Chorionicity is the main determinant of the perinatal outcome in twin pregnancies: perinatal mortality and morbidity are significantly higher in monochorionic versus dichorionic twins. This is mainly due to complications associated specifically with monochorionicity, such as twin to twin transfusion syndrome (TTTS), selective fetal growth restriction (FGR) and twin reverse arterial perfusion syndrome (TRAP), consequences of the presence of inter-twin vascular anastomoses in the common placenta. For this reason the diagnosis of chorionicity in twins is of clinical importance in order to plan an increased surveillance in monochorionic gestations and to recognize the appearance of complications in their early stages.
Different sonographic signs may be used to evaluate chorionicity: number of placental masses, sex of the fetuses, characteristics of the intertwine membrane. The last one is surely the most useful and valuable tool: the take-off of the membrane from the placental surface shows the typical “lambda” appearance in dichorionic pregnancy and the typical “T” appearance in the monochorionic ones. In this article the sonographic features that help in the accurate depiction of chorionicity are reviewed.
La corionicidad es el principal determinante del desenlace perinatal en los embarazos gemelares: la morbimortalidad perinatal es significativamente superior en los gemelos monocoriónicos que en los bicoriónicos. Esto se debe principalmente a las complicaciones asociadas con la corionicidad, a saber, el síndrome de transfusión fetal-fetal (STFF), la restricción del crecimiento fetal selectivo (CIR) y la perfusión arterial reversa (secuencia TRAP), que son debidas a anastomosis vasculares intergemelares en la placenta común. Por esta razón, el diagnóstico de corionicidad en gemelos es de importancia clínica, ya que así se podrá planificar una mayor vigilancia en gestaciones monocoriónicas y detectar la aparición de complicaciones en las primeras etapas.
Distintos signos ecográficos sirven para evaluar la corionicidad: número de masas placentarias, sexo de los fetos, características de la membrana intergemelar. Esto último es, sin duda, la herramienta más útil y valiosa: la separación de la membrana de la superficie placentaria muestra una típica forma «lambda» en el embarazo bicoriónico y un típico signo en «T» en el monocoriónico. En este artículo se revisan las características ecográficas que ayudan a la descripción exacta de la corionicidad.
Twin pregnancies account for 1–2.5% of all pregnancies1; the prevalence of twins showed an increase of approximately 80% since the beginning of the 1970s, mainly due to the increase of the maternal age at conception over this period and to the widespread diffusion of the assisted reproduction technology.
Twin pregnancies are at significant risk for adverse outcome with perinatal mortality and morbidity rate three to six times that of singletons. The main determinant of fetal outcome in twin pregnancy is placental chorionicity: monochorionic twins have a three to five higher risk of perinatal loss and handicap in comparison to dichorionic.2,3 This is mainly due to complications specifically associated with monochorionicity such as twin to twin transfusion syndrome (TTTS), twin reversed arterial perfusion syndrome (TRAP), selective intrauterine growth restriction (IUGR), which are consequences of the vascular anastomoses between the two twins sharing the single placenta.4
The determination of chorionicity in a twin pregnancy is important to plan an intensive monitoring of the monochorionic pregnancies with the aim of improving their outcome, to perform accurately prenatal invasive procedures and to early diagnose and manage complications such as TTTS and selective IUGR.
Zygosity and chorionicityZygosity refers to the type of conception: monozygotic twins result from the mitotic division of a zygote originating from a single ovum fertilized by one sperm; dizygotic twins are the result of a double conception originating from two ova fertilized by two sperms. Monozygotic twins account for 30% of all twin pregnancies. The origin of monozygotic twinning is still unclear and different hypotheses have been proposed, the most exiting one suggesting that monozygotic and dizygotic twinning events arise from the same embryogenic mechanism.5–7
Chorionicity refers to the type of placentation and does not reflect zygosity. Whereas dizygotic twins are always dichorionic, monozygotic twins may be monochorionic or dichorionic depending on when the zygote divides. This is the hypothesis of the Corner's theory8 which has never been demonstrated in humans,6 but is useful for understanding the development of different types of chorionicity in monozygotic twins. According to this theory, if the split of the zygote occurs during the first three days following fertilization, dichorionic diamniotic twins develop: this occurs in 29% of the cases. When the split occurs 4–7 days after fertilization a monochorionic diamniotic twin pregnancy develops: this occurs in 70% of the cases. In 1% of the cases the split occurs later (between 8 and 12 days) resulting in monochorionic monoamniotic twins. When a partial splitting of the zygote occurs between 13 and 16 days following fertilization conjoined twins develop; this is an extremely rare condition occurring in up to 200,000 births.
