Psychosis is a subjective and experiential phenomenon of the mind, influenced by cognitive and socio-cultural patterns of the individual. The neurobiological correlate of this phenomenon is the dysfunction of brain dopaminergic pathways. This article reviews the scientific evidence on the theoretical approaches of the dopaminergic hypothesis of psychosis and its relationship with the reward and salience systems. The aberrant salience occurs when the dysregulation of dopamine transmission produces a mistaken interpretation of neutral or irrelevant stimuli as a source of reward or punishment. Advances in neuroscience achieved in the last decade have led to the conceptualisation of the constructs of visual, social and emotional salience, to test the hypothesis of aberrant salience in psychosis. Psychosis appears, therefore, as a trans-nosological pathological process, relatively non-specific, which alters the attribution system of reality.
La psicosis es un fenómeno subjetivo influenciado por los esquemas cognitivos y socioculturales del individuo, que tiene como correlato neurobiológico la disfunción de las vías dopaminérgicas cerebrales. Este artículo revisa la evidencia científica que sustenta los planteamientos teóricos de la hipótesis dopaminérgica de la psicosis y su relación con los sistemas de recompensa y asignación de relevancia. La salience aberrante o asignación de relevancia aberrante acontece cuando la desregulación de la transmisión de dopamina provoca que estímulos neutros o irrelevantes se interpreten, anómalamente, como generadores de recompensa o castigo. Los avances en neurociencia alcanzados en la última década han servido para conceptualizar los constructos de salience visual, emocional y social, y para testar parcialmente la hipótesis de la salience aberrante en la psicosis. La psicosis aparece, por tanto, como un proceso patológico trans-nosológico, relativamente inespecífico, en el que se altera el sistema de atribución de la realidad.
It can be affirmed that, in genetically susceptible individuals, psychosis represents the last phase of a long path. Delirium, hallucinations and disorganised thought can manifest in a wide range of psychiatric illnesses (schizophrenia, bipolar disorder, cycloid psychosis, postpartum psychosis, chronic delusional disorder, etc.), neurological illnesses (Alzheimer's disease, Parkinson's, Huntington's, frontotemporal dementia, etc.) and even autoimmune diseases (Morvan's syndrome, encephalopathy in potassium channels), although much less frequently.1 Before these psychotic symptoms develop, patients are affected by abnormalities in sensory perception, mood and cognition, which limit and change their ability to process experiences.
In the last few decades, many theories have been proposed, based on neurobiological and psychosocial findings. Unfortunately, none of them have been able to completely cover the complexity of the disorder. Recent advances in neuroscience have allowed the identification of a series of structural, genetic, molecular, biochemical and epidemiological abnormalities that have served as support in forming new proposals.2,3 In this review, scientific evidence was gathered that supported the concept of psychosis as an aberrant state of salience,* as a consequence of dopamine dysregulation and a “common final pathway” of many psychiatric and neurological disorders. Thus, this model did not seek to explain the physiopathology of underlying schizophrenia or Alzheimer's disease (probably more complex, in which several interconnected neurotransmission systems are participating), but rather the final production of psychotic symptoms (delirium, hallucinations and disorganised thought) in an already damaged or dysfunctional brain, where the dopamine system seems to have a central role indeed.
ObjectiveTo review the scientific evidence that supports theories regarding the dopamine hypothesis of psychosis and its relationship with salience and reward systems.
DevelopmentMethods and materialsThe “PubMed” database, up to December 2011, was systematically searched. The following search criteria were used: Salience and Psychosis OR Salience and Schizophrenia OR Salience and Bipolar OR Salience and Delusion. Of the 145 results found, publications based on animal subjects and in languages other than English or Spanish were excluded. Quality of scientific tests was categorised following the recommendations from the Oxford Centre for Evidence-Based Medicine (CEBM).
