Transcatheter “valve-in-valve” (VIV) implantation into a deteriorated mitral bioprosthesis is an uncommon technique worldwide. Reintervention of a mitral degenerated valve is associated with an elevated surgical risk, especially in elderly patients with multiple comorbidities. This technique provides an alternative to conventional surgery that avoids myocardial dissection, extracorporeal circulation, and myocardial ischemia.1 Most prostheses are implanted via a transapical approach; however, another route to access the degenerated prostheses is through the femoral vein, followed by perforation of the interatrial septum and anterograde valve implantation.2 Herein we present the first experience with this technique performed in our facilities and for our concern the first in Latin America.
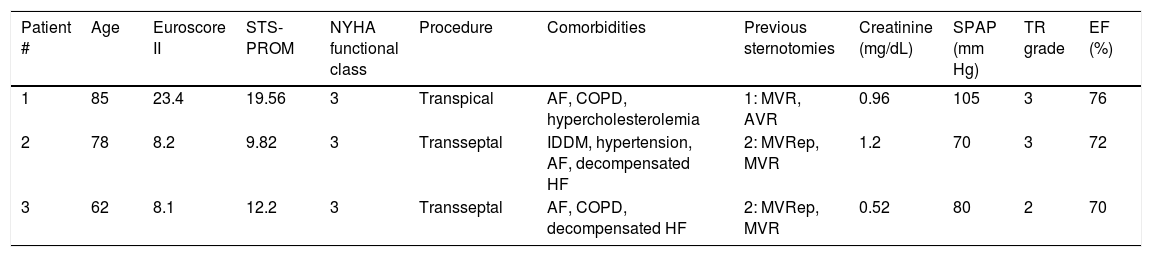
From January to December 2016, 3 patients (age 74±12 years) were admitted in our institution with signs of valve dysfunction long term after bio-prosthetic mitral valve replacement (MVR), 2 patients received transseptal implantation and 1 patient transapical implantation of a balloon-expandable pericardial heart valve into a degenerated bioprosthesis (range 24–29mm) in mitral position at our institution. All patients were considered in high risk for surgical valve replacement (EuroSCORE II 15.755.75±7.6%, STS PROM [Society of Thoracic Surgeons predicted risk of mortality] 14.69±4.8%) (Table 1) after the Heart Team evaluation, therefore were eligible for a VIV procedure via a transseptal or transapical approach.
Baseline clinical parameters.
| Patient # | Age | Euroscore II | STS-PROM | NYHA functional class | Procedure | Comorbidities | Previous sternotomies | Creatinine (mg/dL) | SPAP (mm Hg) | TR grade | EF (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 85 | 23.4 | 19.56 | 3 | Transpical | AF, COPD, hypercholesterolemia | 1: MVR, AVR | 0.96 | 105 | 3 | 76 |
| 2 | 78 | 8.2 | 9.82 | 3 | Transseptal | IDDM, hypertension, AF, decompensated HF | 2: MVRep, MVR | 1.2 | 70 | 3 | 72 |
| 3 | 62 | 8.1 | 12.2 | 3 | Transseptal | AF, COPD, decompensated HF | 2: MVRep, MVR | 0.52 | 80 | 2 | 70 |
AF, atrial fibrillation; AVR, aortic valve replacement; COPD, chronic obstructive pulmonary disease; EF, left ventricular ejection fraction; HF, heart failure; IDDM, insulin dependent diabetes mellitus; MVR, mitral valve replacement; MVRep, mitral valve surgical repair; NYHA, New York Heart Association; SPAP, systolic pulmonary artery pressure; STS PROM, Society of Thoracic Surgeons predicted risk of mortality; TR, tricuspid regurgitation.
