Pituitary tumors secreting thyroid-stimulating hormone (TSH) tumors are uncommon. Most series suggest a frequency less than 1%, and are, therefore, a rare cause of hyperthyroidism.1 A somewhat higher frequency (less than 2%) has recently been reported, with an incidence of one case per million population.2 It is not clear whether this incidence has increased or whether tumors are being more accurately diagnosed because of improved diagnostic methods.
The clinical signs are similar to those of other more common forms of hyperthyroidism. Tumors are biochemically characterized by elevated peripheral hormone levels with normal or elevated TSH levels. It is important to make a differential diagnosis with thyroid hormone resistance (THR), both peripheral and pituitary. The main difference between them is the absence of symptoms in peripheral THR, while mild symptoms of hyperthyroidism usually occur in pituitary THR.
Only a few cases of subacute thyroiditis after surgery for a TSH-secreting adenoma have been reported in the literature.3
We report the case of a 37-year-old female patient who attended the endocrinology clinic reporting sweating, vomiting, diarrhea, and nocturnal palpitations during the previous two months. She had no personal or family history of thyroid disease. When specifically questioned again, she reported no weight loss or other associated symptoms, and did not usually use iodinated salt. Physical examination findings included a weight of 62kg, a body mass index of 26kg/m2, blood pressure values of 130/80mmHg, a heart rate of 100 beats per minute, and no distal tremor or exophthalmos. She had elastic, grade 1 goiter with no apparent nodules.
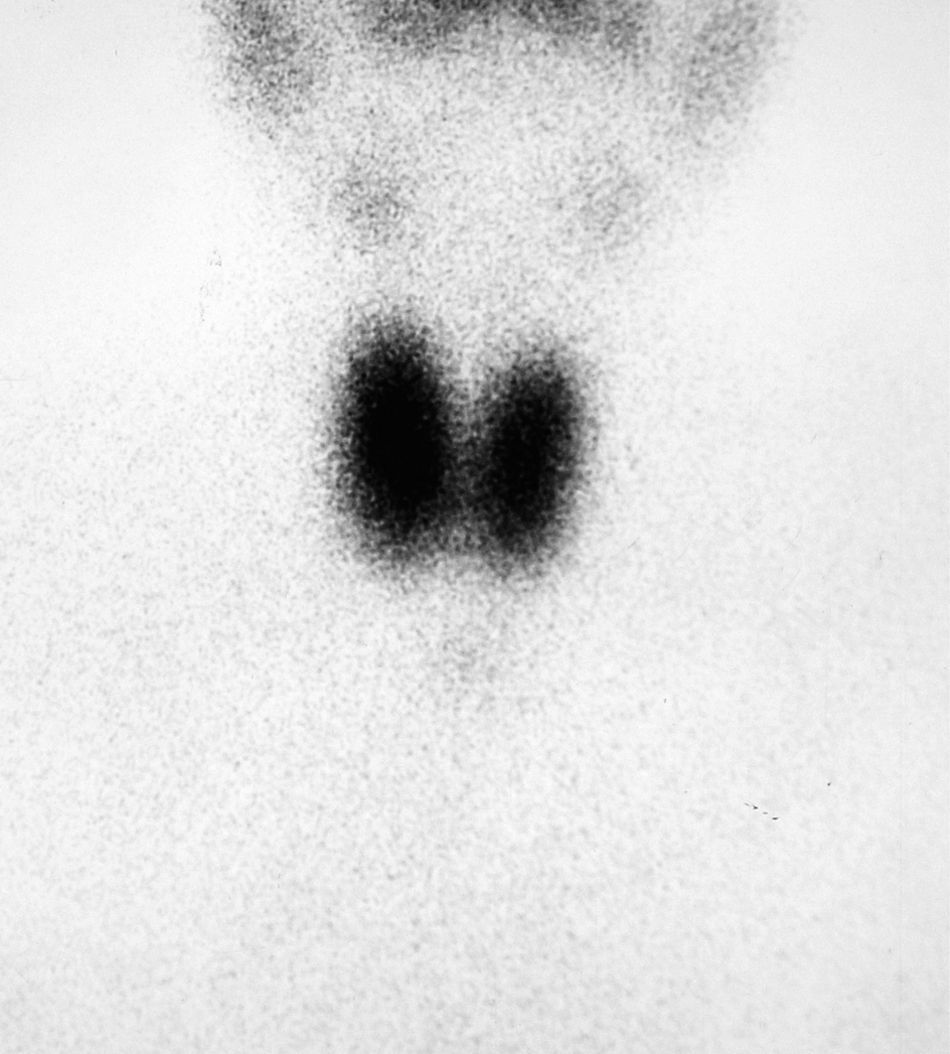
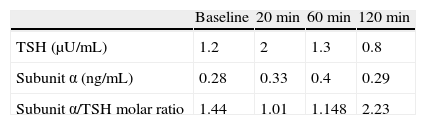
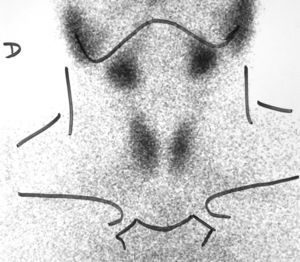
Hyperthyroidism was suspected, and laboratory tests and a thyroid scan were performed. Laboratory test results included free T4 levels above the normal range [2.2ng/dL (NR 0.9–2)] and inappropriately normal TSH [0.72μU/mL (NR 0.3–4.5)], and Tc-99 scintigraphy showed diffuse hyperuptake (Fig. 1).
