The aim of this study was to determine the prevalence of HIV and the acceptability of rapid testing in an Emergency Department (ED), Barcelona (6/07/2011 to 8/03/2013).
MethodA convenience sample was used, depending on nurse availability in the ED. Participants signed an informed consent. Results were confirmed by conventional methods.
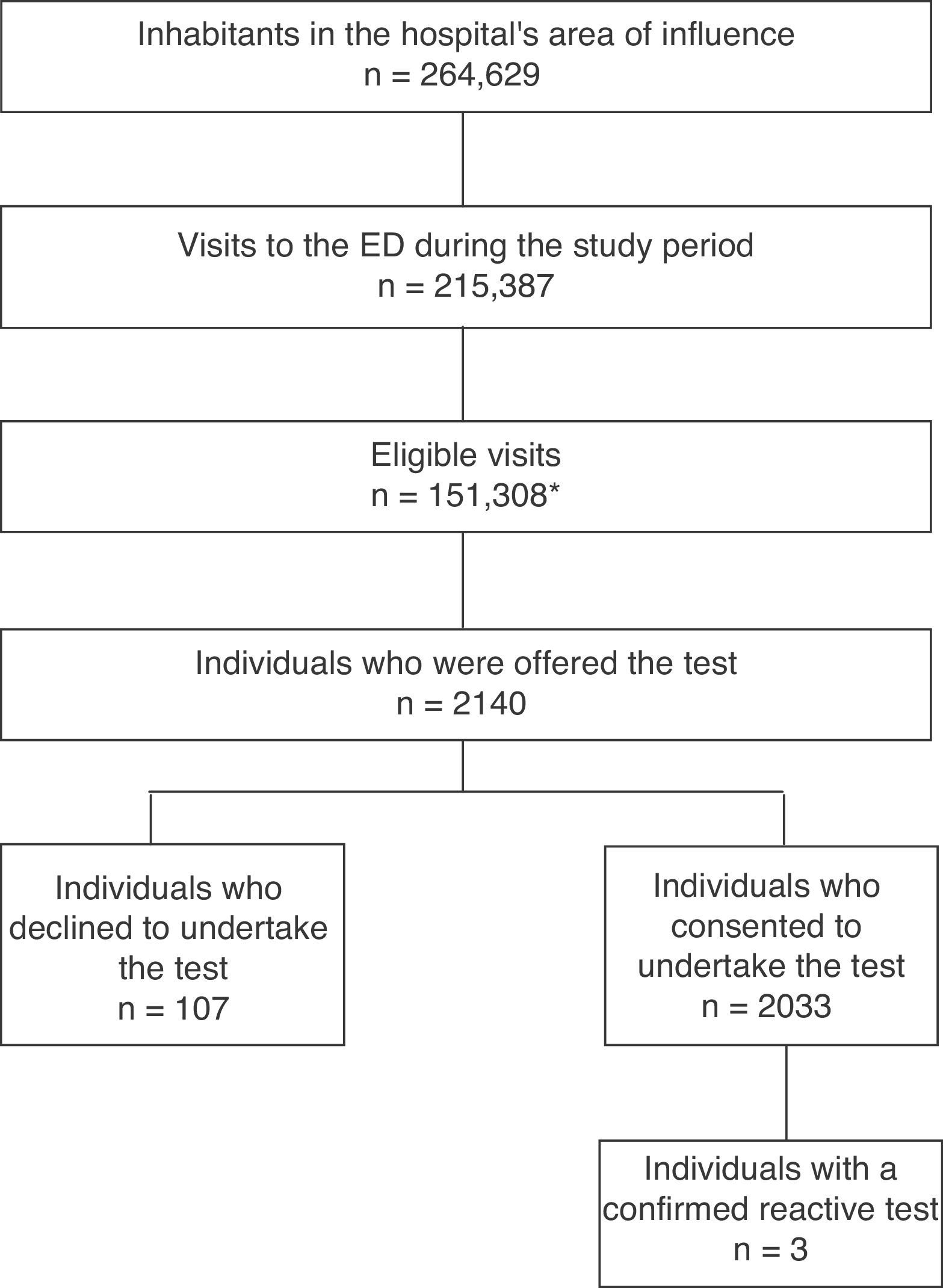
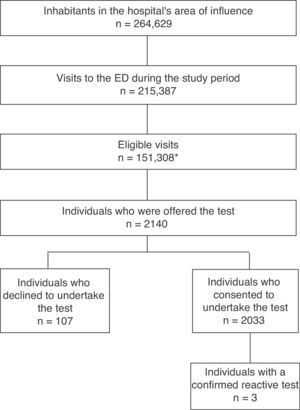
ResultA total of 2140 individuals were offered testing, and 5% rejected taking part (107/2140). Three subjects (3/2033 [0.15%]) had confirmed reactive test. Individuals with a higher education were more likely to perform a rapid HIV test in ED (P<0.005).
