Sphingomyelinase activityThe measurement of SMase was based on the method described by Vargas-Villarreal et al.26, with minor modifications. The mycobacterial whole cell extracts were the sources of enzymes for all the described experiments. In brief, 2.5μCi of [N-methyl-14C]-sphingomyelin (47mCi/mmol; PerkinElmer Life and Analytical Science, Boston, MA) was mixed with 200mM sodium acetate (pH 5.5), 2mM Zn2+, 4% Triton X-100 and 0.3mM sphingomyelin. The mixtures were sonicated in an Ultratip Labsonic System (Lab-Line Instrument Inc., Melrose Park IL), applying one pulse of 40W/min. The assays were performed by mixing 10μl of the substrate preparation with 10μl of whole cell extract suspension of M. tuberculosis strain H37Rv or CDC-1551 containing 0–2μg of total protein. The mixture was vortexed for 10sec and incubated at 37°C for 60min. The reaction was stopped by adding 25μl of 1mg/ml sphingomyelin, 1mg/ml phosphorylcholine and 1mg/ml choline (Sigma Chemical Co., St. Louis, MO) in 5% trichloroacetic acid in n-butanol. Non-digested sphingomyelin was separated from the SMase hydrolysis products (phosphorylcholine and choline) by thin-layer chromatography (TLC). The assay mixtures were applied (45μl) on 10cm×10cm silica-gel plates (0.25-mm thickness, 60-mesh; Merck, Germany). The plates were placed in a TLC tank containing chloroform:methanol:water (65:25:4, v/v) as a mobile phase. The tank was loaded 15min before starting each chromatographic run with the mobile phase-vapors to saturate the internal atmosphere. The spots corresponding to choline and phosphorylcholine (origin), and sphingomyelin (Rf=0.29) were developed by exposing the TLC plates to iodine vapors21. To identify [14C]-sphingomyelin, [14C]-phosphorylcholine and [14C]-choline spots, their respective relative migration coefficients (Rf) were compared with those of their corresponding non-radioactive standards (Sigma Chemical Co., St. Louis, Mo.) obtained in the same run. The two spots corresponding to [14C]-sphingomyelin, and [14C]-phosphorylcholine and [14C]-choline mixed were scraped from the TLC silica gel plates and placed into plastic vials containing 5ml scintillation liquid (BCS, Biodegradable Counting Scintillation; Amersham International Corporation, Buckinghamshire, England). Radioactivity in each vial was determined with a 1600 Tri-Carb liquid scintillation spectrometer (Packard Instrument Company, Inc., Downers Grove, IL). The device was adjusted to work with unquenched samples at 96% efficiency. One unit of SMase activity was defined as 1pmole of [14C]-sphingomyelin hydrolyzed (equivalent to picomoles of [14C]-phosphorylcholine plus [14C]-choline released) in 1h of incubation. The specific activity was given as the number of units of SMase activity per mg of total mycobacterial proteins. The classification of the SMase type was determined as described previously26.
Effect of incubation time, dose, pH and cations on sphingomyelinase activityTime: The rate of hydrolysis of sphingomyelin was determined at various time points over 60min by mixing 10μl of whole mycobacterial extracts (containing 2μg of total proteins) with 2.5μCi [N-methyl-14C]-sphingomyelin (final concentration 1mg/ml) dissolved in 20μl of 200mM sodium acetate buffer (pH 5.5), 1mM Zn2+, and 0.2% Triton X-100.
Dose: Variable amounts of protein from whole cell extracts from strains H37Rv and CDC1551 were assayed by mixing 10μl of whole mycobacterial extracts (containing various concentrations of protein between 0 and 2μg) with 2.5μCi [N-methyl-14C]-sphingomyelin (final concentration 1mg/ml) dissolved in 20μl of 200mM sodium acetate buffer (pH 5.5), 1mM Zn2+, and 0.2% Triton X-100 at 37°C, for 1h at pH 5.5.
pH: pH values were adjusted with appropriate proportions of sodium acetate (pH 3–6) or Trizma-Base (pH 7–10) buffers to mycobacterial extracts containing 2μg of total protein and incubated for 1h at 37°C prior to running the standard assay as described above.
Cations: Each metal cation was assayed separately. Assay mixtures with 1 or 10mM MgCl2, CaCl2, ZnSO4, HgCl2, MnCl2, or CoCl2, or with 1 or 10mM of chelating agent EDTA (Sigma Chemical Co., St. Louis, MO) were incubated for 1h at 37°C to run the standard assay as described above.