Ultrasound diagnosis of chorionicityAlthough dizygotic twins always have a dichorionic placenta, the appearance of the placentas would depend on the sites of the blastocysts implantation. In case of distant implantations in the uterine cavity two separate placentas can be demonstrated; in case of close implantation they will appear fused in a single placental mass, similar to that of the monozygotic monochorionic twins. However, due to the higher risk of complications, monochorionic twins need an increased surveillance with strict follow-up and for this reason they must be differentiated from the dizygotic dichorionic twins with fused placentae as early as possible.
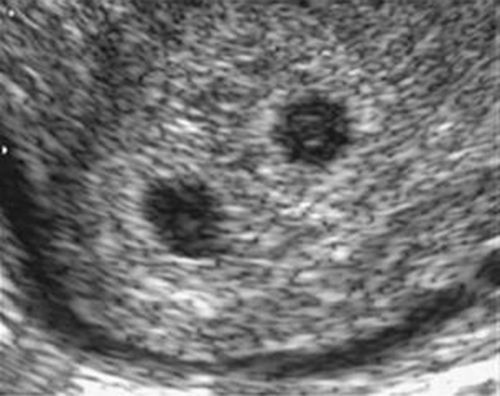
First trimester The ideal time to determine chorionicity is the first trimester. As early as 4–5 weeks postmenstrual weeks, by using transvaginal sonography, it is possible to recognize the chorionic sacs inside the uterine cavity within the tick deciduas. They appear as sonolucent round structures with a brightly echogenic rim. By simply counting the chorionic sacs it is possible to establish whether the pregnancy will be monochorionic, dichorionic, trichorionic and so on (Fig. 1).
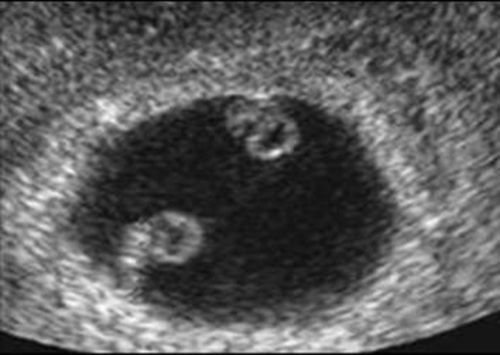
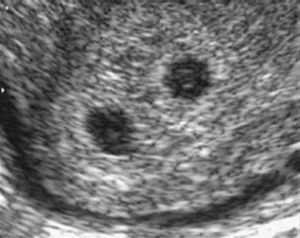
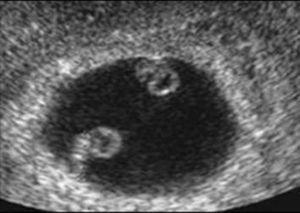
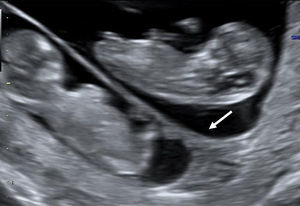
The visualization of a single chorionic sac at 4–5 weeks, however, does not exclude the possibility of a monochorionic twin pregnancy. By the sixth postmenstrual week the yolk sac and the embryo can be visualized inside the chorionic sac and the definitive diagnosis of a single pregnancy or monochorionic twin pregnancy can be done. If a single chorionic sac is visualized containing two yolk sacs and two embryonic poles, then a monochorionic twin pregnancy can be diagnosed (Fig. 2).
