We present the case of a 75-year-old male with a history of alcohol consumption of 25g/day, hypertension, diabetes, dyslipidaemia, hepatic steatosis and angina on exertion. The patient was being treated with insulin glargine, metformin, sitagliptin, enalapril, rosuvastatin, ASA and paracetamol.
He sought care due to the following signs and symptoms: fever >39°C with chills for the last 24h in the context of flu syndrome and asthenia for the last 2–3 weeks. He denied increasing his alcohol consumption or ingesting high doses of paracetamol. His physical examination was unremarkable, with no jaundice of the skin or mucous membranes, pain, or hepatomegaly on abdominal palpation.
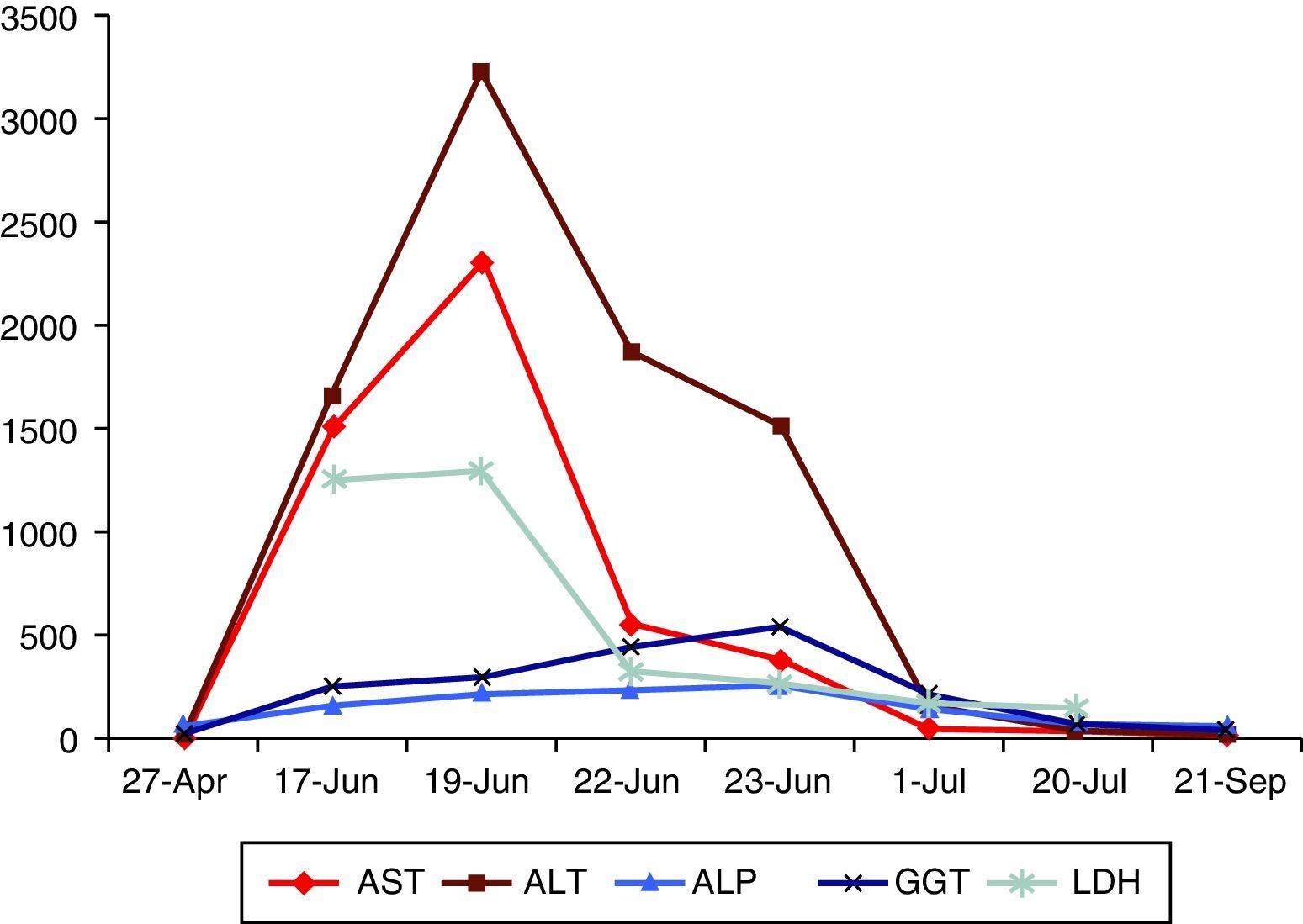
Laboratory testing revealed an abnormal liver biochemistry profile with a predominance of cytolysis (AST: 1516U/l; ALT: 1663U/l; BRB: 1.34mg/dl; ALP: 156U/l and GGT: 250U/l). Ultrasound revealed gallstones, with no dilation of the bile duct. HBV, HCV, HAV and CMV serologies were performed and came back negative. Blood cultures were sterile. His ferritin was 4849ng/ml, and his iron saturation index was 53%. His ceruloplasmin, thyroid hormones and proteinogram were normal. His ANA and HIV tests were negative.
