The renin–angiotensin system (RAS) is one of the most complex hormonal regulatory systems, involving several organs that interact to regulate multiple body functions. The study of this system initially focused on investigating its role in the regulation of both cardiovascular function and related pathologies. From this approach, pharmacological strategies were developed for the treatment of cardiovascular diseases. However, new findings in recent decades have suggested that the RAS is much more complex and comprises two subsystems, the classic RAS and an alternative RAS, with antagonistic effects that are usually in equilibrium. The classic system is involved in pathologies where inflammatory, hypertrophic and fibrotic phenomena are common and is related to the development of chronic diseases that affect various body systems. This understanding has been reinforced by the evidence that local renin–angiotensin systems exist in many tissue types and by the role of the RAS in the spread and severity of COVID-19 infection, where it was discovered that viral entry into cells of the respiratory system is accomplished through binding to angiotensin-converting enzyme 2, which is present in the alveolar epithelium and is overexpressed in patients with chronic cardiometabolic diseases. In this narrative review, preclinical and clinical aspects of the RAS are presented and topics for future research are discussed some aspects are raised that should be clarified in the future and that call for further investigation of this system.
El sistema renina angiotensina es uno de los sistemas de regulación hormonal más complejos, pues participan varios órganos qué interactúan entre sí para regular múltiples funciones corporales. En un inicio el estudio de este sistema se enfocó en investigar su papel en la regulación, tanto de la función cardiovascular como de las enfermedades relacionadas. A partir de este enfoque se desarrollaron estrategias farmacológicas para el tratamiento de enfermedades cardiovasculares. Sin embargo, en las últimas décadas y con nuevos hallazgos se ha planteado que el sistema renina angiotensina es un sistema mucho más complejo constituido por 2 subsistemas, uno clásico y otro alternativo que tienen efectos antagónicos, normalmente en equilibrio. El predominio del sistema clásico está involucrado en enfermedades donde los fenómenos inflamatorios, hipertróficos y fibróticos son comunes y se relacionan con el desarrollo de enfermedades crónicas que afectan diversos sistemas. Esto se ha reforzado por la evidencia de que existen sistemas renina angiotensina locales en muchos tejidos, y por el papel del sistema renina angiotensina en la propagación y severidad de la infección por la COVID-19, en donde se descubrió que el ingreso del virus en el sistema respiratorio se realiza a través de la enzima convertidora de angiotensina 2, presente en el epitelio alveolar y que se sobreexpresa en pacientes con enfermedades cardiometabólicas crónicas. En la presente revisión narrativa se presentan aspectos preclínicos y clínicos de ese sistema y se plantean algunos aspectos que se deben aclarar en el futuro y que demandarán más investigación de este sistema.
Renin was first identified more than a century ago, and since then the renin–angiotensin system (RAS) has been studied for its role in regulating blood pressure. Various elements and processes that are key in both cellular and systemic physiology have been incorporated, which has led to the suggestion that the RAS plays a role in pathologies that affect multiple organs and systems. The current work provides an overview of the RAS, covering preclinical and clinical aspects and raising some perspectives regarding the pathologies in which the RAS is involved. For this purpose, a review was carried out in the PubMed, Google Scholar, and Scopus databases using combinations of the keywords renin, angiotensin, ACE, angiotensin receptors, renin–angiotensin system, physiopathology and pharmacology.
The renin–angiotensin systemFor many years, the study of the renin–angiotensin system focused on the kidney–heart axis. The kidney and heart form an axis whose function is to ensure tissue perfusion and body homeostasis; for this, the cardiovascular system provides blood circulation and the kidney maintains the blood volume and the amount of water in the body.1
This cardiorenal interaction requires very fine regulatory mechanisms, one of which is the RAS, which involves a cascade of enzymatic reactions that result in a final effector, angiotensin II. This molecule works by modulating vascular tone and body water volume. However, accumulating evidence has identified the presence of an RAS in different tissues, including muscle, nervous system, bone, gonadal, gastrointestinal, immune, adipose, pancreatic, liver and circulatory tissue.2
The RAS includes renin, which is produced by the juxtaglomerular apparatus in the kidney, and a liver protein called angiotensinogen, which is a target of enzymatic reactions that results in angiotensin II. Angiotensin II acts at several levels to ensure the regulation of cardiovascular and renal function and body homeostasis.