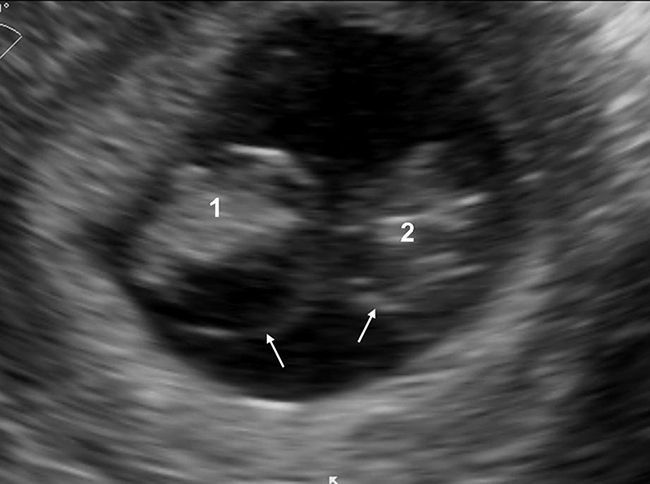
Determination of amnioticity is difficult until 8 weeks of gestation. Before that time the amnion is still too close to the embryo and is identifiable in only 50% of the cases.9 The amnions appear as thin echogenic membranes surrounding the two embryos inside the celomatic cavity (Fig. 3).
Lack of visualization of the amniotic membranes after 8 weeks allows the diagnosis of monochorionic monoamniotic twins. In this case the number of yolk sac seen may be one or two depending on the timing of cell division.10
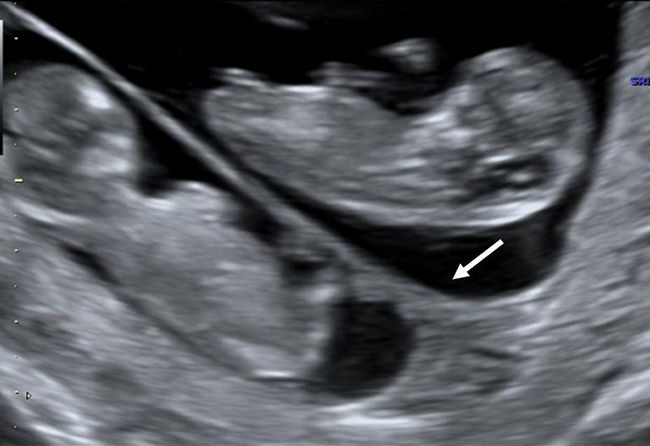
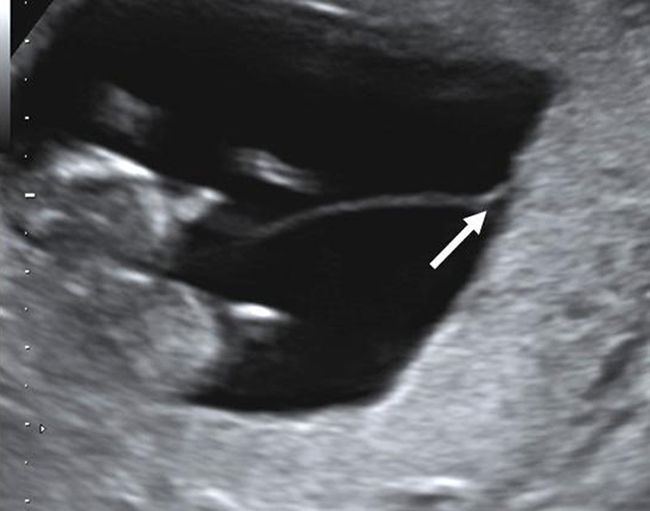
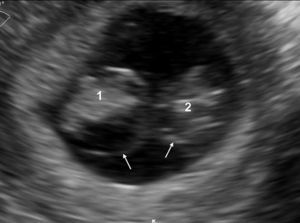
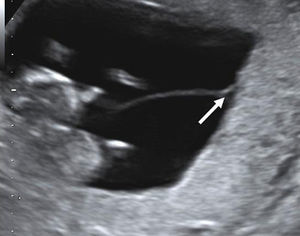
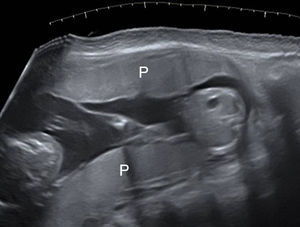
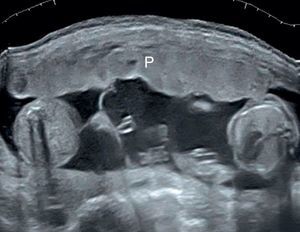
After 10 weeks of gestation chorionicity is determined by the evaluation of the number of the placental masses and, in case of a single placental mass, by the characteristics of the membrane dividing the amniotic sacs. In dichorionic twins with fused placentas, amnios and chorion reflect away from the placental surface, creating a potential space into which villi can grow. Sonographically this space appears as a triangular-shaped extension of placental tissue into the base on the inter-twin membrane. This sign was first described in 1981 as the “lambda” sign11 and is used interchangeably with the “twin peak” sign described in 199212 (Fig. 4). In monochorionic pregnancy there is a single layer of continuous chorion limiting the villous growth; the intertwin membrane takes off perpendicularly to the placental surface producing the sonographic sign known as “T” sign (Fig. 5).