Repeat laboratory tests performed one month later confirmed the same biochemical changes, and measurements of autoimmune markers including TSH receptor antibodies (TSI) and antithyroglobulin and antiperoxidase antibodies were requested. Total T3 levels were also requested and were found to be above the normal limit [169ng/dL (NR 80–160)]. The presence of elevated peripheral hormones, inappropriately normal TSH levels, and clinical signs and symptoms led to a TSH-secreting pituitary tumor being suspected as the first diagnostic possibility. A thyrotropin-releasing hormone (TRH) test was therefore performed with 150μg IV for diagnosis and differential diagnosis with THR, as well as magnetic resonance imaging (MRI). Alpha subunit levels were also measured (NR<0.1ng/mL), and the subunit α/TSH molar ratio was calculated. Table 1 shows the results of the TRH test.
The absence of a TSH response to TRH and the presence of a subunit α/TSH molar ratio higher than 1 at all times supported the diagnosis of a TSH-secreting tumor.
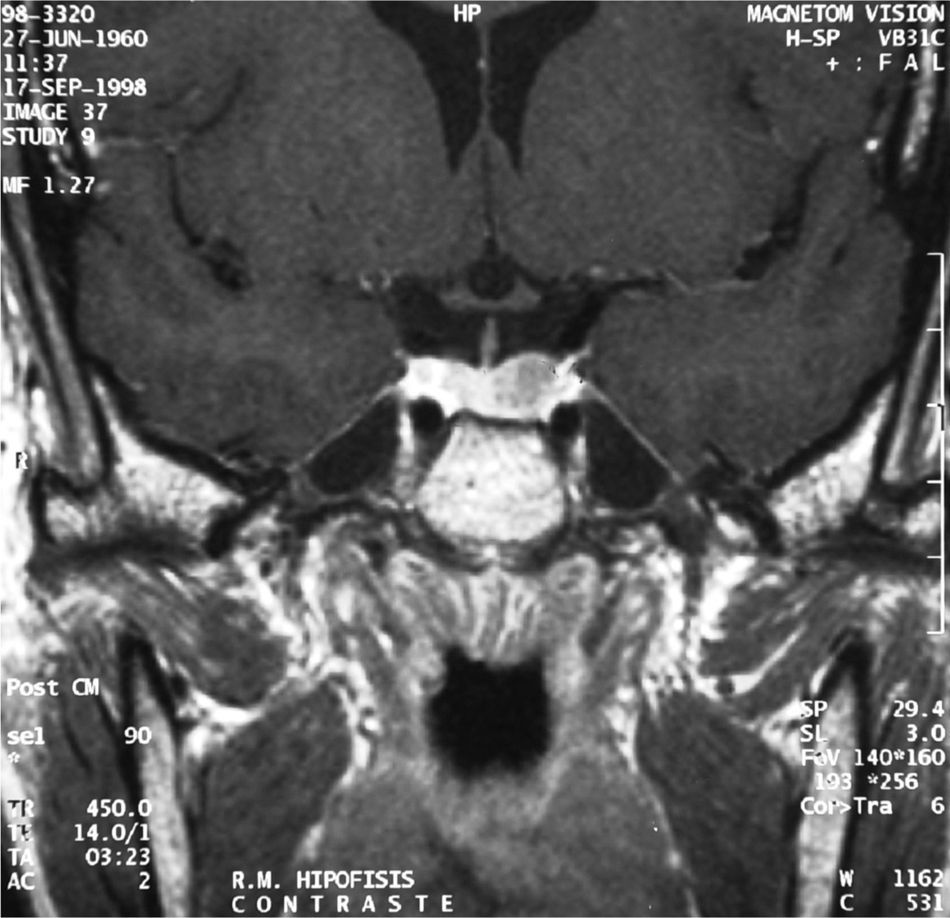
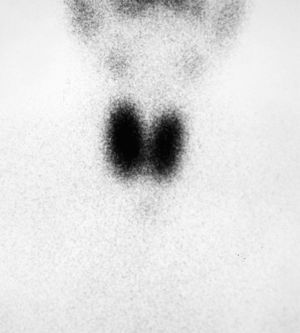
MRI showed a pituitary gland enlarged in its left part. It had not displaced the stem or caused sella turcica erosion or enlargement, and its size of 1.2cm was consistent with a pituitary macroadenoma (Fig. 2).
