Intrahepatic cholangiocarcinoma (iCCA) is an uncommon neoplasm but increasing in incidence, with dismal prognosis despite surgical treatment due to lack of tools for early diagnosis.1,2 Recent publications have shown its complex molecular landscape.1,2 Despite activating K-Ras mutations tend to be most frequent in iCCA, with usual FGFR family alterations as well as IDH1/2 mutations, extrahepatic cholangiocarcinoma tend to have Her2 and PIK3CA mutations, while Her2 and TP53 mutations are more frequently found among gallbladder cholangiocarcinoma.1,2 IDH1/2 mutations have also been correlated with clear cell changes and poorly differentiated histology.2,3 Nevertheless, clear cell variant iCCA is extremely rare, with no specific molecular profile associated. We present a case of a clear cell papillary cholangiocarcinoma in which we performed an extensive molecular characterization.
A 57 year old woman was referred to our institution due to incidental abnormal liver function tests (high alkaline phosphatase and GGT). Image studies revealed an intrahepatic mass in V–VI segments, adjacent to the gallbladder (see Fig. 1). A fine needle aspiration cytology specimen revealed a malignant cell proliferation with clear cytoplasm. Image studies discarded other malignancies. With the preoperative diagnosis of cholangiocarcinoma, a right hepatectomy with cholecystectomy was performed. The patient developed pulmonary and liver metastases three months after surgery. She received chemotherapy under the GEMOX schema (Gemcitabine combined with oxaliplatin) but died a year later.
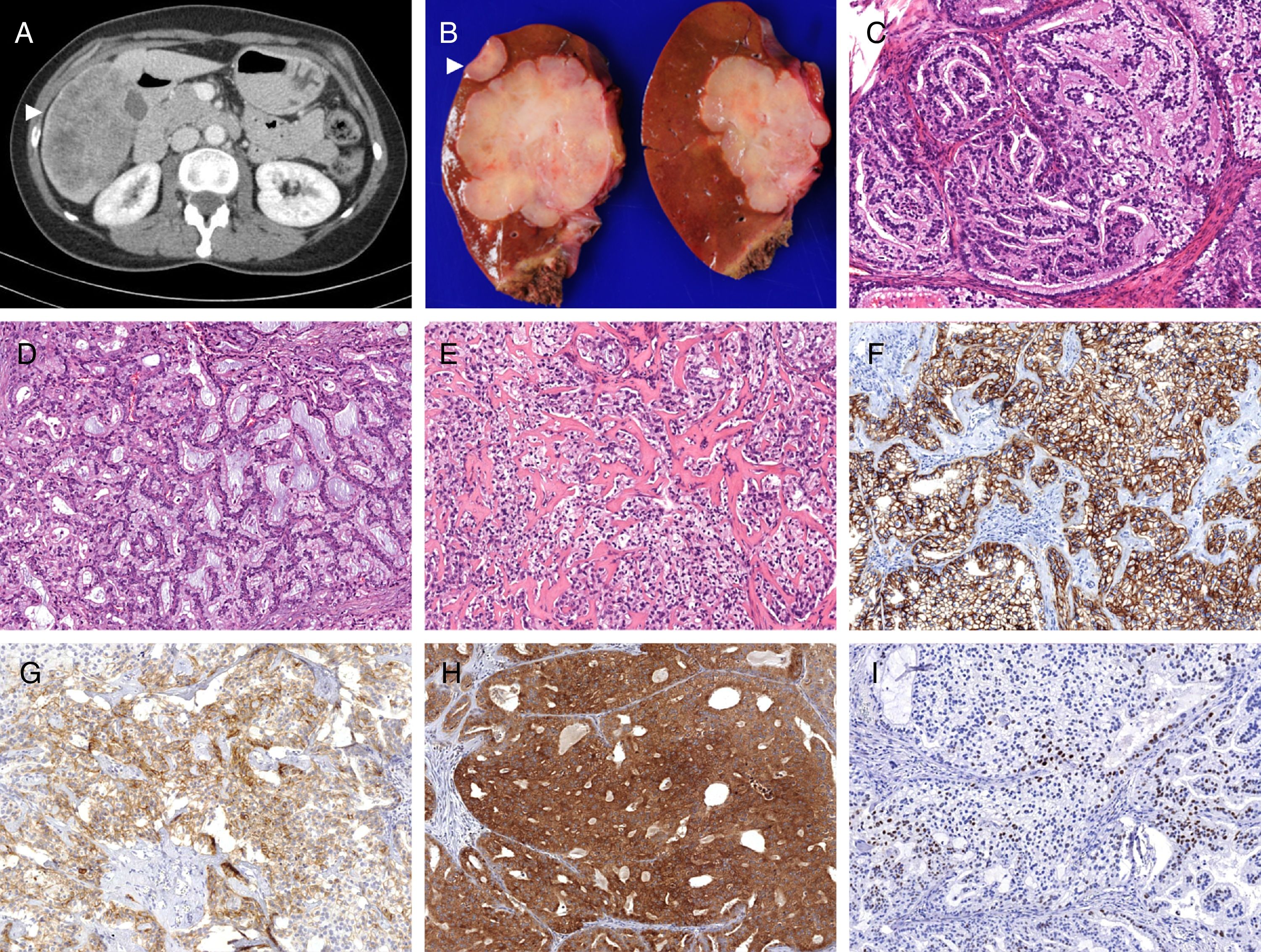
(A) The scanner showed an heterogeneous intrahepatic mass in V–VI segments, adjacent to the gallbladder (white arrowhead). (B) Grossly, the mass was firm white-tan, with adjacent satellite lesions (white arrowhead). The lesion comprised clear cells arranged in tubulopapillar (C) and cribiform (D) structures, and infiltrating areas with stromal sclerosis (E) A diffuse and strong expression of BerEp4 (F) was observed. CD56 had focal positivity (G), and Bap-1 positivity was retained (H). Cyclin-D1 held heterogeneous expression (I). C–E 100×.