ReninRenin (R) is an acid protease synthesised and stored by secretory vesicles in the juxtaglomerular cells of the dense renal macula. It is initially synthesised as a proenzyme, prorenin (PR), a 406 amino acid (aa) protein, which is cleaved and released into circulation as active renin (340aa). A small proportion is released as prorenin; the renin/prorenin ratio in the plasma of healthy people is 10. This R/PR ratio is similar in individuals with primary high blood pressure (HBP) but increases to 40–50 in individuals with type 2 diabetes mellitus (DM2). The action of plasma renin is known as plasma renin activity and is measured through the concentration of angiotensin I that appears in plasma per unit of time (ng/ml/h). Stimuli for renin secretion include decreased blood pressure, sympathetic stimulus and humoral factors, such as angiotensin II, endothelins, prostaglandins, histamine, vasopressin, nitric oxide and dopamine.3–5
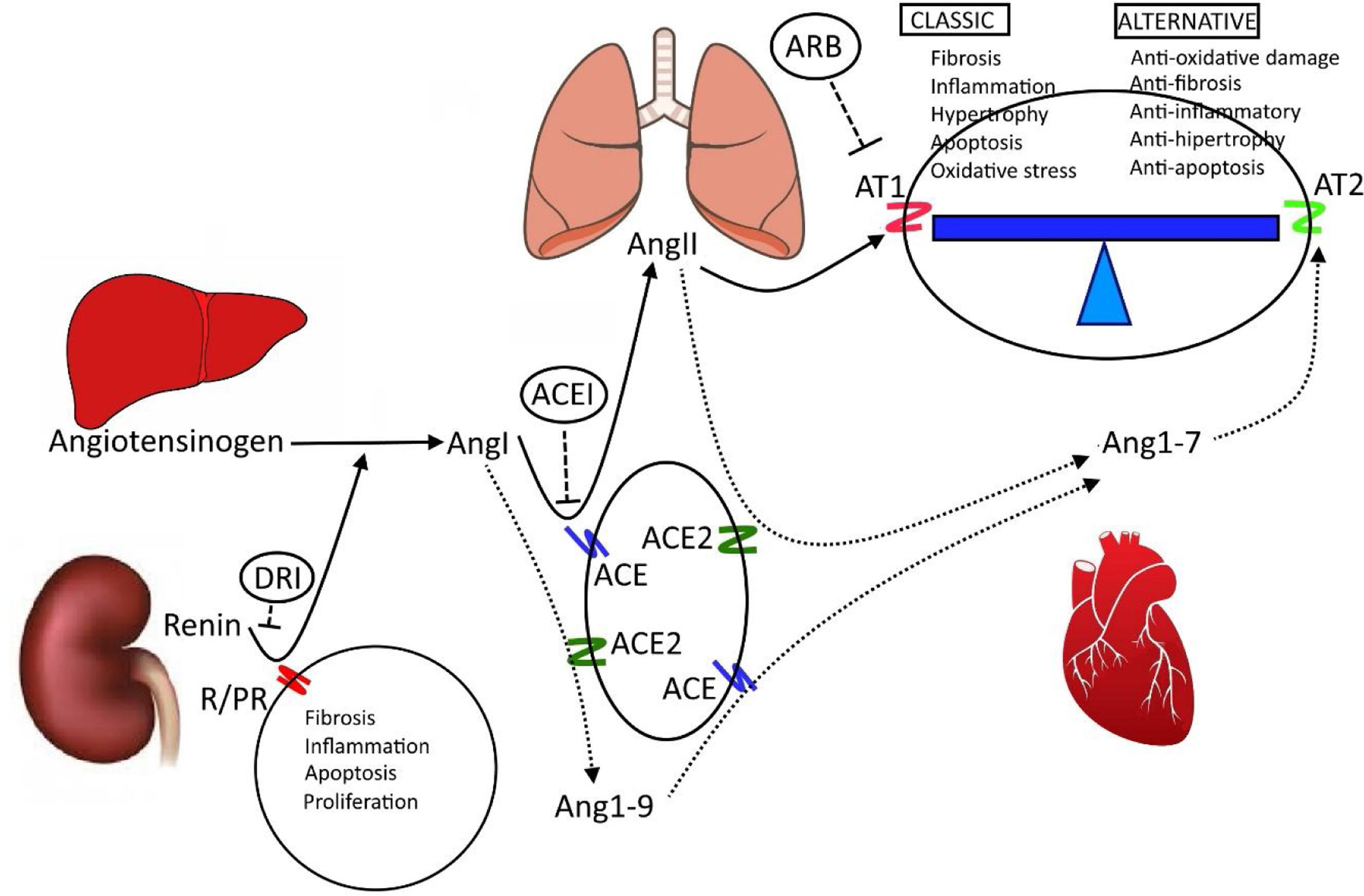
AngiotensinAngiotensin II (AngII) originates from preangiotensinogen, a 485aa liver-synthesised precursor protein, which by enzymatic action is transformed into angiotensinogen, a 452aa protein that is then secreted into the circulation.6 Glucocorticoids, oestrogens, thyroid hormones, cytokines and AngII stimulate the secretion of angiotensinogen. Once in circulation, angiotensinogen interacts with R to generate the peptide angiotensin I (AngI), a 10 aa peptide. The interaction of AngI with angiotensin-converting enzyme (ACE) gives rise to AngII, an active 8 aa peptide that targets angiotensin receptors 1 and 2 (AT1 and AT2). AngII has a half-life of one to two minutes and is degraded by peptides. The effect of AngII is derived from its action mainly on AT1 receptors present in vascular smooth muscle and other tissues. Effects include vasoconstriction, inflammation, fibrosis and cell proliferation.2,7,8 However, other metabolites produced by the action of ACE2 and other peptidases have also been described (Table 1). ACE2 promotes the conversion of AngI to angiotensin 1-9 (Ang1-9) and the conversion from AngII to angiotensin 1-7 (Ang1-7). Similarly, the conversion from Ang1-9 to Ang1-7 is carried out by ACE and neprilysin (membrane metalloendopeptidase, MME). ACE also converts Ang1-7 to Ang1-5. Many of these angiotensin molecules are expressed in parallel to AngII in the heart, kidney and testis, while there is lower expression in the colon, small intestine and ovaries.2,9–13 Other angiotensin molecules include Ang2-10, derived from AngI and produced by the action of aminopeptidases. Ang2-10 gives rise to Ang2-8 (also called AngIII) and is produced through the action of ACE. Ang3-8 (or AngIV) is produced from AngIII through the action of aminopeptidase (Table 1). Another angiotensin is Ang3-7, which is derived from AngIV. AngII gives rise to AngA, which in turn gives rise to the metabolite alamandine.9,14–17
Classic renin angiotensin system vs alternative renin angiotensin system.