The remaining pituitary function was assessed, and a campimetric study was performed with normal results.
The patient was admitted to hospital for surgery, after receiving a cycle of octreotide (100μg/8h) to fight symptoms of hyperthyroidism. Laboratory tests upon admission showed free 2.1ng/dL of free T4 with 1.4μU/mL of TSH. On the fourth day of treatment with octreotide, TSH and free T4 levels were 0.08μU/mL and 1.5ng/dL, respectively. Apparently total adenoma resection was performed through transsphenoidal surgery. Pathological and immunohistochemical diagnosis was consistent with pituitary adenoma positive for TSH, prolactin, and GH.
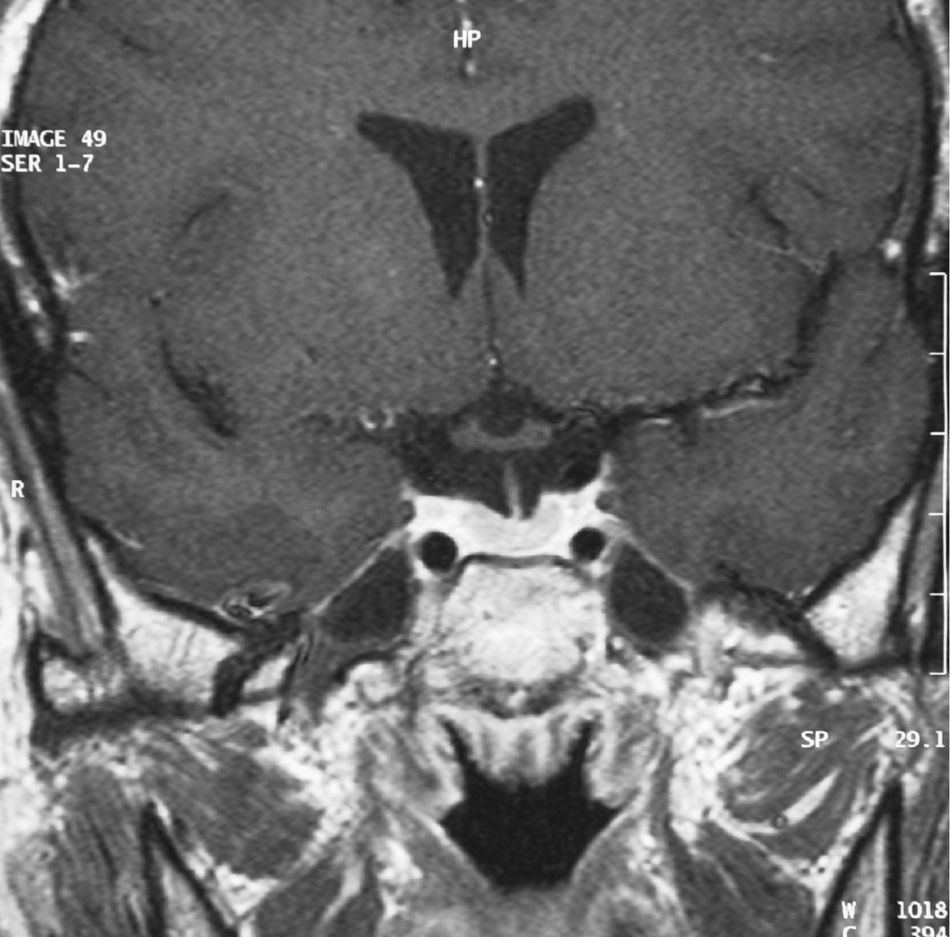
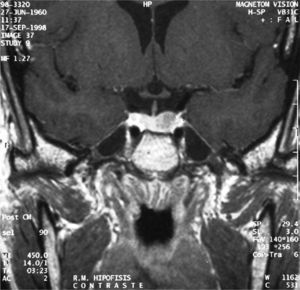
One week after surgery, laboratory test results were within normal ranges (free T4 1.22ng/dL, TSH 0.11μU/mL, total T3 85ng/dL) and the patient showed no symptoms, thus suggesting cure. Three months later, stimulation tests performed to study the functioning of the pituitary axis found normal results, and MRI showed no signs of recurrence (Fig. 3).