Dopamine hypothesis of psychosisBefore addressing the role of the dopamine system in clinical expression of psychosis, it is necessary to differentiate the function of its 4 main routes. The mesolimbic pathway (ventral tegmental area–limbic area) is considered to be associated with the modulation of behavioural responses to emotionally gratifying and motivating stimuli. That is to say, it is the cerebral mechanism that processes rewards. The mesocortical pathway (ventral tegmental area–cerebral cortex) has been associated mainly with cognitive function, even though it also modulates responses related to motivation and emotion. The nigrostriatal pathway (substantia nigra–basal ganglia) is involved in motor functioning, and the tuberoinfundibular pathway (hypothalamus–anterior pituitary) regulates prolactin release.
We assign the word psychosis to the altered mental state in which the subject loses their judgment of reality and develops—without awareness of the illness—delirium, hallucinations and disorganised thought. Psychosis is a subjective phenomenon, experienced in the mind. Even if it has a mesolimbic hyperdopaminergic state as a common neurobiological base, it is filtered through the individual's cognitive and socio-cultural circuits. This allows an abnormality in the same chemical (dopamine) to produce different clinical manifestations in different cultures and individuals.4–6
The dopamine hypothesis of psychosis postulates that hyperactivity in the mesolimbic dopamine pathways and disruptions of the D1 and D2 receptors and of the presynaptic terminal are instrumental in the clinical expression of psychotic symptoms.7,8 Imaging studies with radio-labelled l-DOPA showed an increase in presynaptic synthesis of dopamine and in the initial occupation of D2 receptors in the striatum, a finding replicated more frequently in schizophrenia patients.9,10
Pharmacological action on D2 dopamine receptors and its effectiveness in controlling psychotic symptoms, as well as psychotic–mimetic effects of amphetamines, empirically support the dopamine hypothesis of psychosis. Furthermore, there have been neuroimaging studies that support the temporal and quantitative association of this relationship.11
Dopaminergic neurons of the striatum, the main entryway of information directed towards the basal ganglia, have different transmission modes: tonic transmission (low frequency), which seems to be essential in manifesting psychomotor behaviour and allows information to be transmitted to the cortex in a classified and precise way; and phasic transmission (high frequency), which is in charge of detecting sudden changes in stimuli.12 Smith et al.13 proposed that aberrant phasic release of dopamine causes inadequate labelling of internal and external stimuli, thus generating an “aberrant internal model” that constitutes the basis of delusion ideation.
Dopamine and rewardsThe ability to predict rewards and avoid adverse conditions is an essential function for adaptation and survival.14 Dopamine (DA) has the power to modify striatal circuits, strengthening the striated–cortical connections according to reinforcers received through past experiences, thus contributing to future psychomotor behaviours.7,15 This is an example of how dopamine fixes the stimuli–response relationship, favouring learning and predicting reality.
There is universal agreement concerning the central role of the dopamine system in rewards and motivation.5 In a study with monkeys, Schultz et al.16 observed that the unexpected appearance of an award was accompanied by an increase in phasic dopamine transmission and, consequently, learning from the experience. In humans, Jensen et al.14 demonstrated that phasic transmission in response to unexpected events was especially activated in the ventral striatum, while activation of the anterior insula and the orbitofrontal cortex were associated with the valence (attraction–aversion) of the stimulus. Specifically, patients with psychosis presented an abnormal physiological response in the dopamine systems of the middle brain, striatum and limbic region associated with failure in reward prediction.17
What is salience?This refers to a high-order mental process that allows certain objects, mentally perceived or represented, to attract the spotlight, thus being incorporated into thoughts and behaviours. The term assignación de relevancia (relevance assignment), Vargas and Lahera's proposed translation of “salience” into Spanish, can help to better capture and communicate the essence of mental phenomena associated with the dopamine system.18
It has been proposed that during the prodromal period of the psychotic disorder, towards the end of adolescence, there is a disproportionate increase of dopamine neurotransmission in the mesolimbic area, which does not correspond to normal learning and stimulus–response prediction mechanisms.4 This dysregulation of dopamine transmission allows for neutral or irrelevant stimuli, both external and those derived from internal representations, to be interpreted abnormally as reward or punishment generators. Consequently, certain behaviours focused on obtaining a goal are selected.4,19,20 Recently, Palaniyappan21 proposed the concept of proximal salience, referring to a momentary state generated by the assessment of external or internal stimuli in the context of interoceptive awareness. “Aberrant salience” in the acute phase of psychosis would cause rigid emotional states anchored in irrelevant and idiosyncratic stimuli.22 This way, delirium constitutes the cognitive explanation that the individual offers to this anomalous experience in an effort to give it meaning. These explanations temporarily “soothe” the patient and serve as a guiding cognitive framework for future thoughts and behaviours.4 Clinical experience corroborates that the patient's pre-psychotic anguish and bewilderment is considerably reduced when the comprehensive explanation from delirium emerges.