| Molecules | Receptors | Distribution | Effect | Pathology | Pharmacology |
|---|---|---|---|---|---|
| Classic renin angiotensin system | |||||
| Prorenin, reninangiotensinogenAngiotensin 1Angiotensin 2 | R/PRACEAT1 | Kidney, heart, vessels, pituitary gland, adrenal gland, brainLiver, plasmaLung, Gonads, Brain, Intestine, Fibroblasts | Inflammation cell proliferation apoptosisVasoconstrictionOxidative stressFibrosisInsulin resistanceCardiopulmonary dysfunction | Hypertension, hypertensive heart disease, hypertensive nephropathy, diabetes mellitus 2Heart failure | Clinical use: Renin antagonistsACE inhibitorsAT1 receptor blockersExperimental use: Renin agonistsAng II receptor agonists |
| Alternative renin angiotensin system | |||||
| Prorenin, reninangiotensinogenAngiotensin 1Angiotensin 1-7Angiotensin 1-9Angiotensin 2-10Angiotensin 2-8Angiotensin 3-8Angiotensin 3-7Angiotensin AAlamandine | R/PRACEACE2AT2MASMrgDAT4 | Kidney, heart, vessels, pituitary gland, adrenal gland, brain, liver, plasma,endothelium | VasodilationAnti-inflammatoryAnti-proliferativeAntifibroticAnti-apoptotic | All pathologies related to classic RAS involve imbalance with decreased action of alternative RAS | Experimental use:Analogue agonists from Ang1-7, Ang1-9AVE0991CGEN-856S |
The general characteristics of both systems are displayed. R/PR. Renin receptor; AT1–AT2–AT4: angiotensin receptor 1, 2 and 4; MAS: MAS Receptor; MrgD: MrgD Receptor. Ang1-7–Ang1-9: angiotensin 1-7 and 1-9.
In addition to renin, another key enzyme in the RAS is angiotensin-converting enzyme (ACE), a peptidase that transforms AngI into AngII. This enzyme has two subtypes, ACE and ACE2, which are expressed in different tissues. ACE converts AngI into AngII in the pulmonary tissue. Other aminopeptidases, such as cathepsins, also favour this process.13 ACE also interacts with the bradykinin system to degrade bradykinin into inactive peptides.
ACE is a transmembrane protein. Two ACE subtypes have been identified—one expressed in somatic cells and one in germ cells. ACE is expressed in many epithelial cells, especially in lung tissue, renal epithelium cells, adrenal glands, the small intestine and the epididymis. It is coupled to the Gq/11 protein and activates phospholipase C (PLC) by raising the intracellular calcium level and activating protein kinase C (PKC). ACE inhibits adenylate cyclase (AC) and activates tyrosine kinase.11
ACE2 is a membrane protein with a single transmembrane segment, an intracellular segment, and terminal N and C domains, but with a single enzymatic active site that gives it characteristics different from those of ACE. It is located in kidney, intestine, heart, testis and retina tissue. Rodent models that do not express ACE2 show corneal hyperkeratosis that reverses with losartan treatment.18,19
Renin–angiotensin system receptorsThere are specific receptors for each of the RAS molecules (renin, angiotensin), These are described below.
Renin receptorAt the tissue level there is the renin/prorenin receptor, which is activated by renin and renal and extrarenal prorenin. The cleavage of prorenin generates active renin, which can then generate AngI. However, the binding of R and PR to the renin receptor activates intracellular MAPK signalling pathways related to the production of pro-inflammatory cytokines and cell differentiation processes.3
Angiotensin receptors 1 and 2The angiotensin receptors are transmembrane proteins with seven transmembrane domains. Two subtypes have been described: AT1 and AT2. AT1 is a G protein-coupled receptor that increases the intracellular calcium concentration. There are two subtypes of AT1: subtype A, present in the brain, and subtype B, present in the adenohypophysis and cerebral cortex. The genes that encode for this receptor are located on chromosome 3. The AT1 receptor is the primary target of AngII, but AT1 also binds AngIII and Ang A. AT2 is a G protein-coupled receptor that activates various phosphatases, which eventually activate membrane potassium channels. Additionally, AT2 activation triggers the production of nitric oxide and GMPc. AT2 binds Ang1-7, Ang1-9, AngII, Ang A and AngIII. This receptor is present in foetal tissues, neonatal tissues and adult brain tissue. The gene encoding AT2 is located on the X chromosome.12 Another related receptor is AT4, which binds AngIV and Ang3-7 (Table 1). In animal models, AT4 expression has been studied in the cerebral cortex, where it is involved in cognitive processes, but it has also been identified in other tissues including kidney, adrenal cortex, lung and heart. Functions include vasodilation and glucose uptake modulation.14,20
MAS receptorsMAS receptors are transmembrane proteins encoded by the proto-oncogene MAS1 and are coupled to G proteins. This proto-oncogene induces tumours in animal models. In the RAS, MAS receptor activation decreases the sympathetic tone, blood pressure, chronic hypertension, and fibrosis. By contrast, activation increases parasympathetic tone, baroreflex, vasodilation, nitric oxide production and natriuresis. Ang1-7 is the natural ligand for the MAS receptors. A subtype of the MAS receptor is the MrgD receptor, which shows affinity for alamandine.10,15,16,21
Classic RAS vs alternative RASThe RAS is an endocrine axis that includes elements of many origins such as renin, angiotensinogen, ACE, renin receptors and angiotensin (Fig. 1, Table 1).