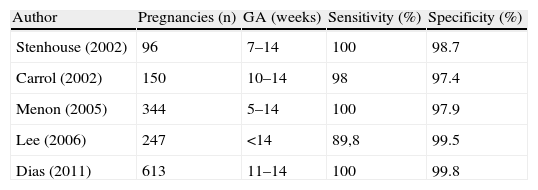
Several studies have confirmed the accuracy of “lambda” and “T” signs in predicting chorionicity in the first trimester, comparing the sonographic diagnosis with the placental hystology or the discordant sex at birth.13–17 The sensitivity reported ranges from 89,8% to 100%; the specificity ranges from 97.4% to 99.8% (Table 1). The gestational age at the time of the sonographic examination varied in the different studies. Stenhouse et al.13 and Menon et al.15report a sensitivity of 100% but they examined the patients at 7–14 and 5–14 weeks respectively, thus including in these good results also the patients examined in the earliest stages of pregnancy (below 10 weeks) when the sonographic diagnosis relies on the number of the chorionic sacs rather then the T/lambda sign. The study by Lee et al.16reported four sex discordant twin pairs with monochorionic placentas. They used this finding to contest the belief that monochorionicity is synonymous with monozygosity. However they did not perform genetic tests to confirm zygosity. Carroll et al.14 compared the accuracy of T/lambda sign and membrane thickness and found that membrane thickness improved sensitivity by 1.3%, although its assessment is time consuming and highly inter- and intraobserver dependent. The largest series is by Dias et al.17: they examined 613 twin pregnancies with transabdominal ultrasound at the time of the routine first trimester screening (11–14 weeks): sensitivity and specificity were 100% and 99.8% respectively. The only error occurred in a pregnancy complicated by a small hematoma running along the site of the membrane insertion.
Sensitivity and specificity of lambda and T signs for determining chorionicity in the first trimester of pregnancy.
| Author | Pregnancies (n) | GA (weeks) | Sensitivity (%) | Specificity (%) |
| Stenhouse (2002) | 96 | 7–14 | 100 | 98.7 |
| Carrol (2002) | 150 | 10–14 | 98 | 97.4 |
| Menon (2005) | 344 | 5–14 | 100 | 97.9 |
| Lee (2006) | 247 | <14 | 89,8 | 99.5 |
| Dias (2011) | 613 | 11–14 | 100 | 99.8 |
Another technique to assess chorionicity in the first trimester is the evaluation of the number of layers of the intertwin membrane, considering that the dividing membrane is made by only two layers of amnios in monochorionic and four layers (two amnios and two chorions) in dichorionic twins. This technique, mainly used in the second and third trimester, has also been applied in the first trimester using transvaginal approach and depicting the amnios and chorion at the membrane to placenta insertion.18
In conclusion the accuracy of ultrasound in the assessment of chorionicity in the first trimester is high and is mainly based on the evaluation of lambda and T-sign.