ConclusionA low prevalence of new HIV diagnoses was found among participants, although there was a high acceptability rate to perform rapid testing in the ED.
El objetivo fue determinar la prevalencia de VIH y la aceptabilidad del uso de la prueba rápida en un servicio de Urgencias (SU) en Barcelona (6/07/2011-8/03/2013).
MétodoEstudio descriptivo que analizó la realización de la prueba rápida del VIH en fluido oral en un SU. La muestra se conformó por conveniencia y los participantes firmaron el consentimiento informado. Resultados reactivos confirmados por métodos convencionales.
ResultadoA 2.140 se les ofreció realizar la prueba. La tasa de rechazo fue de un 5% (107/2.140). Tres sujetos (3/2.033 [0,15%]) tuvieron la prueba reactiva con confirmación del diagnóstico. Sujetos con mayor nivel educativo tenían una mayor probabilidad de aceptar realizar la prueba rápida de VIH en el SU (p<0,005).
ConclusiónSe encontró una baja prevalencia de nuevos diagnósticos de VIH entre los participantes y una alta aceptabilidad a realizar la prueba rápida en el SU.
In 2013, 3278 new human immunodeficiency virus (HIV) diagnoses were reported in Spain, signifying a rate of 7.04/100,0001 (uncorrected due to delayed diagnoses) which stands above the European average (5.7/100,000).2 In Catalonia, it is estimated that 33,600 people live with HIV, of which approximately 8000 do not know they are infected. In 2013, 754 cases of HIV were reported in Catalonia (overall rate of 10.4/100,000); 42% were diagnosed late (CD4 count <350/μl),3 with delayed diagnosis (DD) being highest in heterosexuals, older adults and parenteral drug users, much like the rest of Spain.4
DD is the main preventable cause of HIV-related morbidity and mortality,5 incurs a substantial increase in healthcare costs5 and means that individuals with a late diagnosis may contribute to the ongoing transmission of the infection due to being unaware of their serostatus.6 Different preventative approaches have been suggested in an effort to improve access to the test and thus to reduce DDs as well as the rate of undiagnosed HIV. In the United States, since 2006 the CDC has recommended testing on an opt-out basis in individuals aged between 13 and 64 attending healthcare centres in areas where HIV prevalence is above 0.1%.7 Likewise, in Europe only the United Kingdom8 and France9 promote universal screening at healthcare centres. The other European countries follow the recommendations of the ECDC and WHO/UNAIDS, which advise targeting screening programmes at high-risk groups, thus increasing the positive predictive value of the test and its profitability.10 In Spanish healthcare centres, HIV testing is recommended in people with a clinical suspicion of HIV infection, as well as in asymptomatic patients who relate high-risk practices, prioritising targeted testing.11
Universal screening strategies include offering the rapid HIV test at Emergency Departments (EDs).8,9 EDs have been identified as key departments for offering the rapid HIV test as they may be the first point of access to the health service for the vulnerable population with a DD; they also treat a diverse population similar to the demographic tendencies of the local epidemic, and may lead to rapid diagnosis and the timely initiation of treatment.7 Screening at EDs increases detection rates of new HIV diagnoses,7 improves quality-adjusted life expectancy12 and generates a reduction in DD-related costs.12 The underuse of the test has been identified as one of its drawbacks, due to few ED users agreeing to undertake it.13
In Spain, universal screening has been studied as a preventive practice in various healthcare contexts, albeit rarely in EDs. As a result, this study has the following objectives: firstly, to determine the prevalence of new HIV infection diagnoses detected in participating individuals; and, secondly, to describe the rate of consent of ED users to using the rapid HIV test in a Barcelona hospital.
MethodsA descriptive study that analysed rapid HIV testing and the prevalence of the infection at an ED of a Barcelona hospital, from 06/07/2011 to 08/03/2013. A survey was designed to collect information regarding sociodemographic characteristics; the opinion on and consent to the test at the ED; risk behaviours; and the test result. Inclusion criteria: age between 16 and 65 years; being attended by any ED specialty except Obstetrics and/or Midwifery; negative or unknown HIV serostatus; being conscious in order to consent to participation. The participants signed an informed consent form. At the beginning of the interview, patients who stated that they were HIV-positive were excluded, along with those who had been surveyed at a previous ED visit to thus control duplicate results. The study was approved by the Ethics Committee of the Hospital de Mataró.
The sample was formed out of convenience. Selection was performed consecutively in triage according to the availability of the two nurses recruited for the study, who took turns so that there was only one nurse selecting patients during working hours, on weekdays and according to times of the year. The financial remuneration of the recruited nurses was fixed for the time taken for selection. The screening criteria were only applied to patients the nurses were able to approach to offer the test. To ensure a precision rate of 3%, with a 95% confidence level and an expected unknown proportion (0.8) of positive results, the sample calculated was 2978.