Renin–angiotensin system. The image illustrates the main organs and components of the renin–angiotensin system. Similarly, it illustrates the elements that make up the classic RAS and the alternative RAS, which are in balance. Cardiopulmonary and renal pathologies are caused by a greater influence of the classic RAS. R/PR. Renin receptor prorenin; ACE–ACE2: angiotensin-converting enzyme and angiotensin-converting enzyme 2; AngI–AngII–AngI 1-7–Ang1-9: angiotensin I, II, 1-7 and 1-9; AT1–AT2: angiotensin receptor 1 and 2; ML: smooth muscle. ACEI: angiotensin-converting enzyme inhibitors; ARB: angiotensin receptor blockers; DRI: direct renin inhibitors. Author: own source.
Renin, ACE, AngII and its AT1 receptor have been studied for decades. These molecules are involved in regulating cardiovascular function, and they participate in the development of some chronic pathologies of renal and heart origin, such as high blood pressure and hypertensive heart disease. Together, these molecules make up what is called the classic RAS system, also called the ACE/Ang II axis/AT1 receptor.11,13,22 The general action of this subsystem has a vasopressor effect, with increased peripheral vascular resistance and water and sodium retention caused by stimulating the release of aldosterone into the adrenal cortex (Fig. 1, Table 1). The alternative RAS consists of some elements common to the classic RAS along with others that counter-regulate the actions of the classic RAS (Fig. 1, Table 1). The common elements are renin, angiotensinogen, AngI and ACE. The action of angiotensin-converting enzyme 2 (ACE2) generates a series of molecules with individual activities (Ang1-9, Ang1-5 and Ang1-7); Ang1-7 constitutes the final molecule in this series and acts on the MAS receptor. For this reason, this alternative system is called the ACE2/Ang1-7/MAS receptor axis.7,9,12,15,21,23,24
Local renin–angiotensin systemsIn addition to the endocrine action of the RAS, several studies have shown paracrine and autocrine effects that depend on local actions in many organs and tissues, including heart, kidney, lung, muscle, central nervous system, blood vessel, pituitary gland, adrenal gland, liver, immune system, erythrocytes, digestive tract and adipose tissue. Similarly, an intracellular RAS is proposed, with AT1 receptors present in the mitochondrial membrane. The presence of a RAS in each tissue gives rise to their specific processes.
Adipose tissueIn adipose tissue, AngII increases inflammatory processes, increases the mass of adipose tissue, alters lipogenesis and lipolysis, decreases insulin sensitivity and increases glucose uptake.25–27
Skeletal muscleIn skeletal muscle, AngII decreases blood flow, decreases insulin-stimulated glucose uptake and alters insulin signalling. In contrast, angiotensin 1-7 increases the insulin-stimulated glucose uptake and improves average insulin signalling.28
PancreasIn the pancreas, AngII produces a reduction in blood flow, diminishes insulin secretion and increases oxidative stress, leading to increased inflammation. As a counterpart, angiotensin 1-7 decreases inflammation and apoptosis of the pancreatic islets.29,30
LiverIn the liver, the RAS reduces insulin sensitivity and increases fibrosis. There is experimental evidence of the relationship between RAS and non-alcoholic fatty liver. The expression of AngII modifies the insulin receptor, inducing resistance. In addition, activated AT1 favours the production of reactive oxygen species (ROS) and pro-inflammatory cytokines and promotes fibroblast differentiation.31 At the clinical level, retrospective studies have shown a possible positive association between age, diabetes and the likelihood of developing non-alcohol fatty liver disease, while there is a negative relationship between treatment with statins and/or ARBs and the development of liver fibrosis.32
Blood vesselsThe action of the RAS in blood vessels has been studied experimentally in the retina, where ACE, renin and AT receptors are present. The activation of AT1 generates vasoconstriction of the retinal vessels and increases ROS, decreases endothelial nitric oxide (NO), and increases endothelial dysfunction. AT2 activation produces vasodilation and has anti-inflammatory effects.33
HeartIn the heart, the activation of AT1 by AngII increases ROS, hypertrophy, inflammation, metabolic changes and alters conductance. AT2 activation generates the opposite effects.2,7,22,24,34
KidneyThe presence of a local RAS has been found in the kidney. AT1 activation increases ROS production, hypertrophy and inflammation. Therefore, a relationship between intrarenal RAS, HBP and renal parenchyma damage has been suggested.1,22,35
LungIn animal models, a relationship has been identified between ACE, AT1 receptors and inflammation, vascular remodelling, and endothelial dysfunction. These damaging processes can lead to cardiopulmonary dysfunction. In these cases, pretreatment with captopril or losartan decreases the pro-inflammatory effect.36,37 ACE2 is expressed in type II pneumocytes and has a protective anti-inflammatory role.23,38
Bone marrowThere is experimental evidence for the presence of a RAS in bone marrow. AT1 receptors are expressed in haematopoietic cells, and their stimulation promotes differentiation of erythroid cells, a phenomenon that is blocked by losartan. In pathological conditions, high levels of ACE have been found in patients with leukaemia.39,40
Nervous systemThere is evidence for the expression of an RAS in neurons and glia due to the presence of ACE and the AT1, AT2 and MAS receptors. AT2 and MAS activation decreases the production of inflammatory factors and increases the levels of BDNF and proteins that facilitate phagocytosis. AT1 and MAS activation promotes inflammation, cell death, demyelination and alterations in cellular communication, which modifies plasticity and alters the development of cognitive processes.41,43 Microglia activation induces cytokine elevation and neuroinflammation. Additionally, preclinical studies have shown a neuroprotective effect of AT2 agonists after a cerebrovascular accident, and it has been suggested that ECAI or ARBs could help to minimise neuronal damage.21,44 The endogenous agonists of these receptors and their origins remain unknown.
