Nontuberculous mycobacteria (NTM) can cause skin and soft-tissue infection. Although uncommon, cases of surgical wound infection have been reported. Their diagnosis is important for proper treatment.
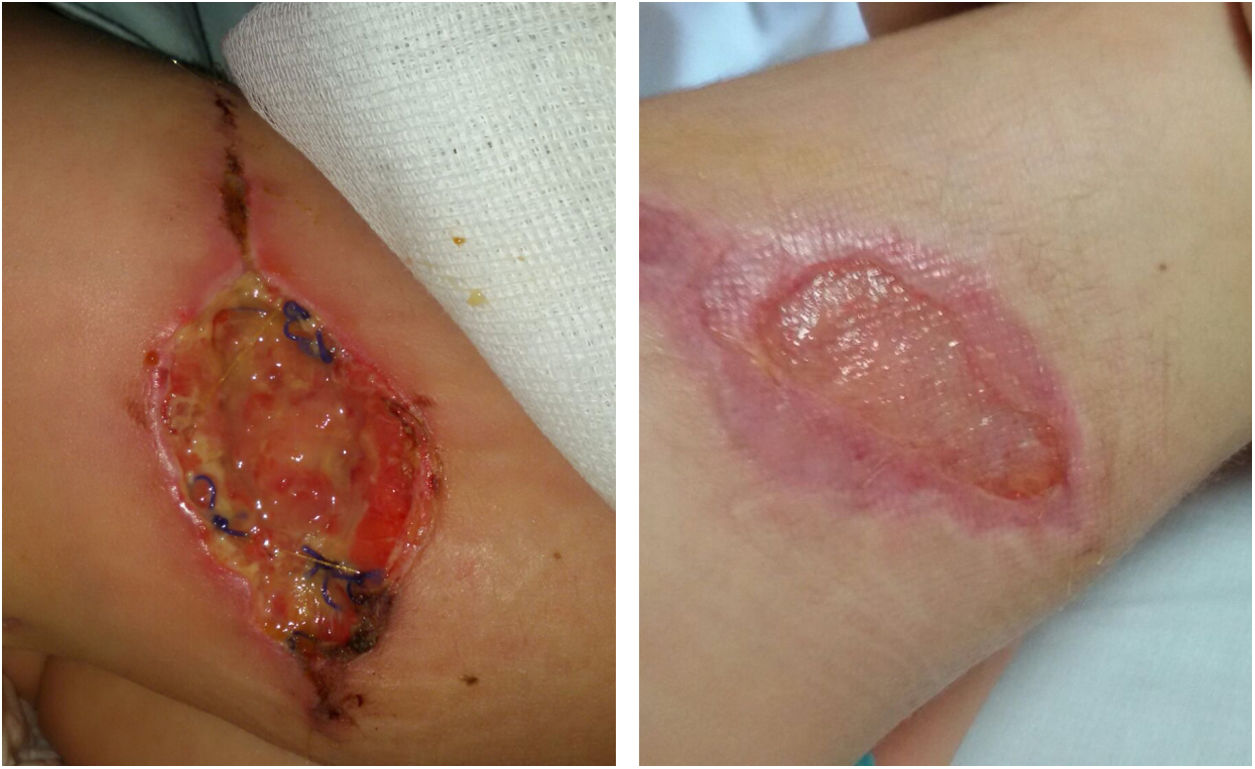
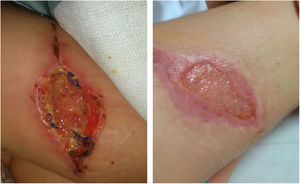
We report the case of a 4-year-old girl who underwent surgery for a congenital melanocytic nevus 6cm in diameter on her right thigh. Five days after surgery, she presented swelling of the surgical site. This was treated with amoxicillin/clavulanic acid. Nevertheless, the lesion increased in size, and wound dehiscence occurred. She underwent surgery again after 20 days and debridement was performed.
