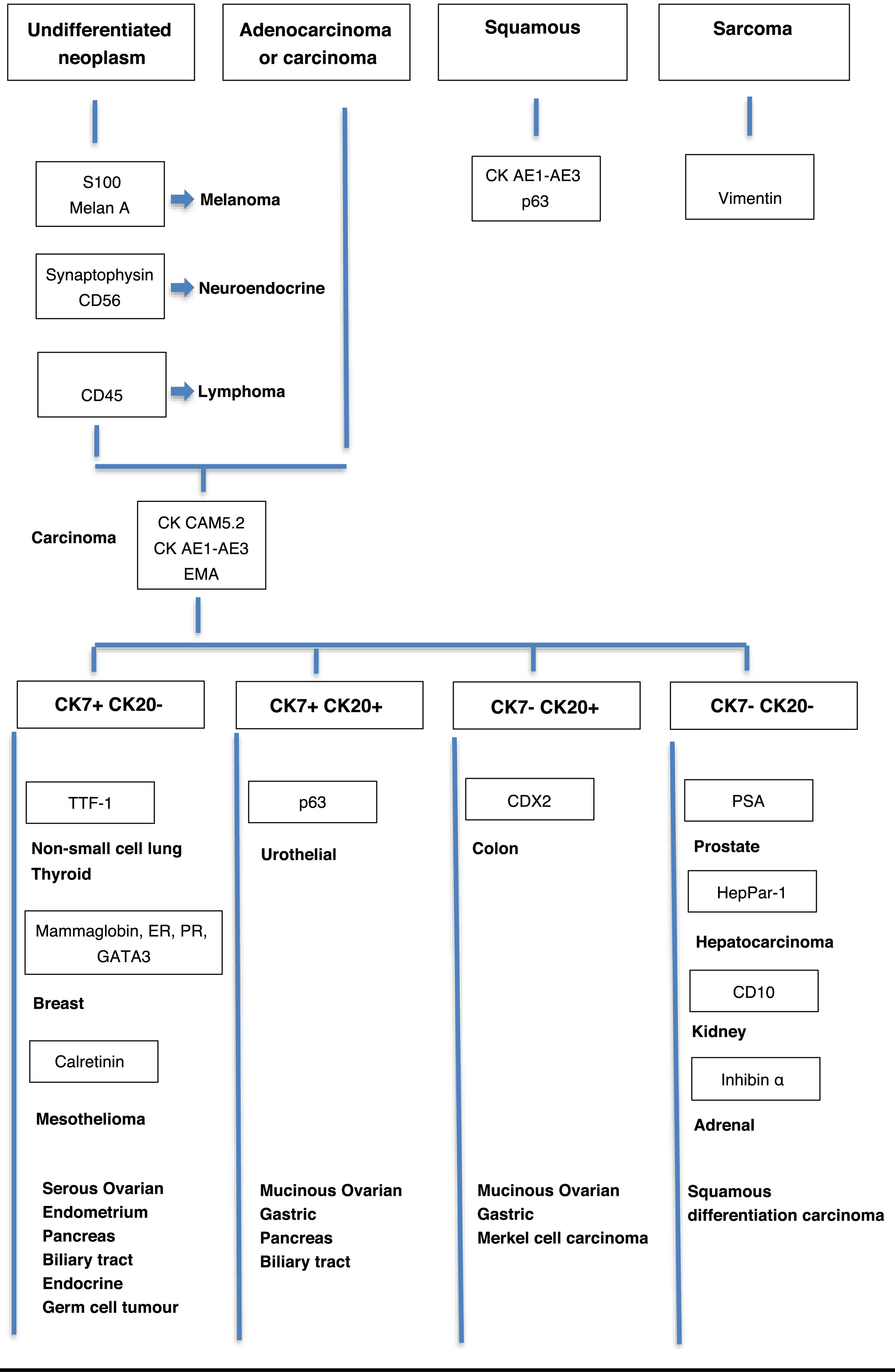
Cancer of unknown primary (CUP) is a clinical type of metastatic malignancy where the primary tumor remains unidentified despite thorough diagnostic evaluation. It presents a significant challenge in modern oncology due to its complex management and diagnostic difficulties. Although CUP accounts for approximately 2 to 5% of cancer cases worldwide, recent advancements in diagnostic techniques, such as immunohistochemistry and genomic sequencing, have improved the ability to classify and treat CUP using targeted therapies. However, limitations persist, including the biological heterogeneity of CUP and the frequent need for empirical treatments. This review discusses advancements in CUP diagnosis and treatment, emphasizing the importance of a multidisciplinary approach that integrates precision oncology and palliative care to enhance patient quality of life.
El cáncer de origen desconocido (COD) es un tipo de neoplasia metastásica cuyo tumor primario no se identifica tras una exhaustiva evaluación diagnóstica. Representa un desafío importante en la oncología moderna debido a la complejidad en su diagnóstico y tratamiento. Aunque el COD constituye entre el 2 y el 5% de todos los casos de cáncer, los avances recientes en técnicas diagnósticas, como la inmunohistoquímica y la secuenciación genómica, han mejorado la capacidad de clasificar y tratar el COD mediante terapias dirigidas. No obstante, persisten limitaciones, incluidas la heterogeneidad biológica del COD y la necesidad de tratamientos empíricos todavía en muchos casos. La presente revisión discute los avances en el diagnóstico y tratamiento del COD, enfatizando la importancia de un enfoque multidisciplinar que integre la oncología de precisión y los cuidados paliativos para mejorar la calidad de vida de estos pacientes.