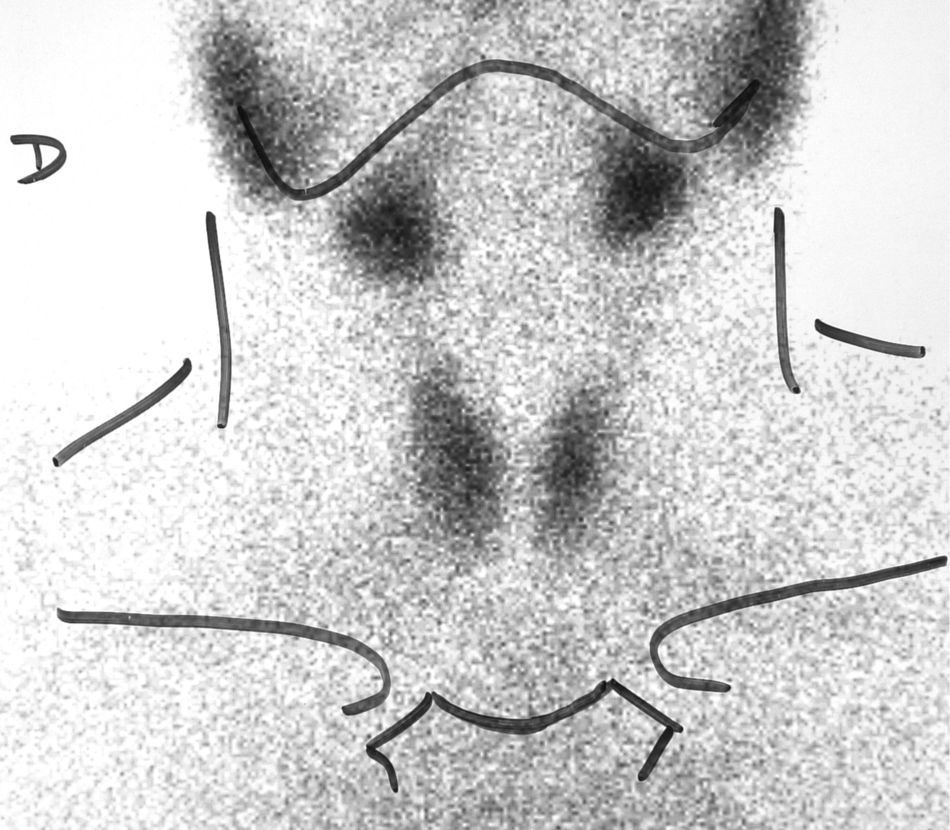
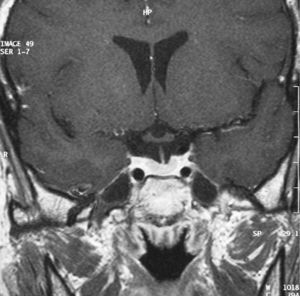
Four months after surgery, the patient reported sudden pain in the right side of the neck and palpitations, nervousness, and tremor. Test results included elevated free T4 level (2.6ng/dL), TSH 0.04μU/mL, and erythrocyte sedimentation rate of 53mm/h. Palpation revealed an enlargement of the right side of the thyroid gland and elevated temperature (38.5°C). A Tc-99 scintigraphy showed diffuse, homogeneous hypo-uptake (Fig. 4). Subacute thyroiditis was suspected, and treatment was therefore started with aspirin 500mg/8h, with gradual dosage reduction. Eight weeks later, the local symptoms had subsided, and the patient reported mild signs of hypothyroidism. Laboratory test results included free T4 0.5ng/dL and TSH 0.1μU/mL. It was decided to wait without prescribing replacement therapy, and two months later the patient was symptom-free and her laboratory values had normalized.
At subsequent visits, thyroid function test results have been normal, and MRI has shown no evidence of tumor recurrence.