The salience model also offers a plausible explanation for negative symptoms of schizophrenia: disturbance in dopamine regulation can increase “noise” in the system, “drowning” the dopamine signals correctly associated with stimuli that indicate rewards, as Roiser et al.23 and Seamans and Yang25 observed. This is to say, stimuli naturally called upon to motivate (those that arouse the subject's interests and motivate him or her to do something) are mitigated by endless aberrant external and internal stimuli that drive the patient to bewilderment and, chronically, inactivity.
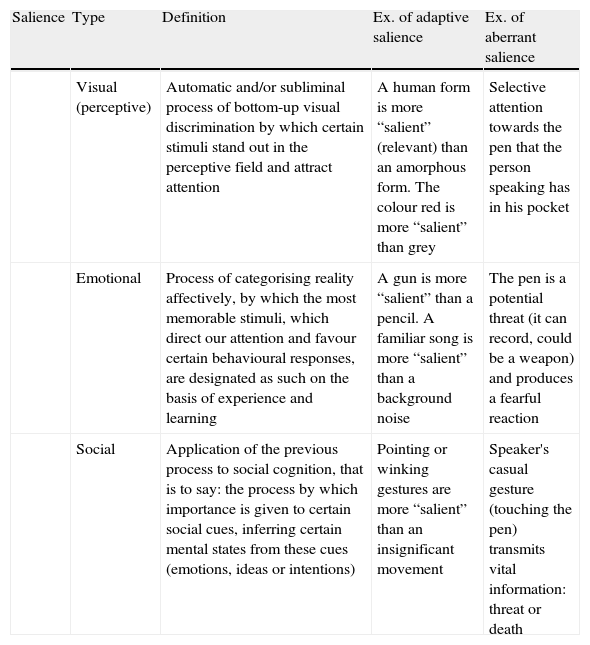
Visual, emotional and social salienceCertain stimuli—relevant to our adaptation and survival—seem to stick out in the perceptual field and powerfully attract our attention. This is the result of an automatic and subliminal process of bottom-up visual discrimination.24 The roles of the thalamus (as the centre of multiple neural connections) and of the thalamic dopamine synapses (as filters of the information sent to the cortex) suggest that the thalamus could be involved in the disturbances in processing sensory stimuli and later in the adaptive learning of rewards.25 Brébion and Ohlsen26 found that patients with visual hallucinations used less serial and semantic coding when they were familiar with the words, as somehow these words allowed them to form mental images. The authors suggested that this finding was due to aberrant salience of mental images that the patients experienced upon perceiving the stimulus (word).
An anomaly in perception and interpretation of reality immediately translates to a social anomaly. Social cognition includes the set of mental processes necessary to infer and predict the mental states of others, thus effectively managing social relationships. Adequate social cognition implies the integrity of the very system involved in emotional processing and regulation, which depends in part on the connections between the amygdala and prefrontal cortical areas.27,28 These pathways are those in charge of assigning emotional relevance to a stimulus: they convert it into something more or less memorable, they direct attention to it or they neglect it, favouring certain behavioural responses towards it or the opposite.29 In psychosis, an aberrant phasic level of dopamine with a dysfunction in emotional processing in the amygdala (for example, interpreting a neutral facial expression as anger or rage) can interfere with cortical operations and prompt threatening perceptive anomalies.29 Such perceptive disturbances with negative emotional states may cause benign interactions to be misinterpreted as hostile ones. Consequently, an attributional bias appears that leads to paranoia and secondary isolation30,31 (Table 1).