Second and third trimester. Determination of chorionicity in the second and third trimester is more problematic and the diagnostic accuracy is lower than in the first trimester. The diagnosis relies on the evaluation of fetal gender, number of the placental masses and characteristics of the intertwin membrane.19
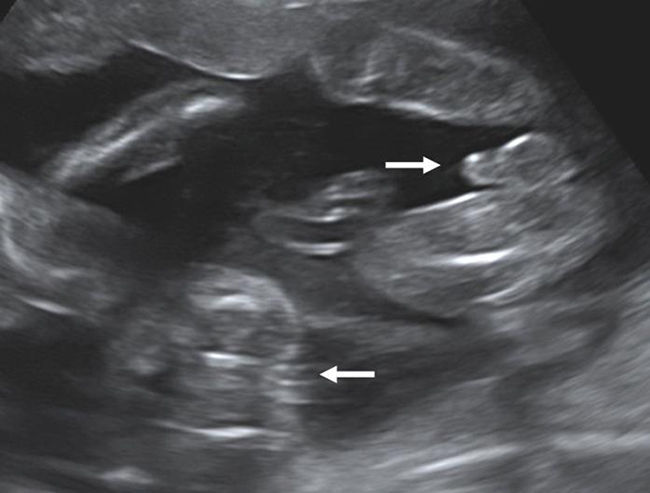
Fetal gender The identification of discordant fetal gender indicates dichorionic twinning (Fig. 6). The positive predictive value of discordant gender (when correctly identified) is 100%. However, considering that around 50% of concordant sex twins are dichorionic, the definition of chorionicity needs further sonographic signs.
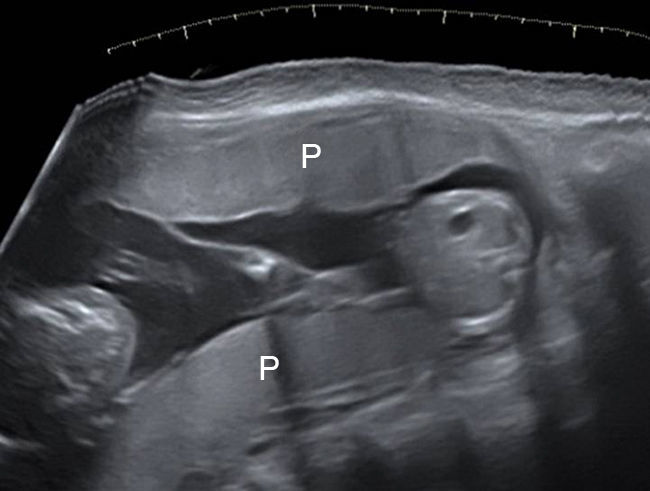
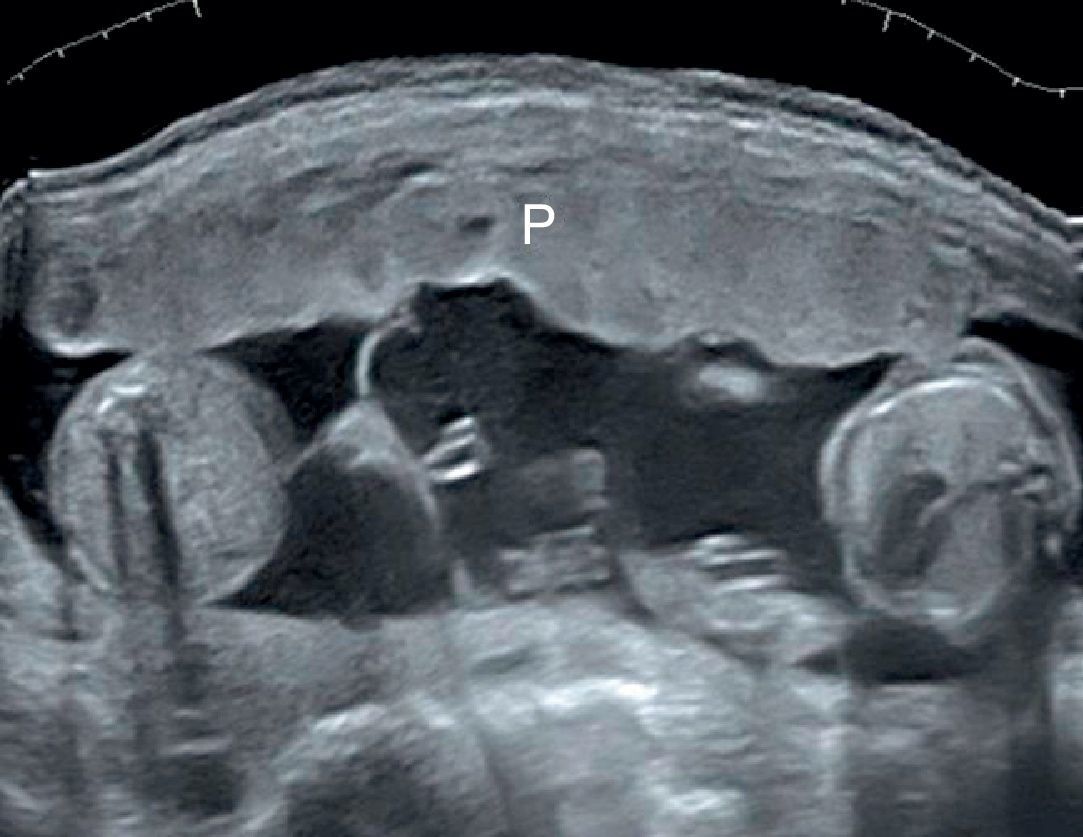
Number of the placental masses The location of the placental masses has a limited value in the assessment of chorionicity. Separate placental sites are an indicator of dichorionic twins (Fig. 7). However widely separated placentas are found in only one-third of all twin gestations. Furthermore two separated placental masses may be seen in the rare cases of a monochorionic placenta which is bilobed or has a succenturiate lobe. For this reason the sensitivity of two placental sites is 32% and the PPV is 97.7%.20 A single placental mass may be indication of both monochorionic and dichorionic twins with fused placentas (Fig. 8).