Cellular renin–angiotensin systemSeveral studies have highlighted the presence of AT1 in the mitochondrial membrane. The presence of this receptor can alter the function of the mitochondrial oxidative chain, which can lead to the production of free radicals and oxidative stress that in turn causes mitochondrial damage. Likewise, excess free radicals can lead to cell destruction. This oxidative phenomenon can be present in different tissues. In the nervous system, inflammatory phenomena in neurons and glia might explain the cell destruction present in some neurodegenerative diseases. Changes related to corneal inflammation resulting in hyperkeratosis and retinopathy have also been reported in preclinical trials.33 This inflammatory phenomenon is common to many tissues and explains the presence of fibrotic processes in the cardiac, pulmonary, liver and renal systems.27,32,34
RAS in the physiopathology of chronic diseasesGiven the wide distribution of the RAS, it has been investigated for its role not only in cardiovascular diseases but also in other pathologies including metabolic, respiratory and neurological diseases, among others. Much of the evidence comes from preclinical studies where, through experimental genetic, pharmacological and experimental surgery techniques, relationships between the RAS and numerous pathologies have been identified.
RAS and cardiometabolic risk factorsChronic diseases of cardiometabolic origin represent an important group in the global disease burden. Therefore, special attention has been given to the role of the RAS in cardiometabolic risk factors.
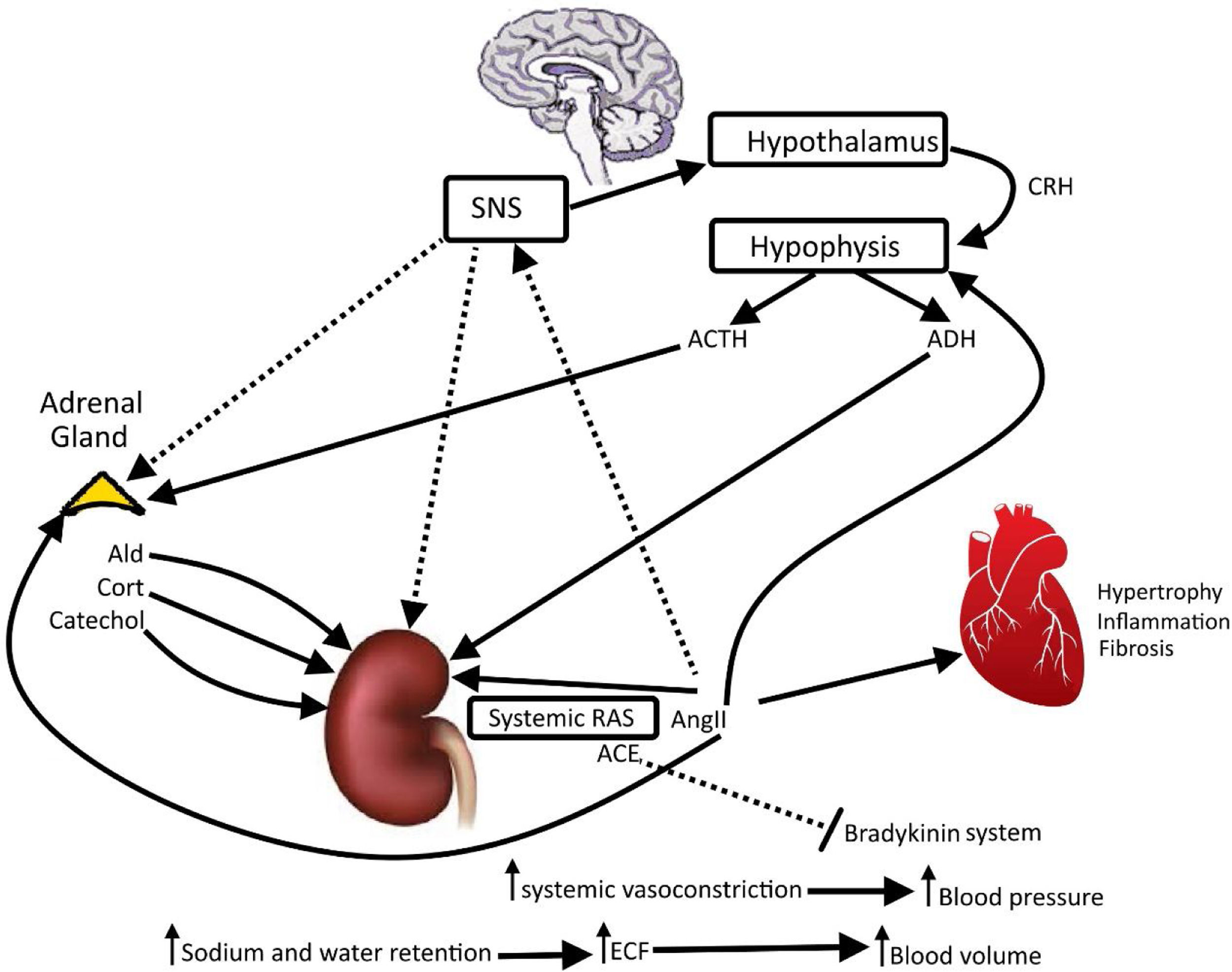
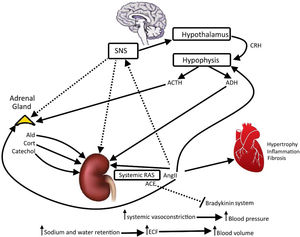
High blood pressureThe activation of AT1 receptors increases the activity of the sympathetic nervous system (Fig. 2). It increases the blood pressure, vasoconstriction, ADH secretion and aldosterone secretion, and it generates cardiac hypertrophy, fibrosis, inflammation and ROS production (Figs. 1 and 2). In parallel, AT1 activation decreases parasympathetic tone, baroreflex sensitivity, NO production and natriuresis (Fig. 2).24,45 Opposing effects are seen when AT2 is activated, which causes a decrease in blood pressure, fibrosis and inflammation. AT2 activity increases vasodilation, NO production, baroreflex sensitivity, cardioprotection and natriuresis.7,34 Additionally, the activation of AT4 increases renal blood flow at the renal cortex and increases vasodilation, NO production and natriuresis. It is also cardioprotective and decreases inflammatory processes. MAS receptors decrease sympathetic tone, blood pressure, chronic hypertension and fibrosis, and increase parasympathetic tone, baroreflex, vasodilation, NO production and natriuresis.1,8
Renin–angiotensin system, adrenal gland, sympathetic nervous system (SNS), hypothalamus and hypophysis. The image illustrates the positive interactions among systemic renin angiotensin system and vital organs. There are feedbacks positives (displayed) and negatives (not displayed) to maintain homeostasis, body water and osmolarity in equilibrium. Thick arrows indicate main regulator. CRH: corticotropin-releasing hormon; ACTH: adrenocorticotropic hormone; ADH: antidiuretic hormone; Ald: aldosterone; Catechol: catecholamines; Cort: cortisol; AngII: angiotensin II; systemic RAS: systemic renin angiotensin system; ACE: angiotensin-converting enzyme; ECF: extracellular fluid. Author: Own source.