Visual, emotional and social salience.
| Salience | Type | Definition | Ex. of adaptive salience | Ex. of aberrant salience |
| Visual (perceptive) | Automatic and/or subliminal process of bottom-up visual discrimination by which certain stimuli stand out in the perceptive field and attract attention | A human form is more “salient” (relevant) than an amorphous form. The colour red is more “salient” than grey | Selective attention towards the pen that the person speaking has in his pocket | |
| Emotional | Process of categorising reality affectively, by which the most memorable stimuli, which direct our attention and favour certain behavioural responses, are designated as such on the basis of experience and learning | A gun is more “salient” than a pencil. A familiar song is more “salient” than a background noise | The pen is a potential threat (it can record, could be a weapon) and produces a fearful reaction | |
| Social | Application of the previous process to social cognition, that is to say: the process by which importance is given to certain social cues, inferring certain mental states from these cues (emotions, ideas or intentions) | Pointing or winking gestures are more “salient” than an insignificant movement | Speaker's casual gesture (touching the pen) transmits vital information: threat or death |
In this vein, McBain et al.32 used high-resolution (defined facial features) and low-resolution (blurry facial features) images of faces to study the relationship between schizophrenia patients’ perception of emotional expressions and basic visual attributes. They concluded that an abnormal and important association between affective and basic visual systems underlies the psychotic patient's emotional perception. The findings of Seiferth and Pauly33 were also interesting, as they demonstrated that subjects at high clinical risk of psychosis presented hyper-activation of the brain regions involved in processing emotional and facial expressions (fusiform gyrus, right lingual gyrus and left middle occipital gyrus). This suggests that these disturbances may be present before the illness is manifested at the cognitive level.
Speechley and Whitman34 assessed the reasoning method called “jumping to conclusions”—the tendency to make hasty and not very informed conclusions—in patients with schizophrenia diagnoses (delusional and non-delusional), bipolar patients and a control group. The results suggested that delusional ideation in schizophrenia is related to a reasoning bias that leads to hasty conclusions based on the hyper-relevancy of coincidences.
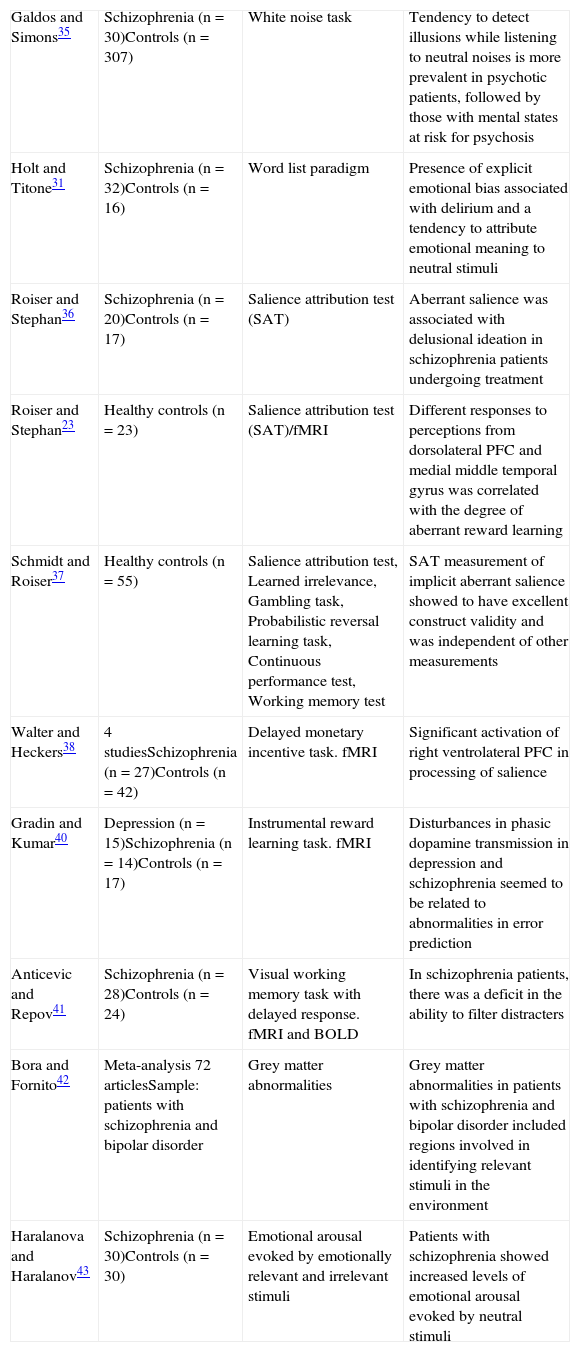
Aberrant salience in psychosis: FindingsIn the last decade, different paradigms have been applied to test the hypothesis of aberrant salience in psychosis (in Table 2, the main studies in this vein are summarised). Galdos and Simons35 developed a tool called the “white noise task” to detect the emotional significance of randomised neutral sounds and its association with variables of vulnerability to psychosis. They found that the tendency to detect relevant illusions while listening to randomised neutral noises was more prevalent in the group of patients with a psychotic disorder, followed by the group with mental states at risk of psychosis. They also observed that those illusions were associated with high levels of positive schizotypy (and not negative schizotypy) in healthy controls.