There are only a few reports in the literature of the concomitant occurrence of subacute thyroiditis and a TSH-secreting tumor.3,4 The pathophysiological mechanism of this association is unknown, and is probably the result of chance. There have also been reports of concomitant autoimmune thyroid disease and thyrotropinoma, but this is also an uncommon association.5
As noted above, THS-secreting pituitary tumors, or thyrotropinomas, are the most uncommon adenohypophysial tumors. Thyrotropic cells account for less than 5% of all hypophysial cells, hence the low frequency of these tumors.6 They are benign tumors, and only one case of conversion of a benign thyrotropinoma into a carcinoma with multiple metastases and loss of subunit α has been reported.7 They are usually big tumors, and only 10% are microadenomas.8
They may occur at any age, but are more common in middle age, and are not more prevalent in females, as occurs with most endocrine diseases.
Most thyrotropinomas only secrete TSH. Twenty-five percent are characterized by a concomitant secretion of other pituitary hormones, mainly growth hormone (GH, 15%) and prolactin (10%),9 because they share common transcription factors such as PROP-1, Pit-1, and HESX-1.10,11 Our patient had positive immunohistochemistry for TSH, prolactin, and GH, but functionally only had increased TSH.
The molecular mechanisms which lead to the formation of these tumors are unknown, but may include somatic point mutations or abnormal oncogene expression, as occurs in other tumors.
Clinically, patients with TSH-secreting tumors have symptoms related to either tumor size (visual disturbances, headache) or to hormone hyperproduction. Palpitations, nervousness, or goiter commonly occur. Many patients have a long history of thyroid dysfunction, diagnosed as Graves’ disease, and up to 30% of patients eventually undergo thyroidectomy or radioiodine ablation.12
Diagnosis is sometimes delayed when concomitant autoimmune hypothyroidism exists, and it is inadequate TSH suppression with replacement therapy that should lead us to suspect autonomous TSH production. Gonadal axis disorders, including menstrual changes and decreased libido, are common.
Approximately two-thirds of patients with thyrotropinoma have goiter with increased radioiodine uptake.
The main finding in central hyperthyroidism is the presence of elevated circulating thyroid hormone levels with high or inappropriately normal THS levels.
Eighty percent of patients with TSH-secreting tumors have hyperproduction of subunit α and a subunit α/TSH molar ratio higher than 1. However, it has recently been reported that subunit α is normal in up to 60% of microadenomas.2
Stimulation with TRH allows for differentiating TSH hyperproduction by a secreting tumor from thyroid hormone resistance. Upon stimulation, TSH response is decreased in tumors, while in healthy people and patients with THR, TSH increases in response to TRH. Measurement of subunit α during the test is usually helpful, because the molar ratio of subunit α to TSH is elevated in patients with tumors. Our patient had a TRH test consistent with a TSH-secreting tumor.
A test with T3 (80–100μg/day for 8–10 days) may also be performed. In this test, complete TSH inhibition does not occur in patients with thyrotropinoma.1
MRI is the most sensitive imaging test for detecting the pituitary tumor.
The treatment of choice for these tumors is surgery, performed after the normalization of thyroid function to prevent surgical complications. Preoperative treatment of hyperthyroidism with short- or long-acting somatostatin analogs is currently accepted. This normalizes thyroid hormone levels in 75–95% of cases, and decreases tumor size by up to 50%.13 Somatostatin analogs inhibit TSH pulse amplitude and may suppress TRH. Antithyroid drugs should not be used because they may increase TSH levels and stimulate tumor growth. In our patient, the suppression of TSH levels and the normalization of peripheral hormones were achieved after four days of treatment.
Radiotherapy has been used as an adjuvant to surgery when the latter does not achieve cure.
Surgery is curative in 40% of patients, and the remission rate after radiotherapy is low. Therefore, somatostatin analogs are also the treatment of choice when surgery and radiotherapy fail.
Subacute thyroiditis usually has a viral origin and causes pain of a sudden or gradual onset in the thyroid area, together with occasional fever. Apoptosis of follicular epithelium occurs, resulting in the release of thyroglobulin and peripheral hormones, which causes the clinical signs of hyperthyroidism. At this time, radioiodine uptake is low, and hormone synthesis ceases. At subsequent disease stages, when preformed hormone deposits are already depleted, serum T3 and T4 levels decrease, sometimes to the hypothyroid range, and TSH increases. When the condition has been resolved, hormone levels almost always return to normal values, as occurred in our patient.
Please cite this article as: Guijarro de Armas MG, et al. Tirotoxicosis por tiroiditis subaguda en el postoperatorio de un tirotropinoma. Endocrinol Nutr. 2012;59:270–3.