The rapid HIV test was performed in oral fluid (OraQuick ADVANCE® Rapid HIV-1/2 Antibody Test, OraSure Technologies); according to the manufacturer, the sensitivity and specificity of this test in samples of oral fluid is 99.3% (95% confidence interval [95% CI] 98.4–99.7) and 99.8% (95% CI 99.6–99.9), respectively.14 The reactive results were referred to the functional HIV unit of the same hospital, where they were confirmed using conventional techniques.
A descriptive analysis was performed of the sociodemographic characteristics, sexual conduct, high-risk practices for HIV transmission and opinions on the rapid HIV test, as well as a logistic regression model. The response variable was “declining the survey at the ED”; the sociodemographic variables were explanatory. 95% CIs were calculated for the odds ratios (OR). P values of <0.05 were considered to be statistically significant. Version 17 of the SPSS® statistical package was used (SPSS Inc.).
ResultsFig. 1 shows the selection diagram for patients at the ED. During the study period, 151,308 patients were eligible (this figure does not distinguish repeat visits, so refers to eligible visits rather than individuals); of this population, 53% were female and the median age was 53 years. In the same period, the hospital's referral laboratory also reported 29 (0.46% [29/6143]) positive HIV tests requested for the entire area of influence, including primary care. 2140 individuals were offered the test; the decline rate was 5% (107/2140). The mean age was 37 years (SD 13.8); 49.2% were male, 14.5% were immigrants and 96.9% identified as heterosexual.
Three subjects (3/2033 [0.15%]) took the reactive test with subsequent confirmation (95% CI 0.03–0.43). Case 1: 41-year-old MSM; CD4 113/mcl, CD8 228/mcl, viral load 378,207copies/ml (log5.6); reason for consultation: disseminated TB. Case 2: 32-year-old MSM; CD4 460/mcl, CD8 837/mcl, viral load 57,438copies/ml (log4.8); reason for consultation: superinfected sebaceous cyst on the abdominal wall. Case 3: 55-year-old female; CD4 512/mcl, CD8 397/mcl, viral load 225copies/ml (log2.4); history of active breast cancer under chemotherapy treatment on admission to the ED; reason for consultation: fingernail abscess. No indeterminate results were reported.
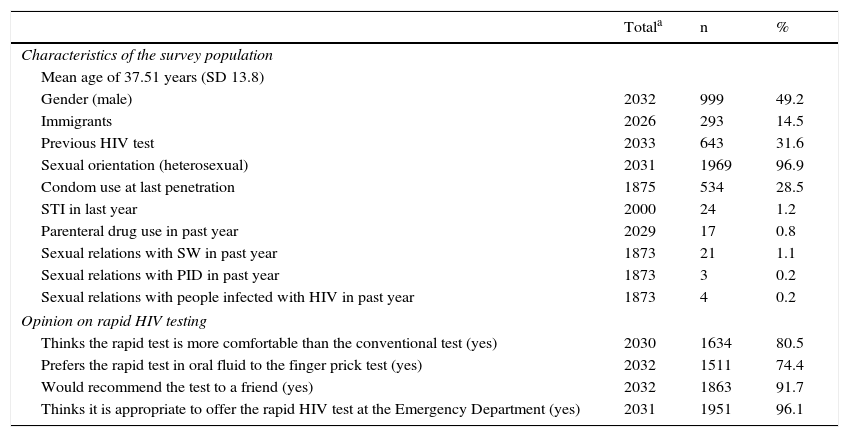
Of the entire sample, 31.6% stated that they had undergone an HIV test in the past. As regards information on risk behaviours, 28.5% stated that they had used a condom the last time they had penetrative sex, and 1.2% had been diagnosed with at least one sexually transmitted infection in the last year (Table 1). As for opinions on rapid HIV testing, 80.5% of the survey respondents felt it was more comfortable than the conventional method, 74.4% preferred the oral fluid method to the finger prick test, 91.7% would recommend it to a friend and 96.1% felt it was appropriate to offer it at an ED (Table 1).