StatisticsAll the experiments were performed by triplicate (n=9). Slopes of the effect of incubation time and dose of mycobacterial extracts were calculated by linear regression. Statistical significance was determined using ANOVA.
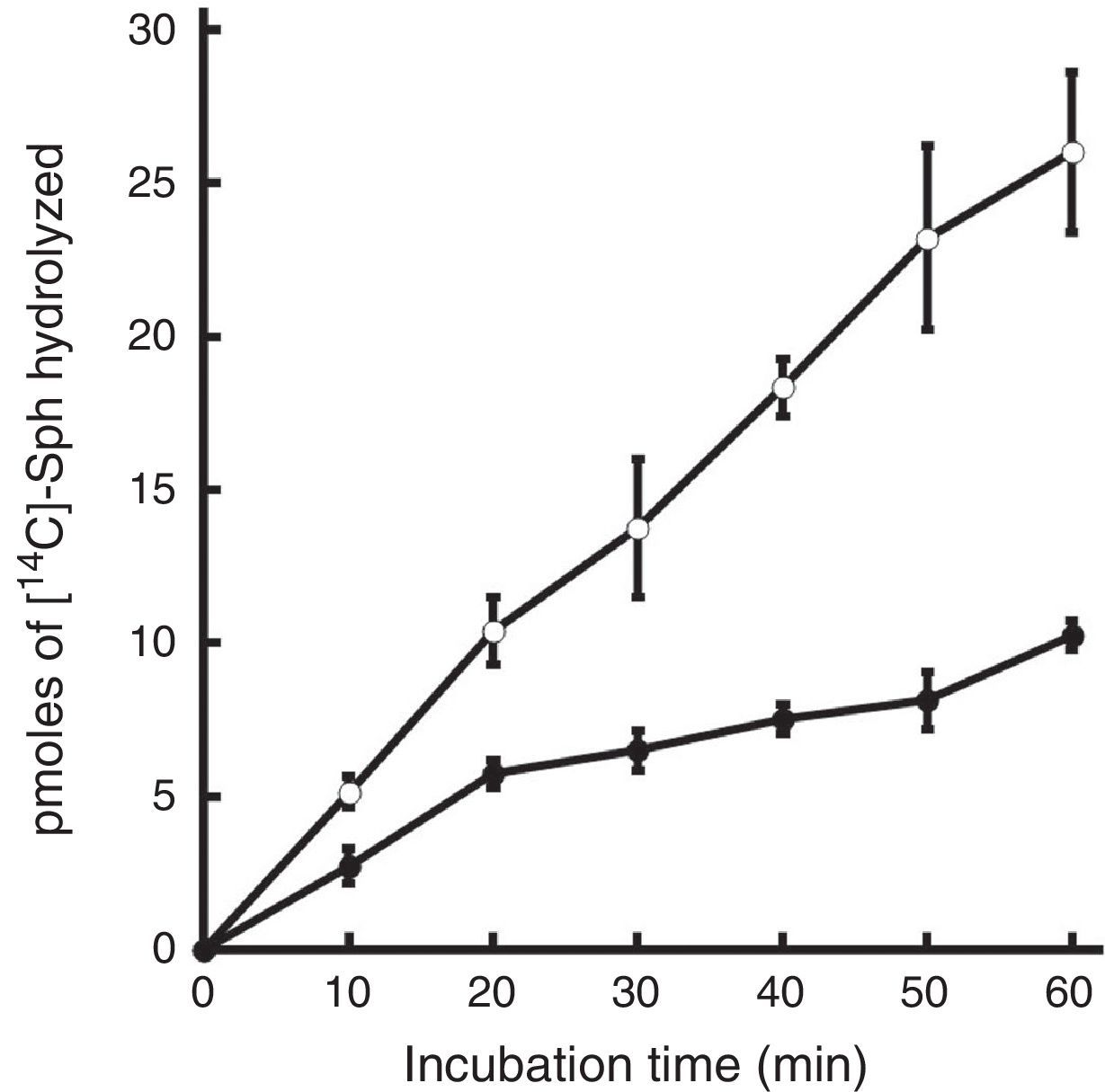
ResultsDependence of mycobacterial sphingomyelinase on incubation time and dose of extractsThe SMase activity of both H37Rv and CDC1551 strains increased linearly between 0 and 60min of incubation (r2=0.99). After 60min of incubation 37.89pmol and 16.1pmol of [14C]-phosphorylcholine were recovered from chromatography plates of H37Rv and CDC1551, respectively (Fig. 1).