Experimental findings with respect to aberrant salience in psychosis.
| Galdos and Simons35 | Schizophrenia (n=30)Controls (n=307) | White noise task | Tendency to detect illusions while listening to neutral noises is more prevalent in psychotic patients, followed by those with mental states at risk for psychosis |
| Holt and Titone31 | Schizophrenia (n=32)Controls (n=16) | Word list paradigm | Presence of explicit emotional bias associated with delirium and a tendency to attribute emotional meaning to neutral stimuli |
| Roiser and Stephan36 | Schizophrenia (n=20)Controls (n=17) | Salience attribution test (SAT) | Aberrant salience was associated with delusional ideation in schizophrenia patients undergoing treatment |
| Roiser and Stephan23 | Healthy controls (n=23) | Salience attribution test (SAT)/fMRI | Different responses to perceptions from dorsolateral PFC and medial middle temporal gyrus was correlated with the degree of aberrant reward learning |
| Schmidt and Roiser37 | Healthy controls (n=55) | Salience attribution test, Learned irrelevance, Gambling task, Probabilistic reversal learning task, Continuous performance test, Working memory test | SAT measurement of implicit aberrant salience showed to have excellent construct validity and was independent of other measurements |
| Walter and Heckers38 | 4 studiesSchizophrenia (n=27)Controls (n=42) | Delayed monetary incentive task. fMRI | Significant activation of right ventrolateral PFC in processing of salience |
| Gradin and Kumar40 | Depression (n=15)Schizophrenia (n=14)Controls (n=17) | Instrumental reward learning task. fMRI | Disturbances in phasic dopamine transmission in depression and schizophrenia seemed to be related to abnormalities in error prediction |
| Anticevic and Repov41 | Schizophrenia (n=28)Controls (n=24) | Visual working memory task with delayed response. fMRI and BOLD | In schizophrenia patients, there was a deficit in the ability to filter distracters |
| Bora and Fornito42 | Meta-analysis 72 articlesSample: patients with schizophrenia and bipolar disorder | Grey matter abnormalities | Grey matter abnormalities in patients with schizophrenia and bipolar disorder included regions involved in identifying relevant stimuli in the environment |
| Haralanova and Haralanov43 | Schizophrenia (n=30)Controls (n=30) | Emotional arousal evoked by emotionally relevant and irrelevant stimuli | Patients with schizophrenia showed increased levels of emotional arousal evoked by neutral stimuli |
BOLD: blood oxygenation level dependent; fMRI: functional magnetic resonance imaging; PFC: prefrontal cortex; SAT: salience attribution test.