Gross examination showed an intrahepatic firm white-tan 11×11×7cm solid mass with three satellite lesions measuring 0.2–2cm. The gallbladder and the extrahepatic ducts showed no lesions. Microscopically the lesion was composed of a proliferation of medium-sized malignant epithelial cells with clear microvacuolated cytoplasms and well-defined borders. They were arranged in tubular, tubulopapillar and cribiform structures. Foci of comedo-type necrosis, perineural and angiolymphatic invasion, and a mitotic index of 7 mitoses/10 HPF were observed, as well as focal perihilar extrahepatic infiltration. Immunohistochemistry demonstrated strong CK7 and BerEp4 positivity, with focal expression of EMA, Cyclin D1, Bap-1 and CD56 (Fig. 1), and microsatellite stability. TTF-1, CEA, CDX2, neuroendocrine and renal cell carcinoma markers were negative. The proliferative index (MIB-1) was above 50%. The Oncomine 22 Solid Tumour DNA (Termo Fisher Scientific) next generation targeted sequencing (NGS) panel was performed. It encompasses mutation hot-spot sites of 22 genes including BRAF, EGFR, ERBB2, FGFR1-3, KRAS, and NRAS, among others. IDH 1/2 were assessed using hot spot exon PCR amplification and Sanger sequencing. No mutations were found in any of the studies.
Intrahepatic clear cell cholangiocarcinomas are extremely rare neoplasms that may require extensive immunohistochemical studies in order to discard clear cell neoplasms from different origins (i.e. renal, ovary or thyroid), as well as clear cell hepatocarcinoma.4 Among them, CD56 immunostain is useful, since it is frequently expressed in clear cell cholangiocarcinoma, and scarcely in clear renal cell carcinoma and in lung PEComa.4 In addition, usual ductal pancreatic carcinomas tend to be CD56 negative whereas gallbladder neoplasms may express it.5
IDH1 and 2 mutations seem to be of importance in the tumorigenesis of intrahepatic cholangiocarcinoma,1 with controversial results regarding IDH mutations to predict survival.1,2 Alterations in FGFR, mainly in the form of fusions and translocations, have also been found in intrahepatic cholangiocarcinoma and seem to harbour a better prognosis.1,2 We did not find mutations in the IDH1/2 genes and/or FGFR genes in our case, although we were not able to perform fusion or translocation studies. Solid tumour NGS panels are already available in the clinical context of a growing number of institutions. Yet, the joint analyses of DNA mutations and RNA alterations are relatively novel and may be limited to selected laboratories in a daily setting.
Some reports have suggested that clear cell intrahepatic cholangiocarcinoma may harbour better prognosis than conventional cholangiocarcinoma.4 Our case recurred within three months after surgery and died the following year, being the usual outcome of a locally advanced intrahepatic cholangiorcarcinoma with high risk features.
In conclusion, we present a patient of an intrahepatic papillary clear cell cholangiocarcinoma with targeted sequencing studies that revealed the absence of the usual mutations observed in this setting. The findings from a single case study may not be representative. Nevertheless, the potential benefit of selecting clinically relevant targeted therapies may worth the molecular profiling of rare variants of aggressive tumours such as the intrahepatic cholangiocarcinoma.
Specific author contributionsIban Aldecoa: Wrote the manuscript. Made substantial contributions to its conception and design. Acquisition, analysis and interpretation of stains and data. Wrote the manuscript, interpreted stains and data.
Carla Montironi: Participated in writing the manuscript, interpreted stains and data.
Guillermo Madrigal Vargas: Participated in writing the manuscript, interpreted stains and data.
Rosa Miquel: Participated in writing the manuscript, interpreted stains and data.
Miriam Cuatrecasas: Participated in interpretation of stains and data, drafting and writing the manuscript. Critical revision for important intellectual content and final approval of the version to be submitted.
Conflict of interestsWe are indebted to Elena Martinez and Pedro Jares for support in the molecular studies. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The authors declare no conflict of interests.