Time-course of mycobacterial SMase activity. Mycobacterial cell fractions (2μg of total protein) were tested for SMase activity at incubation time points between 0 and 60min. Symbols represent the mean±SE of [14C]-phosphorylcholine released by M. tuberculosis whole cell extracts of strains H37Rv (○) or CDC1551 (●) in three independent experiments performed in triplicate.
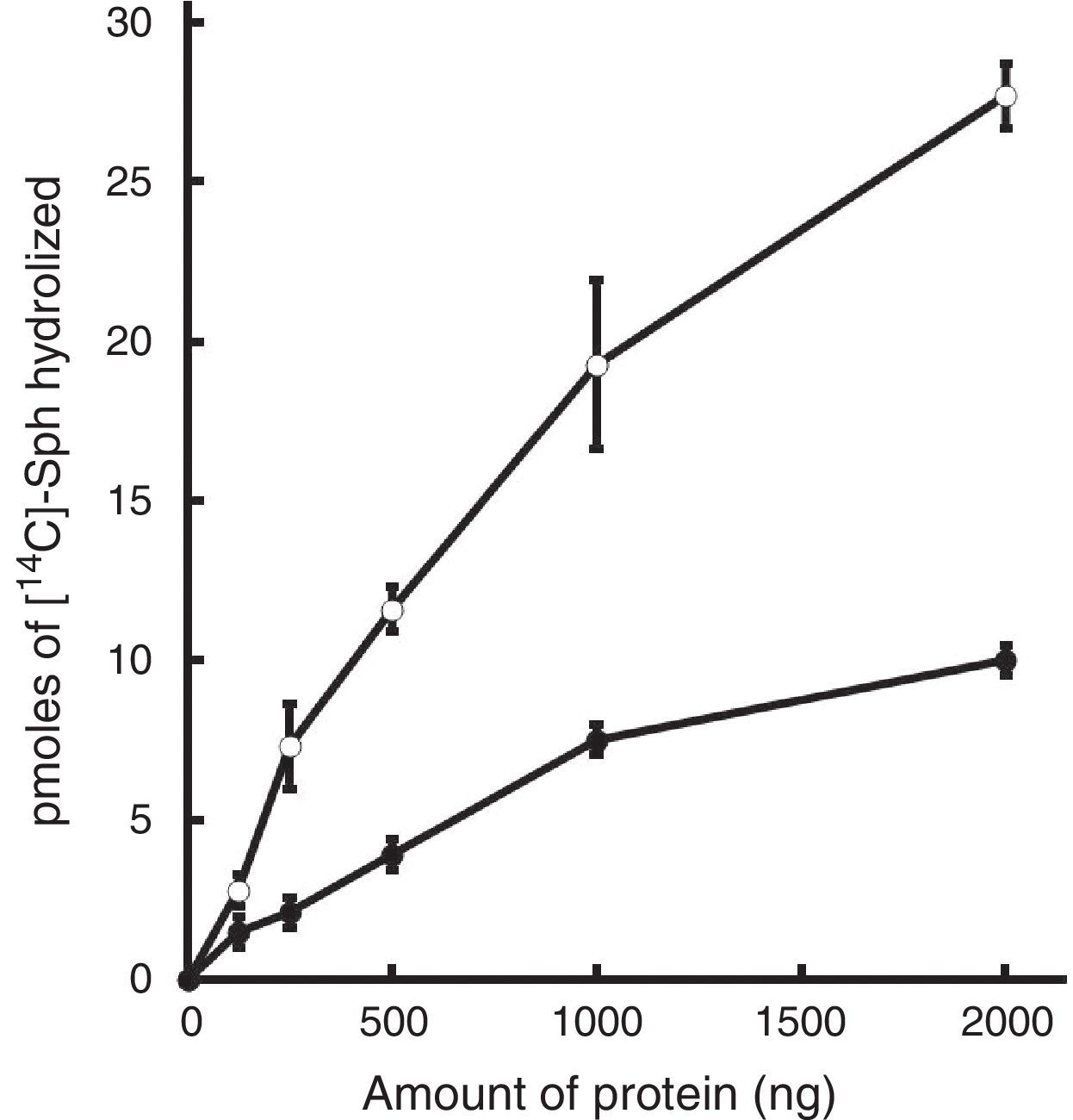
The amount of [14C]-phosphorylcholine released from [14C]-sphingomyelin also increased linearly as a function of extract dose of both strains (r2=0.99). After 60min incubation with 2μg of total protein extract 37.94pmol and 17.65pmol of [14C]-phosphorylcholine were recovered from strains H37Rv and CDC1551, respectively (Fig. 2). Those values corresponded to a specific SMase activity of 37.9×106U/mg/h for H37Rv, which was 2.15 times higher (p<0.001) than that of CDC1551 (17.6×106U/mg/h).