Given these limitations, identifying chorionicity must rely on other sonographic parameters particularly when a single placental mass is seen.
Take-off of the intertwin membrane The ultrasonic visualization of the intertwin membrane origin (take-off) from the placental surface in the second and third trimester has a limited diagnostic value as compared to the first trimester. The membrane take-off to recognize lambda and T-sign may be obscured by the fetus, particularly in the third trimester and when the placenta is posterior. Furthermore, it must be taken into account that as the pregnancy progresses the lambda sign may disappear as the consequence of the progressive regression of the chorion frondosum to form chorion leave at the base of the intertwin membrane. The lambda has been shown to disappear by the 20th week of gestation in about 7% of dichorionic twins with fused placentas.21 This supports the conclusion that while the presence of the lambda sign indicates dichorionicity, its absence does not always rule it out.22
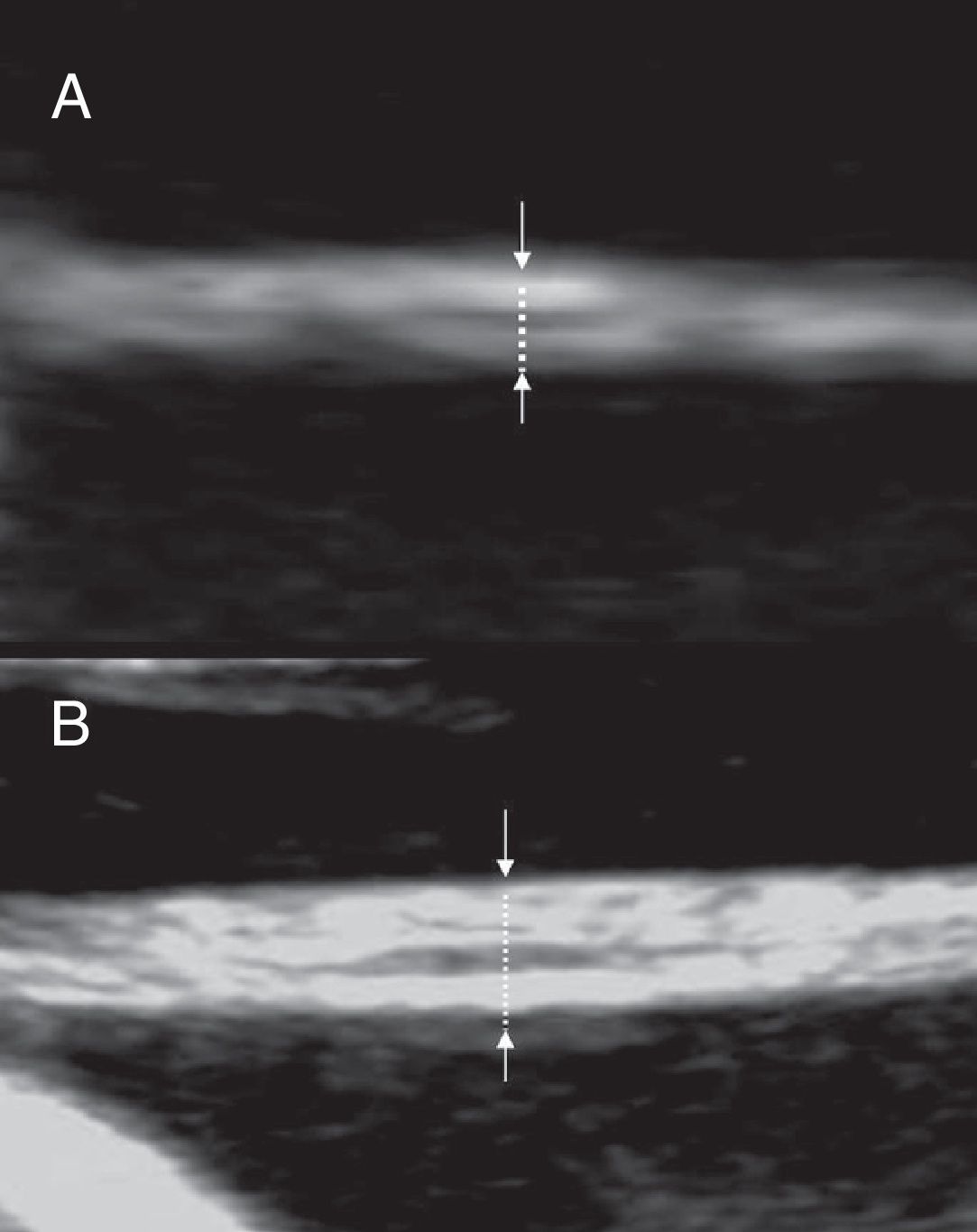
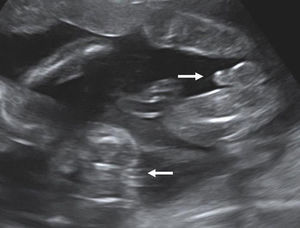
Thickness of the inter-twin membrane The inter-twin membrane thickness is greater in dichorionic twins because it is made of four layers (two chorions and two amnios) as compared to monochorionic twins, whose membrane is made by two layers (two amnios) and consequently is thinner and hairlike.