Adipose tissue produces pro-inflammatory cytokines and metabolites that are involved in the control of energy metabolism, body weight, glycaemic homeostasis and lipid metabolism.46,47 Adipose tissue has been shown to express RAS components, AngII, ACE and AT1 (i.e. the Ang II/ACE/AT1 axis), which is hyperactive in metabolic diseases. AngII induces lipogenesis and reduces lipolysis; these processes are related to obesity, insulin resistance and inflammation. This axis is opposed by the alternative RAS (ACE2/Ang1-7/MASR axis), which is also expressed in adipose tissue. Alternative RAS activation induces metabolic actions including increased lipolysis, reduced body weight, an improved lipid profile, the attenuation of metabolic syndrome, increased glucose absorption and a reduction of oxidative stress. Due to these protective responses, the ACE2/Ang1-7/MAS axis counteracts the negative effect of the ACE/Ang II/AT1 axis and has been proposed as a target to reduce obesity and DM2. In diet-induced obesity animal models, a decrease in SREBPs (sterol binding proteins) that are associated with lipogenesis, adipogenesis and cholesterol homeostasis helps to prevent lipotoxicity. In ACE2-knockout models, SREBPs are increased and lipogenesis is enhanced in the liver48 and skeletal muscle,49 supporting the hypothesis for the role of ACE2/angiotensin 1-7 in lipid metabolism.19
AtherosclerosisThe effect of the RAS may be anti-atherogenic or pro-atherogenic, and the key elements in this system are AngII, ACE2 and Ang1-7. Depending on which system predominates and whether there is inflammation, the effect will be pro-atherogenic (derived from the activation of the classic RAS) or anti-atherogenic (derived from the anti-inflammatory action of the alternative RAS).7–9,24,33
Fatty liverThe presence of a hepatic RAS allows the supposition that its activation accompanies the development of non-alcoholic fatty liver disease. AT1 activation generates local changes that induce lipid accumulation and inflammatory phenomena that trigger a liver fibrotic process.31,32,35
SmokingIn animal models, nicotine exposure has been shown to induce HBP and cardiac changes including hypertrophy and cardiac fibrosis.50,51 Related studies have not been conclusive in humans, but it is proposed that nicotine can induce an imbalance between the classic RAS and the alternative RAS that increases the likelihood of developing chronic cardiopulmonary disease.52
Type 2 diabetes mellitusThe mechanisms through which the RAS participates in the development of DM2 are unclear. Preclinical studies have highlighted the concurrency of various phenomena. AT1 has been described to activate extracellular matrix proteases, which stimulate the release of epidermal growth factor (EGF). When EGF interacts with its receptor at the peripheral tissue level, it activates a common intracellular pathway that is also activated by AT1. This pathway includes ERK 1/2 and mTOR/S6K-1 signalling, which phosphorylates insulin-sensitive receptors, resulting in insulin resistance. AT1 activation also affects the pancreas, which decreases pancreatic infusion and increases oxidative stress in pancreatic beta cells, with a consequent reduction in insulin production. These effects are reduced through the administration of ACEI or ARBs. AT1 activation is accompanied by the production of pro-inflammatory adipokines that enhance the effect of the classic RAS.26–28,53
Kidney fibrosisThe presence of a RAS in the renal parenchyma can also determine kidney function or kidney damage based on the balance between the classic RAS and the alternative RAS. The latter system is related to anti-inflammatory, vasodilator and antifibrotic processes that are opposed to the classic RAS, which favours phenomena related to vasoconstriction, inflammation and fibrosis. The predominance of the classic system at the renal level would explain the fibrotic damage that can lead to chronic renal failure.1,35,54
Other pathologiesThe viral S (spike) protein of the SARS-CoV-2 virus (the virus that causes COVID-19) binds to ACE2 as its receptor for host cell entry.55 This has revealed the participation of the RAS in hyperinflammation processes, a phenomenon that can be a common result in diseases as diverse as digestive,56 pulmonary,36,38 haematological,57,58 hypothalamic,41,42 neurodegenerative,43,44 bone,40 reproductive,59 immunological,60,61 obstetric62 and cancer diseases.63,64 This further broadens the RAS field and the research on its role in chronic diseases.