Dose-dependence of M. tuberculosis SMase activity. Mycobacterial cell fractions containing 0–2μg of protein per assay were tested. Symbols represent the mean±SE of [14C]-phosphorylcholine released [in pmol] by M. tuberculosis whole cell extracts of strains H37Rv (○) or CDC1551 (●). All assays were performed at pH 5.5 with 1h incubation.
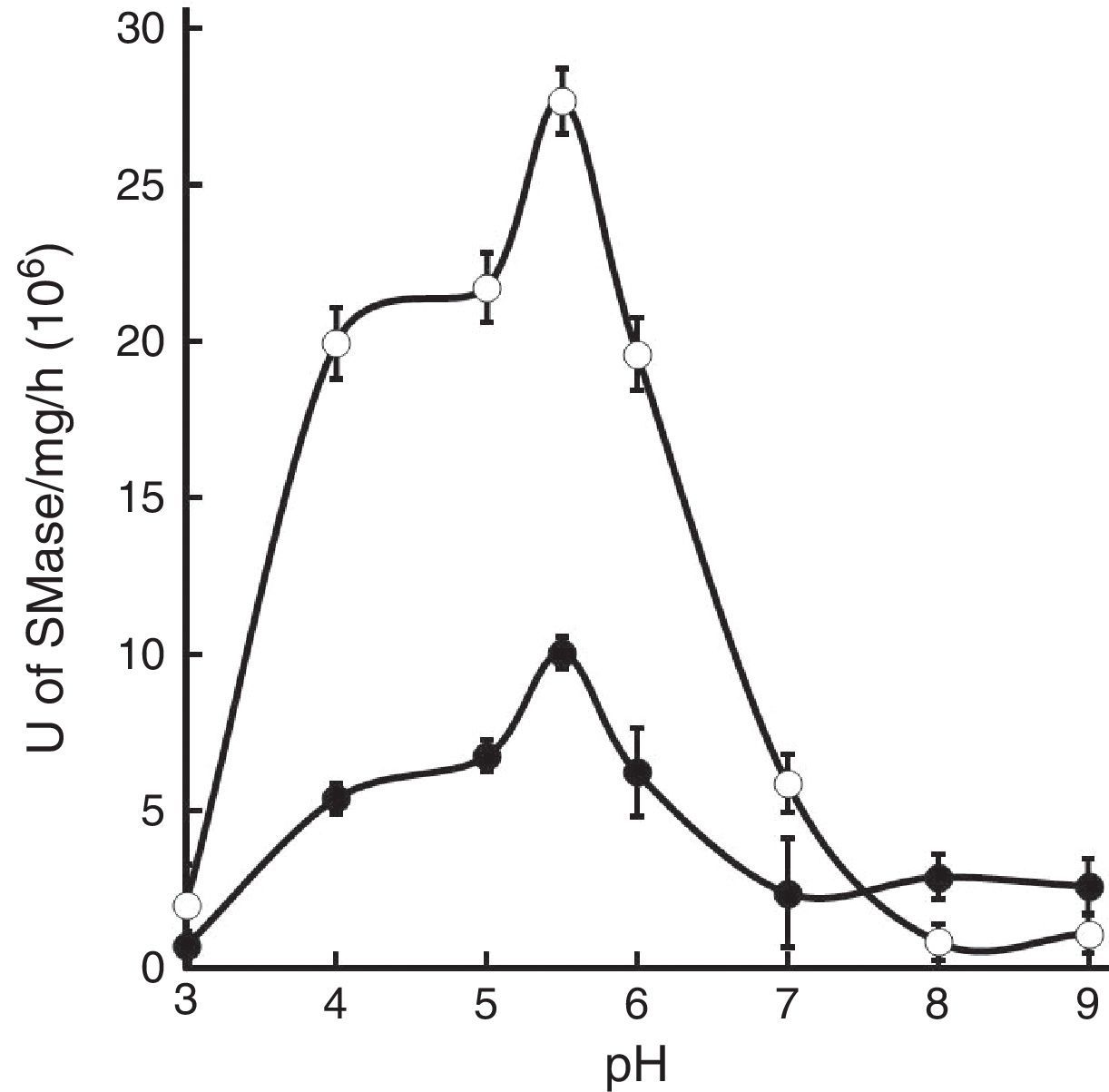
Assayed extracts from both strains produced one primary peak of SMase activity at pH 5.5, which corresponds to the maximum specific activity. The peak from strain H37Rv extracts was almost three times higher than that of strain CDC1551 (Fig. 3).