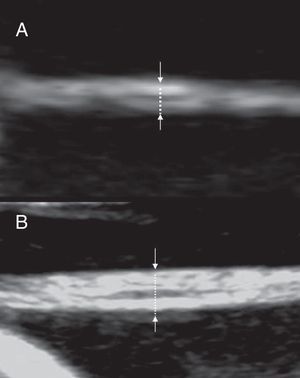
The membrane thickness can be measured with high resolution ultrasound equipments. The membrane should be imaged perpendicularly to the ultrasonic beam after magnification (Fig. 9). The most reliable site for measurement with the most reproducibility is close to the placenta (<3cm).23 The cut-off value firstly suggested to differentiate dichorionic from monochorionic twins is 2mm. Using that value Winn et al.24 report a diagnostic accuracy of 82% for monochorionicity and 95% for dichorionicity. Bracero and Bryne25 report a sensitivity of 75.7%, a specificity of 85.7%, a positive predictive value of 96.6%, and a negative predictive value of 40.0%, for determining dichorionicity. A lower cut-off of 1,5mm has been suggested for measurements made in the first trimester.14The sensitivity of membrane thickness for determining chorionicity declines with increasing gestation with a sensitivity of only 52% for dichorionicity in the third trimester.26
Evaluation of the inter-twin membrane in the second trimester. The membrane is imaged perpendicularly to the ultrasonic beam and magnified. (A) monochorionic twins: two layers may be seen and the thickness is below 2mm (B) dichorionic twins: more than two layers may be seen and the thickness is above 2mm.
