This case refers to a 47-year-old woman who attended the Emergency Department complaining of swelling and pain in the left hand and forearm for the previous 12h and pyrexia without shivering. The patient, a veterinarian by profession, had been bitten within the previous 24h by a stray cat that had signs of osteomyelitis in its right leg. The patient had been taking amoxicillin/clavulanic acid (500/125mg/8h) prophylactically since she had been bitten. The physical examination revealed three punctate wounds on the palmar surface of the upper left extremity at the level of the thenar eminence, with signs of local inflammation. Also observed was induration of the anterior aspect of the forearm, which ascended from the palm to the axilla, following the course of the lymphatic drainage pathway.
Blood analysis revealed slight leucocytosis (12,500cells/μl) and an elevation of acute phase reactants (CRP: 49.7mg/l), with no other abnormalities in the blood count or clinical biochemistry. No significant findings were observed on hand X-rays. As tenosynovitis was suspected, and to assess the extent of the lesion, we requested a soft tissue ultrasound, which showed signs of cellulitis and a fluid collection on the thenar eminence. We decided to perform surgical drainage, finding three abscesses: the one visualised by ultrasound and another two of smaller volume in the hypothenar eminence. The patient had pyrexia and poorly controlled pain. She was therefore admitted for intravenous antibiotics, continuing with the same drug. Drained purulent material and blood cultures were sent to the microbiology laboratory.
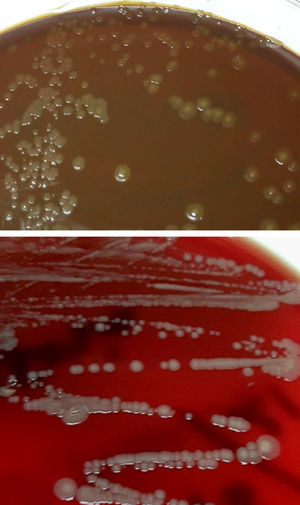
Microbiological diagnosisThe abscess material was spread in general media (blood agar, chocolate agar, Brucella agar and brain-heart infusion [BHI] broth) and selective media (Columbia-CNA agar and MacConkey agar). They were incubated at 36±1°C aerobically, 5% CO2 atmosphere (chocolate agar) and anaerobically (Brucella agar). After 48h, growth was observed in the enrichment medium, and so a subculture was performed on blood and chocolate agar. At 24h, circular, convex colonies, shiny, yellowish-brown, opaque and creamy in appearance, had grown in these plates (Fig. 1). In the Gram staining, large, pleomorphic Gram-negative coccobacilli were observed, grouped mostly in pairs. The oxidase and catalase tests were positive. These colonies were identified by mass spectrometry (MALDI-TOF, Bruker-Daltonics, Germany) as Neisseria zoodegmatis (score=2.4) and with less likelihood as Neisseria weaveri (1.51) and Achromobacter insolitus (1.32). Identification was confirmed by amplification and sequencing of a fragment of the 16s rRNA gene.1 The sequence obtained showed a 100% homology with N. zoodegmatis in GenBank® (access number: NR_043459, KX101082 and JN713518). Blood cultures and anaerobic culture were negative at 5 and 15 days of incubation, respectively.
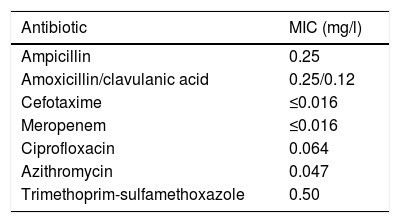
The antibiotic susceptibility testing was carried out using gradient diffusion strips (E-test®, bioMérieux®). In view of the lack of specific cut-off points for this species, we used the epidemiological cut-off values (ECOFF) available for bacteria of the same genus (Neisseria meningitidis, Neisseria gonorrhoeae; http://mic.eucast.org/Eucast2/), for the interpretation of the MIC values (Table 1).
OutcomeThe patient made good progress and was discharged after 48h. She completed the oral amoxicillin/clavulanic acid treatment at home, and had no complications, such as tenosynovitis or arthritis, requiring further hospital assessment.
CommentsOf all animal bites, dog and cat bites are the most common. However, despite being somewhat less common than bites from dogs, cat bites become infected more often (30–50%), and cause far more complications (septic arthritis, tenosynovitis, meningitis, etc.). Infection of wounds from animal bites usually has a polymicrobial aetiology (aerobic and anaerobic microorganisms), resulting from the animal's oral flora or the patient's skin flora.2Pasteurella multocida (one of the most common), Streptococcus spp., Staphylococcus spp., Corynebacterium spp., can all be isolated in cat-bite wounds, in addition to other anaerobic bacteria such as: Peptostreptococcus spp., Prevotella spp., Porphyromonas spp. or Bacteroides spp., among others. Other less common microorganisms, such as Bartonella henselae or Capnocytophaga canimorsus should also be considered, particularly in immunosuppressed patients or patients with asplenia, due to the severity of the symptoms they cause.3
Bacteria of the genus Neisseria are commensals of the oropharyngeal mucosa of healthy dogs and cats, and can also be isolated in bite wounds. N. zoodegmatis (former CDC group EF-4b) is isolated mainly from local infections of wounds that can become complicated, and which are usually underdiagnosed, when mistakenly identified as Pasteurella spp. or contaminating skin flora.4–6 Although it has not been described as a human commensal microorganism, it has been found to be part of the subgingival biofilm in patients with chronic periodontal disease.7
Empirical treatment with amoxicillin/clavulanic acid is the choice for animal bite infections, since it is active against most of the microorganisms involved and, in addition, it can be used in cases in which post-bite prophylaxis is indicated. It is advisable to assess the need in each case for surgical drainage to promote the healing process. In immunosuppressed or splenectomised patients, early intravenous antibiotic therapy and admission are also required, due to the very high risk of the condition developing into fulminant sepsis.8,9
The authors thank the technical staff, L. del Pino and M.J. Yubero, for their collaboration in carrying out this work.
Please cite this article as: Suárez-López A, Ruiz-Garbajosa P, Sánchez-Díaz AM. Infección de herida por mordedura de gato. Enferm Infecc Microbiol Clin. 2018;36:194–195.