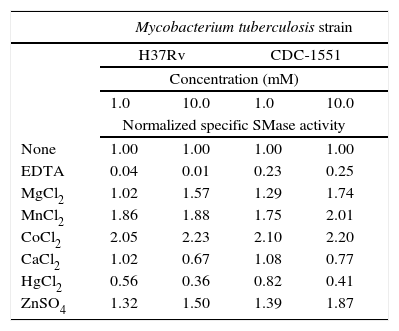
Effect of divalent cationsThe SMase activity detected in whole cell extracts with no extra reagents added was defined as the basal activity equivalent to 100%. Subsequently, the activity was stimulated at different rates by 1 and 10mM concentration of cations Co2+, Mn2+, Zn2+, and Mg2+ or inhibited by Hg2+ and Ca2+. As expected, chelating agent EDTA resulted in the greatest inhibition: 96–99% for strain H37Rv and 75–77% for CDC1551. Results are shown in Table 1.
Normalized sphingomyelinase (SMase) activity modulated by cations or EDTA addition
| Mycobacterium tuberculosis strain | ||||
|---|---|---|---|---|
| H37Rv | CDC-1551 | |||
| Concentration (mM) | ||||
| 1.0 | 10.0 | 1.0 | 10.0 | |
| Normalized specific SMase activity | ||||
| None | 1.00 | 1.00 | 1.00 | 1.00 |
| EDTA | 0.04 | 0.01 | 0.23 | 0.25 |
| MgCl2 | 1.02 | 1.57 | 1.29 | 1.74 |
| MnCl2 | 1.86 | 1.88 | 1.75 | 2.01 |
| CoCl2 | 2.05 | 2.23 | 2.10 | 2.20 |
| CaCl2 | 1.02 | 0.67 | 1.08 | 0.77 |
| HgCl2 | 0.56 | 0.36 | 0.82 | 0.41 |
| ZnSO4 | 1.32 | 1.50 | 1.39 | 1.87 |
Sphingomyelinases (SMases) are enzymes produced by mammalian cells; their main activity is considered to occur during digestive processes. However, these enzymes and the products derived from the hydrolysis of the substrate sphingomyelin are involved in many other cell regulating functions such as cell growth, differentiation, cell migration, adhesion, inflammation and apoptosis2,22,24. A more recent interest in the enzymes has been in their protective roles against pathogens by inhibiting invasion, regulating cytokine responses and inducing apoptosis in infected host cells7.
Pathogens also produce SMases and they may act as virulence factors as demonstrated with C. perfringens20, B. cereus15, P. aeruginosa23, Listeria spp.17 and S. aureus4. Previously, we reported the identification of acidic, alkaline and neutral SMases activities in M. tuberculosis. In that report, neutral SMase had the highest activity and was stimulated by Mg2+, Mn2+ and Co2+ while acidic and alkaline SMase activities remained uncharacterized26. Therefore, in this study, M. tuberculosis acidic SMase activity is further characterized. Maximal SMase activity occurs in a pH range from 5 to 6, with optimal activity at pH at 5.5 (Fig. 3). The assay for cation-induced SMase activity at pH 5.5 resulted in elevated activities by Co2+, Mn2+, Zn2+, and to a lesser extent Mg2+ (Table 1). Zn2+-inducible activity was not previously reported for M. tuberculosis. Thus, it can be classified as Zn2+-dependent acidic SMase activity. By comparing its biochemical characteristics with other SMases, it may correspond to a secretory acidic SMase. In mammalian hosts, this type of SMase regulates immune response10. In this instance, the presence of exogenous SMase produced by M. tuberculosis during infection may interfere with the normal host inflammatory response thus allowing for the establishment of infection and the development of disease. Recently, Roca and Ramakrishnan18 observed that the chemical blockade of host acid SMase activity prevents TNF-induced macrophage programmed necroptosis and mycobacterial release from the cell. Thus, M. tuberculosis acidic SMase may aid in macrophage escape through the induction of necroptosis by its enzymatic product ceramide.