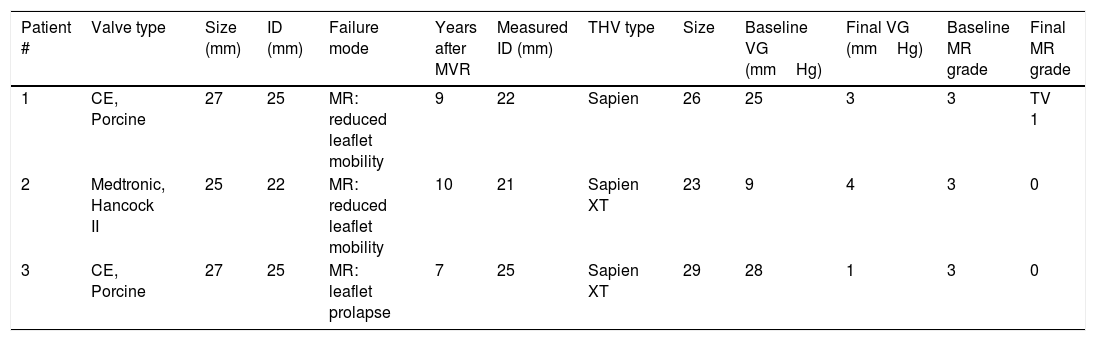
Implantation was successful in all patients, 2 patients received transseptal implantation with an Edwards Sapien XT (Edwards Lifesciences, Irvine, California) valve (#23 and #29) and 1 patient transapical implantation with an Edwards Sapien (Edwards Lifesciences, Irvine, California) #26 valve into a degenerated bioprosthesis (range 24–29mm) in mitral position. Ecocardiography showed a reduction of mean transvalvular gradients from 18.5±9.5mmHg to 3±1mmHg, with no paravalvular regurgitation remaining, the systolic pulmonary artery pressure (SPAP) had a reduction from 92.5±12.5mmHg to 50±10mmHg (Table 2). One patient with the transseptal approach developed a hospital acquired pneumonia and sepsis, then evolves into a septic shock and died 10 days after the procedure. In the remaining patients the New York Heart Association (NYHA) functional class improved from 3.0 to 1.0, over a mean follow-up of 227 (IQR: 90–365) days.
Mitral valve characteristics.
| Patient # | Valve type | Size (mm) | ID (mm) | Failure mode | Years after MVR | Measured ID (mm) | THV type | Size | Baseline VG (mmHg) | Final VG (mmHg) | Baseline MR grade | Final MR grade |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | CE, Porcine | 27 | 25 | MR: reduced leaflet mobility | 9 | 22 | Sapien | 26 | 25 | 3 | 3 | TV 1 |
| 2 | Medtronic, Hancock II | 25 | 22 | MR: reduced leaflet mobility | 10 | 21 | Sapien XT | 23 | 9 | 4 | 3 | 0 |
| 3 | CE, Porcine | 27 | 25 | MR: leaflet prolapse | 7 | 25 | Sapien XT | 29 | 28 | 1 | 3 | 0 |
The valves and their manufacturers are as follows: CE porcine (Carpentier-Edwards Porcine mitral valve, Edwards Lifesciences, Irvine, California); Sapien and Sapien XT (Edwards Lifesciences); Medtronic Hancock II mitral valve (Medtronic, Minneapolis, Minnesota); EOA, effective orifice area; ID, inner stent diameter according to manufacturers’ specifications; measured ID, inner stent diameter according to intraprocedural transesophageal echocardiographic measurements; MR, mitral regurgitation; MVR, mitral valve replacement; THV, transcatheter heart valve; TV, transvalvular; VG, mean transvalvular gradient.
Although the amount of patients reported is small, it exhibits that transapical and transseptal mitral VIV implantation can be feasible in high surgical risk patients, and showed favorable clinical and hemodynamic results in short and medium-term follow-up with low morbidity and low mortality. All patients improved NYHA functional class and had an important reduction in the transvalvular gradients and SPAP however; one of the patients died, cannot be attributed to the procedure.
Among the first multicenter registries: Webb et al. described the results of 23 consecutive patients successfully treated with transcatheter valve implantation through a transapical approach. The success rate of the device was 100%. There was no intra-procedural or 30-day mortality. With a median follow-up of 753 days, the survival rate was 90.4%. Clinical improvement in NYHA class I/II heart failure symptoms was observed in all patients except one (95.6%).3
Favorable results have been reported in cases of implantation through a transseptal approach using the Sapien XT (Edwards Lifesciences, Irvine, California) valve,4 and the Melody (Medtronic, Santa Rosa, California) valve5,6 although the great majority of successful cases have used a transapical approach.3,7–9 The Dvir group reported results on 70 patients enrolled in the global VIVID registry (11.4% valve-in-ring, 88.6% valve-in-valve). All patients were treated with Sapien (Edwards Lifesciences, Irvine, California) valve (23mm 22.9%, 26mm 58.6%, and 29mm 18.6%). Transapical access was used in 85.7%, transeptal in 10% and transatrial in 4.3%. Malposition of the device occurred at 4.3%. The 30-day all-cause mortality rate was 10.3% and 82.3% of the patients remained in the I–II functional class at 30 days.10 The transseptal approach can be feasible in selected patients if the transapical approach is not possible for anatomic reasons or due to the surgical experience in the center.
FinancingThe authors did not receive any payment or economic benefit for the preparation and execution of the article.
Conflict of interestsThe authors declare do not have conflict of interest.