Exudate samples were seeded on standard non-selective media, blood agar and chocolate agar, as well as on specific media for mycobacteria: Löwenstein-Jensen and Bactec MGIT 960 liquid medium (BD Diagnostics, United States). At 72h, tiny, translucent colonies grew. These colonies were identified using MALDI-TOF mass spectrometry (Bruker Daltonics GmbH, Leipzig, Germany). The strain was identified as Mycobacterium senagalense with a score of 2.1. Given the high genetic similarity between M. senegalense and Mycobacterium conceptionense, which was recently reported,1,2 the species were distinguished by biochemical and growth characteristics.3 Specifically, M. senegalense was positive for inositol and negative for mannitol, and it grew at 42°C, which confirmed the diagnosis. In the antibiotic sensitivity study using Etest®, the strain was sensitive to amikacin, ciprofloxacin, clarithromycin, doxycycline, cotrimoxazole and imipenem, and resistant to tobramycin and linezolid.
Treatment was started with ciprofloxacin and clarithromycin. Subsequently, the patient presented surgical wound dehiscence again. A decision was made to close the wound by secondary intention. Co-trimoxazole was added, then suspended after 20 days due to leukopenia.
An ultrasound showed no muscle involvement. The patient followed a favourable course (Fig. 1). She remained afebrile, without elevation of acute-phase reactants. Immune studies were normal. A genetic test for Mendelian susceptibility to mycobacterial disease showed no mutations.
M. senegalense is a pathogen related to diseases of cattle on the African continent (bovine farcy). It belongs to the group of rapidly growing NTMs, and is isolated in water, soil and animals. It is capable of forming biofilms, which makes it difficult to eradicate. M. senegalense belongs to the M. fortuitum group, which includes other species such as M. peregrinum, M. mageritense, M. septicum, M. houstonense, M. boenickei, M. neworleanense and M. brisbanense.
Advances in molecular biology have led to an increase in the diagnosis of these infections.1 As a human pathogen M. senegalense is likely underdiagnosed, due to its similarity to other mycobacteria in this group. Through 16SRNA sequencing, Wallace et al. found that 43 isolates previously identified as M. peregrinum corresponded to M. senegalense.4
We found limited references in the literature to M. senegalense as a cause of disease in humans. The first reported case was in 2005 in Korea and was related to bacteraemia associated with central catheter infection in a patient with non-Hodgkin lymphoma.5 Talavlikar et al. reported the case of a girl who, in a domestic accident in which a fish tank broke, suffered various facial injuries in which M. senegalense was isolated.3 In 2019, Maupin et al. reported a case of tibial osteomyelitis in which this micro-organism was isolated.6 To our knowledge, our case represents the first report of surgical wound infection by M. senegalense. NTMs cause nosocomial infections in healthcare institutions in developed countries, where they are usually concentrated in water pipes. Kohsravi et al. found that 7.7% of the mycobacteria isolated in the water pipes of a hospital were M. senegalense.7
Rapidly growing NTMs are resistant to conventional antituberculosis drugs. This justifies the combined use of antibiotics and points to the need to study the sensitivity of the isolated strain in each case.8
In our case, we used a combination of clarithromycin, ciprofloxacin and cotrimoxazole, in the same way as Talavlikar et al.3 In the other cases reported, the mycobacterium was equally sensitive to these three antibiotics. Currently, there are efficacy and safety data on the use of fluoroquinolones in children, so their use in complicated infections, such as the one we report, would be justified.9,10
The duration of treatment is not defined, although prolonged regimens are recommended; in our case, the duration of treatment was six months. In addition to antibiotic therapy, given said capability of forming biofilms, surgical debridement should be performed in skin and soft-tissue infections.
FundingThe authors declare that they have received no outside funding.
Conflicts of interestThe authors declare that they have no conflicts of interest.
Please cite this article as: Santos-Pérez JL, Delgado-Mainar P, Toro-Rueda C, Baquero-Artigao F. Infección de herida quirúrgica por Mycobacterium senegalense en paciente pediátrico. Enferm Infecc Microbiol Clin. 2021;39:259–261.