Presently, M. tuberculosis genome annotation in Tuberculist predicts that the protein products from genes plcA (Rv2351c), plcB (Rv2350c), plcC (Rv2349c), and plcD (Rv1755c, reported as a fragment) have phospholipase and SMase activities and they are noted as virulence factors implicated in the pathogenesis of M. tuberculosis at the level of intracellular survival by the alteration of cell signaling events or by direct cytotoxicity12,16. Several bacterial phospholipases possess both phospholipase and SMase activities; an example is the alpha-toxin from the pathogen C. perfringens20. The activity we have identified in this study may correspond to M. tuberculosis phospholipase C expressed from gene plcC (Rv2349c) because the proteins from the plcA and plcB genes are membrane-associated phospholipases C. Furthermore, another report where M. smegmatis was used as an expression system of four M. tuberculosis plc genes supports this idea; plcC product was found to be the most active of the four recombinant PLCs under acidic conditions1. However, a novel M. tuberculosis enzyme with SMase activity cannot be ruled out. More research work is needed to make this determination.
Sphingomyelinases (SMases) and their hydrolysis products (ceramide and phosphorylcholine) are regulators of a multitude of cellular processes. Several pathogens produce SMases as virulence factors. In the present study, we report a novel acidic mycobacterial Zn2+-dependent SMase activity. The relevance of this enzyme in tuberculosis pathogenesis remains to be elucidated.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this investigation.
Confidentiality of dataThe authors declare that no patient data appears in this article.
Right to privacy and informed consentThe authors declare that no patient data appears in this article.
Conflict of interestThe authors declare that they have no conflicts of interest.
This study was funded by FIS/IMSS/PROT/G12/1141.




![Time-course of mycobacterial SMase activity. Mycobacterial cell fractions (2μg of total protein) were tested for SMase activity at incubation time points between 0 and 60min. Symbols represent the mean±SE of [14C]-phosphorylcholine released by M. tuberculosis whole cell extracts of strains H37Rv (○) or CDC1551 (●) in three independent experiments performed in triplicate. Time-course of mycobacterial SMase activity. Mycobacterial cell fractions (2μg of total protein) were tested for SMase activity at incubation time points between 0 and 60min. Symbols represent the mean±SE of [14C]-phosphorylcholine released by M. tuberculosis whole cell extracts of strains H37Rv (○) or CDC1551 (●) in three independent experiments performed in triplicate.](https://static.elsevier.es/multimedia/03257541/0000004800000001/v1_201603250025/S0325754116000031/v1_201603250025/en/main.assets/thumbnail/gr1.jpeg?xkr=ue/ImdikoIMrsJoerZ+w96p5LBcBpyJTqfwgorxm+Ow=)
![Dose-dependence of M. tuberculosis SMase activity. Mycobacterial cell fractions containing 0–2μg of protein per assay were tested. Symbols represent the mean±SE of [14C]-phosphorylcholine released [in pmol] by M. tuberculosis whole cell extracts of strains H37Rv (○) or CDC1551 (●). All assays were performed at pH 5.5 with 1h incubation. Dose-dependence of M. tuberculosis SMase activity. Mycobacterial cell fractions containing 0–2μg of protein per assay were tested. Symbols represent the mean±SE of [14C]-phosphorylcholine released [in pmol] by M. tuberculosis whole cell extracts of strains H37Rv (○) or CDC1551 (●). All assays were performed at pH 5.5 with 1h incubation.](https://static.elsevier.es/multimedia/03257541/0000004800000001/v1_201603250025/S0325754116000031/v1_201603250025/en/main.assets/thumbnail/gr2.jpeg?xkr=ue/ImdikoIMrsJoerZ+w96p5LBcBpyJTqfwgorxm+Ow=)