Sociodemographic characteristics, sexual conduct, high-risk practices for HIV transmission and opinions on the rapid HIV test.
| Totala | n | % | |
|---|---|---|---|
| Characteristics of the survey population | |||
| Mean age of 37.51 years (SD 13.8) | |||
| Gender (male) | 2032 | 999 | 49.2 |
| Immigrants | 2026 | 293 | 14.5 |
| Previous HIV test | 2033 | 643 | 31.6 |
| Sexual orientation (heterosexual) | 2031 | 1969 | 96.9 |
| Condom use at last penetration | 1875 | 534 | 28.5 |
| STI in last year | 2000 | 24 | 1.2 |
| Parenteral drug use in past year | 2029 | 17 | 0.8 |
| Sexual relations with SW in past year | 1873 | 21 | 1.1 |
| Sexual relations with PID in past year | 1873 | 3 | 0.2 |
| Sexual relations with people infected with HIV in past year | 1873 | 4 | 0.2 |
| Opinion on rapid HIV testing | |||
| Thinks the rapid test is more comfortable than the conventional test (yes) | 2030 | 1634 | 80.5 |
| Prefers the rapid test in oral fluid to the finger prick test (yes) | 2032 | 1511 | 74.4 |
| Would recommend the test to a friend (yes) | 2032 | 1863 | 91.7 |
| Thinks it is appropriate to offer the rapid HIV test at the Emergency Department (yes) | 2031 | 1951 | 96.1 |
HIV: human immunodeficiency virus; PID: person who injects drugs; SD: standard deviation; STI: sexually transmitted infection; SW: sex worker.
Population surveyed at the Emergency Department of a healthcare institution, Mataró (Barcelona), July 2010–March 2013.
Analysing the association between declining the HIV test at the ED and the sociodemographic variables analysed by means of a logistic regression model revealed a higher probability of rejecting the test in the event of a lower level of education; uneducated (OR 8.21; 95% CI 2.01–33.46) and primary education (OR 5.77; 95% CI 1.75–18.98).
DiscussionThe prevalence of new HIV diagnoses among the people who consented to testing at an ED (0.15%) reported in this study is low, if we compare it to that of the general population of Catalonia (0.4%).3 Conversely, on describing the characteristics of the diagnosed individuals, it is felt that these new cases could have been detected by promoting screening in risk groups, as two of the three subjects with a reactive result were young MSM and the third had an active cancer that would have been diagnosed during her hospital stay. Other authors have reported similar prevalences9 to the one noted in this study, which were located at the limit of 0.1% for cost-effectiveness.15 They also consider that, with the prevalences described and the characteristics of the population diagnosed (high risk and indicator diseases), universal screening at the ED falls within the lower limit of cost-effectiveness; firstly, due to the absence of a hidden epidemic in the low-risk population, and secondly, due to the low efficiency of universal screening at the ED.9 Moreover, it is important to consider that the risk of false positives increases when universal screening is performed on general populations with low prevalences of HIV, thereby leading to a decrease in the test's profitability and thus a reduction in the cost-benefit ratio of the universal screening programme.12
Despite the low offer rate, the consent rate of 95% is quite high in comparison to other studies.9,16 However, on describing the characteristics of the population that declined to undergo the test, different studies have also reported8 that people with a low level of education decline testing due to a low risk perception, poor knowledge of HIV and a low real risk of infection.17 This study did not ascertain the prevalence of HIV among the individuals that declined to undergo the test as no biological samples were available.
The results of this study should be analysed taking into account its limitations: firstly, there are operational limitations, as there was only one nurse offering the test at a heavily visited ED; patient selection depended on her availability and no ED personnel were involved in the study. As described in other studies,8,9 these selection limitations may reflect the operational difficulties that would be present in real conditions, and these studies thus state that in order to reduce these limitations, universal screening should be integrated into EDs, with external personnel and an independent infrastructure, which would imply an elevated cost for health services. Secondly, the offer rate decreased throughout the study period due to the nurses’ exhaustion and lack of financial support; this, in turn, did not allow us to select the total number of the sample calculated. Finally, given that the results are from a single ED and taking into account that the prevalences observed reflect the HIV prevalences among the population attending each ED, this study has limited generalisation.
In conclusion, although there was a high consent rate to rapid resting, the low prevalence detected among the participating subjects reveals that the introduction of universal screening on an opt-out basis at EDs in Spain would identify a non-negligible number of false positives and would have a moderate/low impact on public health; the fact that the only three cases of unknown infection detected were MSM or cancer patients reinforces the recommendation to promote specially-designed voluntary screening for populations with a greater risk, not only via community-based services but also with outreach programmes. Although the authors do not recommend implementing universal screening at EDs, they feel that testing—potentially through rapid diagnostic techniques—should be available at EDs, where HIV infection should be ruled out based on indicator conditions and high-risk behaviours.
Conflicts of interestThis project was funded by Gilead Sciences S.L.
To the men and women who participated in this study and the nurses who carried out the procedure (Mònica Alapont, Maria Carmen Baiges, Aina Ros and Laia Gil).
Please cite this article as: Reyes-Urueña J, Fernàndez-López L, Force L, Daza M, Agustí C, Casabona J. Estudio del impacto a nivel de salud pública del cribado universal del virus de la inmunodeficiencia humana en un servicio de Urgencias. Enferm Infecc Microbiol Clin. 2017;35:434–437.