His flu-like signs and symptoms, fever, pattern of transaminases with AST/ALT<1, non-increased alcohol consumption and non-ingestion of high doses of paracetamol raised suspicion of an infectious cause. Although his iron metabolism was highly abnormal, it had to be taken in the context of fever and hepatocellular necrosis. There were no abnormalities in prior laboratory tests, which was not suggestive of haemochromatosis, since this normally presents as hypertransaminasaemia that is less marked and more consistent over time.
Therefore, the patient was questioned again, and he reported that he often consumed minced beef and pork at home that he neither cooked nor marinated. In view of this, hepatitis E serology was ordered and confirmed acute HEV infection (positive IgM with negative IgG). HEV RNA could not be determined.
The patient's condition quickly improved and he was discharged with no fever 5 days after admission to hospital. An outpatient visit a month after discharge found complete clinical resolution and normalised liver biochemistry (Fig. 1). His ferritin and iron saturation index also normalised during follow-up. Anti-HEV IgG was seen to develop.
Hepatitis due to hepatitis E virus (formerly known as water-borne or enteric non-A, non-B hepatitis) is an acute viral hepatitis transmitted via the faecal–oral route.1–5 HEV is an RNA virus with known 4 genotypes with different signs and symptoms and different epidemiologies.1,5–7 Genotypes 1–2 only affect humans. Genotypes 3–4 affect humans and animals; 3 is less virulent.1,5,7 Genotype 3 is characteristic of non-endemic countries.5,6 It is also the only genotype that may become chronic; this process is promoted by states of immunosuppression.2,7 It is more common in countries with limited resources, with poor access to water and sanitation; however, more and more cases are being reported in developed countries.1,5–7 The prevalence of anti-HEV antibodies is 21% in the United States (more common in those who live with pets and those who consume offal more than once per month).4 It is unknown why, despite these prevalences, acute hepatitis due to HEV is not more common. It has been suggested that mild signs and symptoms may be related to the lower virulence of genotype 3.3,6,7 In Spain, the prevalence of anti-HEV antibodies is 0.6%–7.3% in the general population,2,5–7 and higher (up to 19%) in populations in contact with swine livestock.6,7 A study in Córdoba reported an annual incidence of 7.2% in HIV+ patients.8
In resource-poor countries, its transmission resembles that of HAV, with epidemic outbreaks essentially due to faecal contamination of water.1,2,4,5,7,9 Sporadic cases have been reported in Western countries, both due to travel to endemic areas and due to autochthonous transmission, mainly as a result of consuming undercooked meat or liver.1,2,4,7,9 Transmission through blood transfusions is possible, and transmission through interpersonal contact is uncommon.7
RNA from HEV belonging to a genotype identical to that detected in a patient with acute hepatitis due to HEV who had ingested frozen boar and deer meat intended for consumption has been isolated in this meat.9,10 Similarly, simultaneous cases of acute hepatitis E have been reported in individuals who had consumed undercooked meat from the same animal.9 The prevalence of anti-HEV IgG has also been shown to be higher in those who consume raw meat than in those who do not.1
In conclusion, in acute hepatitis of probable viral origin, it is necessary to perform a suitable medical history that includes information on the consumption of raw or undercooked meat products. This is particularly important if there are no indications of other aetiologies and HAV, HBV, HCV and CMV serologies are negative.
Please cite this article as: de Zárraga Mata C, Escudero Roldán M, Thomas Salom G, Rull Murillo N, Dolz Abadía C. Hepatitis aguda por virus de la hepatitis E tras el consumo de carne cruda. Gastroenterol Hepatol. 2017;40:395–396.