Holt and Titone31 found evidence in favour of an explicit emotional bias associated with delirium, confirming the hypothesis that delusional ideation arises from inadequate attribution of emotional meaning to neutral stimuli. Roiser and Stephan36 used a probabilistic reward-learning task characterised by relevant and irrelevant perceptive stimuli, called the Salience Attribution Test (SAT). It was used to evaluate psychotic patients’ adaptive and aberrant salience when being treated with antipsychotics. The results obtained support the theory that aberrant salience is related to delusional ideation in schizophrenic patients undergoing treatment. Furthermore, it seems related to negative symptoms. Later, Schmidt and Roiser37 applied a series of neuropsychological tests (Salience attribution test, Learned irrelevance, Gambling task, Probabilistic reversal learning, Continuous performance test, Working memory test) to 55 volunteers without a history of psychiatric disorders in order to test the validity of the SAT as an instrument that measures salience. In particular, measurement of implicit aberrant salience was observed to have excellent construct validity and was independent of other measurements, including learning irrelevance.
Roiser and Stephan23 analysed functional magnetic resonance images (fMRI) in healthy controls while they performed the SAT. They demonstrated that: (1) responses in the dorsomedial thalamus and in the central prefrontal cortex (PFC) were strongly correlated with the degree of adaptive reward learning, (2) the different responses from the dorsolateral PFC and the middle temporal gyrus to perceptions with the same reward probability were strongly correlated with the degree of aberrant reward learning and (3) the relationship between aberrant reward learning and the perceptions assigned with identical reward probability varied among subjects, these perceptions being widely associated with responses from the dorsolateral PFC and the middle thalamic gyrus.
Walter and Heckers38 used a task based on the delay of a monetary incentive in an fMRI study. The authors showed normal to high activation in the ventral striatum when a prediction error occurred, with a hypo-activation of the anterior cingulate and the ventrolateral PFC (both mediators of the attentional process and action selection). They also managed to replicate the findings of previous studies that showed significant activation of the right ventrolateral PFC in salience processing.38 In another functional neuroimaging study, Seiferth et al.33 found that subjects at higher risk of psychosis showed greater activation in the frontal gyrus, thalamus and hippocampus upon seeing neutral faces. This suggested that the inclination towards giving greater salience to neural stimuli could constitute a risk marker for psychosis.
ConclusionsIn order to address the physiopathology of a brain system such as the dopamine system, it is necessary to first reflect on the physiology itself. The concept of salience strongly suggests a connection between the different levels of analysis in psychosis (neurobiological, cognitive, behavioural) and the representative and predictive functions of the human brain. Internal and external stimuli attract attention in proportion to their value (relevance) for our adaptation and survival. If this subtle system that categorises and ranks reality is changed, the subject will live in an unpredictable, erratic and anguished reality (the anomalous experience). This leads to searching for explanations in the form of rigid cognitive frameworks (delirium) and confusing external and internal stimuli (hallucinations). This aberrant environmental salience is generically produced by a phasic hyper-dopamine transmission in the mesolimbic, which can be triggered by different pathological states: schizophrenia, consumption of toxic psychostimulants, stress reactions, extreme moods like mania or depression, dementia, etc. Thus, psychosis appears as a pathological trans-nosological process, relatively non-specific, in which the system for attributing reality is disturbed. Identifying and separating the physiopathology from this epiphenomenon (recently called “salience syndrome”39) will allow us to address the authentic physiopathology of the underlying cause (e.g., schizophrenia).
Conflict of interestThe authors have no conflict of interest to declare.
Please cite this article as: Lahera G, et al. Asignación de relevancia (salience) y desregulación del sistema dopaminérgico. Rev Psiquiatr Salud Ment (Barc). 2013;6:45–51.
Salience: As shown throughout the present article, this refers to a high-order mental process that allows certain objects, mentally perceived or represented, to attract the spotlight, thus being incorporated into thoughts and behaviours.