To compare the occurrence of haemorrhages among the different oral anticoagulants (OAC) and to analyse factors that influence it.
Material and methodsSingle-centre, observational, retrospective study. After studying the total population treated with OAC, patients who were treated with an OAC from July 2015 to December 2015 in the II Sector of the Zaragoza Hospital, who consulted the Emergency Department of the Miguel Servet University Hospital and presented a haemorrhagic event, were analysed. Patients’ demographic data, clinical variables and data on the haemorrhagic event characteristics were gathered.
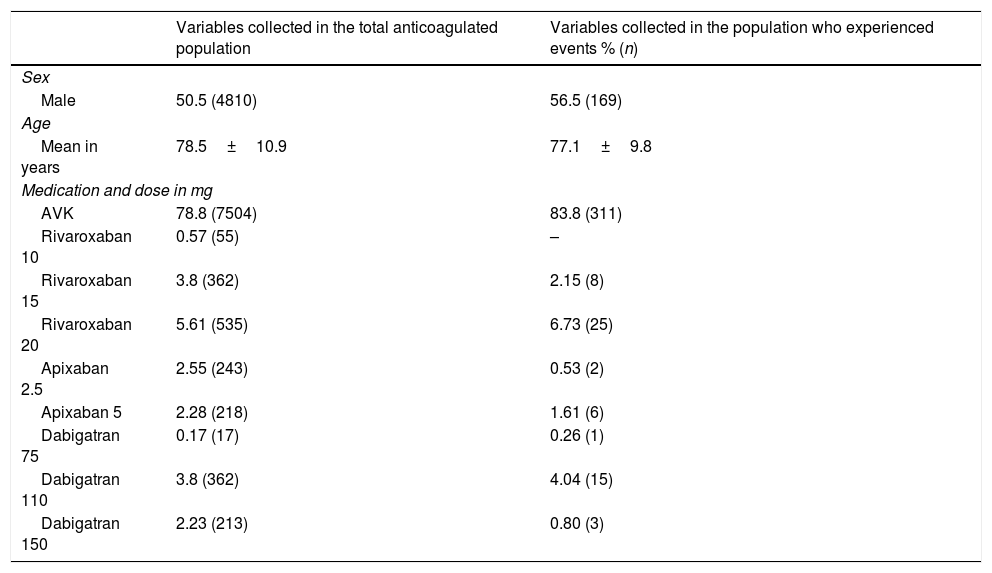
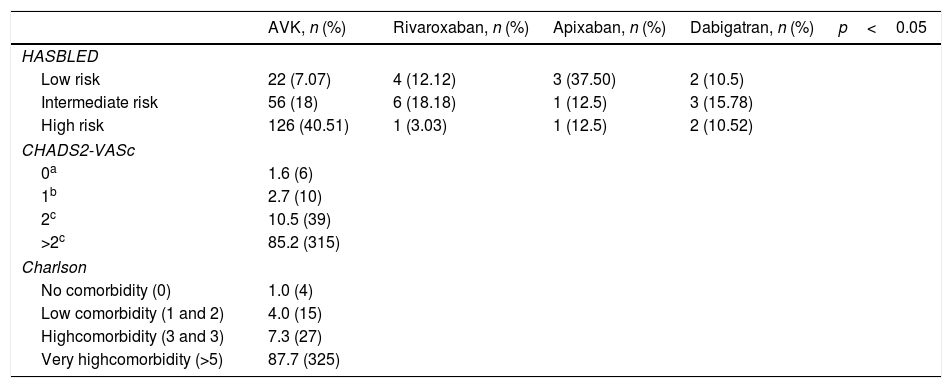
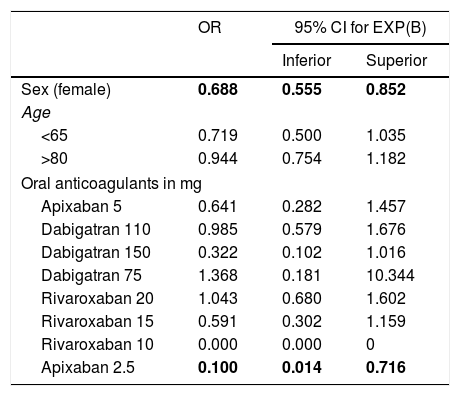
ResultsThere were 9452 patients treated with an OAC, 371 (3.9%) of which presented a haemorrhagic event. The frequency per OAC was; 4.1% (311) in patients treated with vitamin K antagonists, 3.8% (33) with rivaroxaban, 3.3% (19) with dabigatran and 2.1% (8) with apixaban. In the multivariate analysis, only the choice of anticoagulant and sex had a statistically significant influence of a lower risk of haemorrhage, in particular the dose of apixaban at 2.5mg and being female (OR=0.1, CI=0.014–0.71 and OR=0.688, CI=0.55–0.85, respectively).
ConclusionAccording to the results obtained, females and patients undergoing treatment with apixaban presented lower haemorrhagic risk, although there are doubts about whether this better safety profile is related to underdosing, which could influence its effectiveness. Therefore, these results should be analysed with caution and further studies are needed to confirm this data.
Comparar la incidencia de hemorragias entre los anticoagulantes orales (ACO) y analizar los factores que influyen en la aparición de hemorragias.
Material y métodosEstudio observacional, retrospectivo unicéntrico. Tras estudiar la población total en tratamiento con ACO, se analizó a los pacientes en tratamiento con un ACO del sector II de Zaragoza que acudieron al Servicio de Urgencias de Hospital Universitario Miguel Servet de julio a diciembre de 2015 por presentar algún evento. Se registraron datos demográficos, variables clínicas y características del evento hemorrágico. Como factores independientes en la aparición de hemorragias se valoró la dosis, fármacos, el sexo y la edad.
ResultadosHabía 9.452 pacientes en tratamiento con ACO, de los cuales 371 presentaron un evento hemorrágico (3,9%). La frecuencia por ACO fue 4,1% (311) en pacientes tratados con antagonistas de la vitamina K (AVK), 3,8% (33) con rivaroxaban, 3,3% (19) con dabigatran y, por último, con apixaban 2,1% (8) (p<0,05). En el análisis multivariante solo obtuvieron una influencia estadísticamente significativa la selección del anticoagulante y el sexo, en concreto, la dosis de apixaban 2,5mg y ser mujer presentaban menor riesgo de hemorragia (OR=0,1; IC=0,014-0,71 y OR=0,688; IC=0,55-0,85, respectivamente).
ConclusiónSegún los datos obtenidos, las mujeres y los pacientes en tratamiento con apixaban presentaban menor riesgo hemorrágico, si bien existen dudas de si este mejor perfil de seguridad está relacionado con una infradosificación, que podría influir en su efectividad. Por lo tanto, estos resultados deben ser analizados con prudencia y se deben realizar más estudios para confirmar estos datos.