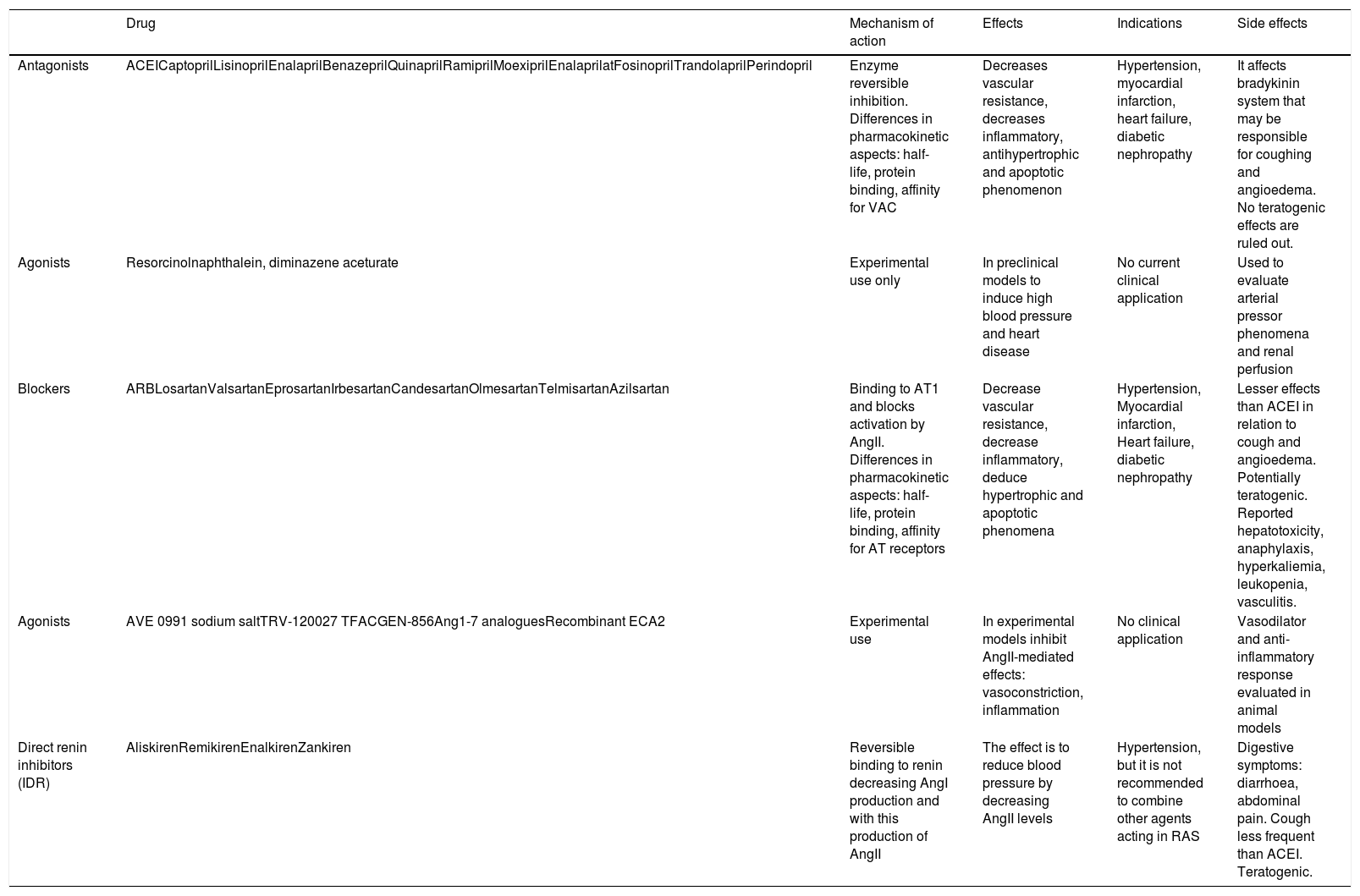
RAS and therapeutic strategiesGiven the relationship between RAS and the development of cardiovascular diseases with essential hypertension blood pressure, congestive heart failure and renal insufficiency, possible therapeutic alternatives have been raised, and one of the first targets was ACE (Fig. 1, Table 2). ACE inhibitors (ACEIs), such as captopril, enalapril, lisinopril and fosinopril, first appeared more than 50 years ago. These progressively became incorporated into clinical practice as an alternative to traditional antihypertensive agents, and today ACE inhibitors are considered to be the first line of treatment. With the widespread use of ACEIs, side effects have been seen in phase IV pharmacological studies; thus, drugs that selectively act on the RAS axis have been identified. As a result, AT receptor blockers (ARBs) were incorporated into clinical practice nearly 30 years ago. These ARBs (e.g. losartan, irbesartan and valsartan) expanded the therapeutic options (Fig. 1, Table 2). In addition, direct renin inhibitors (DRIs) (e.g. aliskiren, remikiren and enalkiren) have been considered as an alternative to intervene in pathologies that involve elevated renin levels.1,37,45,65 Pharmacological interventions have been aimed at blocking the actions of the classic RAS. However, with the discovery of the alternative RAS, scientists began working on alternatives to stimulate this system, with little success until recently. In preclinical studies, alternative RAS agonists have been evaluated for their preventive or protective effects due to their potential anti-inflammatory, antiproliferative and antifibrotic actions, but the results have been inconclusive. These substances have been employed to study the RAS in preclinical studies in animal models with induced pathologies.21 Owing to the recent COVID-19 pandemic, novel therapeutics have been proposed. One of these is recombinant human soluble ACE2, which would bind to the virus and prevent its penetration into cells. It would also activate the alternative RAS, thereby increasing the production of Ang1-7.66
Renin angiotensin system and the main therapeutic strategies developed and used in both the experimental field and the clinical area.