In order to improve the reproducibility of the membrane thickness measurement and to be sure that the membrane is perpendicular to the ultrasonic beam, three dimensional multiplanar sonographic evaluation has been suggested with a very slight increase of sensitivity.27
Due to these limitations, to the technical difficulties and to the operator dependency, the measurement of the inter-twin membrane thickness is of limited value in determining chorionicity in the second and third trimesters.
Number of the layers of the inter-twin membrane The inter-twin membrane is made by four layers (amnios-chorion-chorion-amnios) in dichorionic and only two layers (amnios-amnios) in monochorionic twins. Using high resolution equipment and magnified images with the membrane perpendicular to the ultrasound beam it is possible to recognize the layers; counting more than two layers allows to diagnose dichorionicity19–29 (Fig. 9). It is useful to visualize the membrane close to its placental insertion in order to minimize the risk of error due to the folding back of a monochorionic membrane on itself thus appearing to have four layers.
By counting the number of layers in the inter-twin membrane a 100% predictive accuracy for dichorionic and 94,4% for monochorionic have been reported.29 Another study reported a PPV of 100% for dichorionicity.30 The technique is more accurate in the second rather than the third trimester due to the tinning of the membrane with progressing gestational age.31
Counting of the layers in the inter-twin membrane is not commonly used in the clinical practice, because it is time consuming, technically difficult and operator dependent.
Doppler evaluation of the placenta A further technique suggested to recognize monochorionic twins in the third trimester is the detection of arterio-arterial anastomoses using Doppler evaluation of the placenta. Using this technique Fichera et al.32 report a sensitivity of 75% and a specificity of 100% with detection rate increasing with gestational age.
Also this technique, however, is time consuming, operator dependent and requires highly sophisticated equipments and its use in determining chorionicity in the clinical practice is very limited.
ConclusionsThe prenatal determination of chorionicity is the first step for an accurate managing of twin gestation. Knowledge of chorionicity helps in risk assessment, genetic counseling, invasive procedure and management of TTTS and selective IUGR, death of one twin and discordant fetal anomaly. It is best done in the first trimester, when the diagnostic accuracy approaches 100%. The most reliable sonographic signs are the lambda and T-sign and the number of the placental masses evaluated before 14 weeks of gestation.
Despite the widespread diffusion of ultrasound as a basic screening test, there is still limited knowledge about the correct diagnosis of chorionicity. A recent study showed that only 43% of multifetal pregnancies carried an accurate diagnosis of chorionicity before tertiary care center evaluation.33 For this reason if there is uncertainty in the diagnosis of chorionicity the patient should be referred to a specialist.
The importance of a correct diagnosis of chorionicity in twin gestation has been recently stressed by the report of the National Collaborating Center for Women's and Children's Health on Multiple Pregnancy,34 whose recommendations are:
- •
Determine chorionicity at the time of detecting twin pregnancy by ultrasound using the number of placental masses, the lambda and T-sign and membrane thickness.
- •
Assign nomenclature to fetuses (for example: upper and lower, left and right) and document this clearly in the woman's notes.
- •
If a woman with a twin pregnancy presents after 14 weeks determine chorionicity at the earliest opportunity by using all the possible signs (number of placental masses, lambda and T-sign, membrane thickness, discordant sex).
- •
If it is not possible to determine chorionicity by ultrasound at the time of detecting the twin pregnancy, seek a second opinion from a senior ultrasonographer or offer the woman referral to a healthcare professional who is competent in determining chorionicity as soon as possible.
- •
If it is difficult to determine chorionicity even after referral manage the pregnancy as monochorionic until proved otherwise.
- •
Provide regular training so that ultrasonographers can identify the lambda and T-sign accurately and confidently. Training should cover ultrasound scan measurements needed for women who book after 14 weeks and should emphasize that the risks associated with twin pregnancies are determined by chorionicity and not zygosity.
- •
Conduct regular clinical audits to evaluate the accuracy of determining chorionicity.
The authors declare no conflict of interest.