| Drug | Mechanism of action | Effects | Indications | Side effects | |
|---|---|---|---|---|---|
| Antagonists | ACEICaptoprilLisinoprilEnalaprilBenazeprilQuinaprilRamiprilMoexiprilEnalaprilatFosinoprilTrandolaprilPerindopril | Enzyme reversible inhibition. Differences in pharmacokinetic aspects: half-life, protein binding, affinity for VAC | Decreases vascular resistance, decreases inflammatory, antihypertrophic and apoptotic phenomenon | Hypertension, myocardial infarction, heart failure, diabetic nephropathy | It affects bradykinin system that may be responsible for coughing and angioedema. No teratogenic effects are ruled out. |
| Agonists | Resorcinolnaphthalein, diminazene aceturate | Experimental use only | In preclinical models to induce high blood pressure and heart disease | No current clinical application | Used to evaluate arterial pressor phenomena and renal perfusion |
| Blockers | ARBLosartanValsartanEprosartanIrbesartanCandesartanOlmesartanTelmisartanAzilsartan | Binding to AT1 and blocks activation by AngII. Differences in pharmacokinetic aspects: half-life, protein binding, affinity for AT receptors | Decrease vascular resistance, decrease inflammatory, deduce hypertrophic and apoptotic phenomena | Hypertension, Myocardial infarction, Heart failure, diabetic nephropathy | Lesser effects than ACEI in relation to cough and angioedema. Potentially teratogenic. Reported hepatotoxicity, anaphylaxis, hyperkaliemia, leukopenia, vasculitis. |
| Agonists | AVE 0991 sodium saltTRV-120027 TFACGEN-856Ang1-7 analoguesRecombinant ECA2 | Experimental use | In experimental models inhibit AngII-mediated effects: vasoconstriction, inflammation | No clinical application | Vasodilator and anti-inflammatory response evaluated in animal models |
| Direct renin inhibitors (IDR) | AliskirenRemikirenEnalkirenZankiren | Reversible binding to renin decreasing AngI production and with this production of AngII | The effect is to reduce blood pressure by decreasing AngII levels | Hypertension, but it is not recommended to combine other agents acting in RAS | Digestive symptoms: diarrhoea, abdominal pain. Cough less frequent than ACEI. Teratogenic. |
The RAS and its role in body homeostasis has been studied for more than 100 years and has yet to be fully explored. The research conducted thus far has gradually provided information to explain the relationships among the RAS, health and the development of chronic pathologies. Multiple studies have highlighted the role of the RAS in various pathologies that were not previously associated with it, including metabolic, renal, cardiac, hepatic, digestive, endocrine, neurodegenerative, haematological, reproductive and muscular diseases. The relationship between the RAS and the respiratory infection caused by SARS-CoV was established nearly 20 years ago.18,67,68 This relationship has become more evident in the current COVID-19 pandemic, in which the severity of the clinical manifestations and complications are related to the presence of ACE2 and a hyperinflammatory response, which is a typical response of the classic RAS.67,69 Furthermore, it has been shown that individuals with comorbidities that involve an imbalance between the classic and alternative RAS (obesity, DM2, hypertension, hypertensive heart disease) are more likely to acquire the infection and are more likely to develop severe and fatal complications. There has been a debate about whether pharmacological interventions in the RAS prevent or promote the development of COVID-19 and its complications. This debate is open and requires extensive research to clarify the possible advantages and disadvantages of current therapeutics and to propose valid alternatives that reduce morbidity and mortality in cases where the RAS is involved.
Authors’ contributionRafael Vargas. Conception and design of research. Prepared and edited manuscript. Approved final version.
Jesus M. Varela. Drafted, revised and edited manuscript. Approved final version.
Esperanza Fajardo. Drafted, revised and edited manuscript. Approved final version.
Financial supportThis research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
Conflicts of interestNone.