Giant prolactinomas (tumor size larger than 40mm) are a rare entity of benign nature. Prolactinomas larger than 60mm are usually underrepresented in published studies and their clinical presentation, outcomes and management might be different from smaller giant prolactinomas.
Patients and methodsWe retrospective collected data from patients with prolactinomas larger than 60mm in maximum diameter and prolactin (PRL) serum levels higher than 21,200μIU/mL in our series of prolactinomas (283). Data were collected from January 2012 to December 2017. We included three patients with prolactinomas larger than 60mm.
ResultsAt diagnosis, two patients presented neurological symptoms and one nasal protrusion. All patients received medical treatment with dopamine agonists. No surgical procedure was performed. Median prolactin levels at diagnosis was 108,180 [52,594–514,984]μIU/mL. Medical treatment achieved a marked reduction (>99%) in prolactin levels in all cases. Tumor size reduction (higher than 33%) was observed in all cases. In one patient cerebrospinal fluid (CSF) leak was observed after tumor shrinkage.
ConclusionsDopamine agonists appear to be an effective and safe first-line treatment in prolactinomas larger than 60mm even in life-threatening situations. More studies with a higher number of patients are necessary to obtain enough data to make major recommendations.
Los prolactinomas gigantes (de tamaño superior a 40mm) son una entidad rara de naturaleza benigna. Los prolactinomas mayores de 60mm suelen estar infrarrepresentados en los estudios publicados, y su presentación clínica, resultados y tratamiento podrían ser diferentes de los de prolactinomas gigantes más pequeños.
Pacientes y métodosRecogimos retrospectivamente datos de pacientes con prolactinomas de más de 60mm de diámetro máximo y con concentraciones séricas de prolactina (PRL) superiores a 21.200μIU/ml de nuestra serie de prolactinomas (283). Los datos se recogieron entre enero de 2012 y diciembre de 2017. Se incluyeron 3 pacientes con prolactinomas mayores de 60mm.
ResultadosEn el momento del diagnóstico, 2 pacientes presentaban síntomas neurológicos, y uno protrusión nasal. Todos los pacientes recibieron tratamiento médico con agonistas dopaminérgicos. No se realizó ninguna intervención quirúrgica. La mediana de las concentraciones de PRL al diagnóstico fue de 108.180 (52.594-514.984)μIU/ml. El tratamiento médico logró una reducción notable (>99%) de los valores de prolactina en todos los casos. En todos los casos se observó una reducción del tamaño del tumor (superior al 33%). En un paciente se observó una fuga de líquido cefalorraquídeo (LCR) tras la reducción del tumor.
ConclusiónLos agonistas dopaminérgicos parecen ser un tratamiento de primera línea eficaz y seguro en los prolactinomas mayores de 60mm incluso en situaciones peligrosas para la vida. Se necesitan más estudios con un mayor número de pacientes para obtener datos suficientes para hacer recomendaciones importantes.
Prolactinomas are the most common functioning (hormone-secreting) pituitary tumor with an estimated prevalence of 3.5–5 cases per 10,000 individuals. Most prolactinomas are microadenomas (<10mm diameter), of benign nature and usually found in women. Macroprolactinomas (>10mm diameter), however, are more often found in men, but perhaps this is only due to delay in the diagnosis.1 Prolactinomas larger than 40mm in diameter are defined as giant prolactinomas and are rare, comprising only 0.5–4.4% of prolactinomas. Giant prolactinomas are more frequently observed in men (men to women ratio of 9:1) with a median age at diagnosis of around 40 years.2 The diagnosis of giant prolactinomas might be a considerable challenge. Their size and typical invasive features may result in diagnosis delay, misdiagnosis and/or unnecessary treatments. Given the rarity of giant prolactinomas, information about their clinical management is relatively limited but they seem to have mostly benign nature and rarely develop malignant PRL secreting carcinomas.1
Here, we focus our attention in a particular group of giant prolactinomas, those larger than 60mm in diameter, due to their diagnostic and therapeutic management challenges. Prolactinomas larger than 60mm are underrepresented in giant prolactinomas series. Their presentation includes massive infiltration which simulate highly aggressive tumors at presentation which can lead to aggressive treatments as surgery. To the best of our knowledge, the only series of these type of patients reported in the literature is that published by Shimon et al. in 2016. It was notified a higher surgery rate than in giant prolactinomas (50% vs 15%).1,3 It is our aim to report our experience in three cases with significative response to medical treatment despite their atypical presentation with neurological symptoms.
Patients and methodsRetrospective study of patients with giant prolactinomas larger than 60mm in maximum diameter and prolactin (PRL) serum levels higher than 21,200μIU/mL.
We identified three patients from a series of 283 prolactinomas diagnosed and followed in the Virgen del Rocío University Hospital between 01/01/2012 and 12/31/2017. The prolactinoma cohort was developed by a systematic research from digital medical records of pituitary tumors treated in our department. The study was approved by the IBiS-Virgen del Rocio Hospital Ethics Committee.
Serum PRL levels were measured using a commercially available electrochemiluminiscence immunoassay (Elecsys 2010; Roche) with a sensitivity of 1μIU/mL. The intermediate precision in 2.8–5% for PRL levels between 154 and 4945μIU/mL. Expected values for men are 86–324μIU/mL.
Patients’ treatment as well as follow-up visits were evaluated in a multidisciplinary clinical committee that included endocrinologists, neurosurgeons, and neuroradiologists.
Adenoma size and cavernous sinus invasion data were evaluated using magnetic resonance imaging at both diagnosis and during follow-up and maximum tumor diameter was considered. Following the Response Evaluation Criteria in Solid Tumors (RECIST) guidelines, a reduction of at least 30% in tumor diameter was considered partial response to treatment.
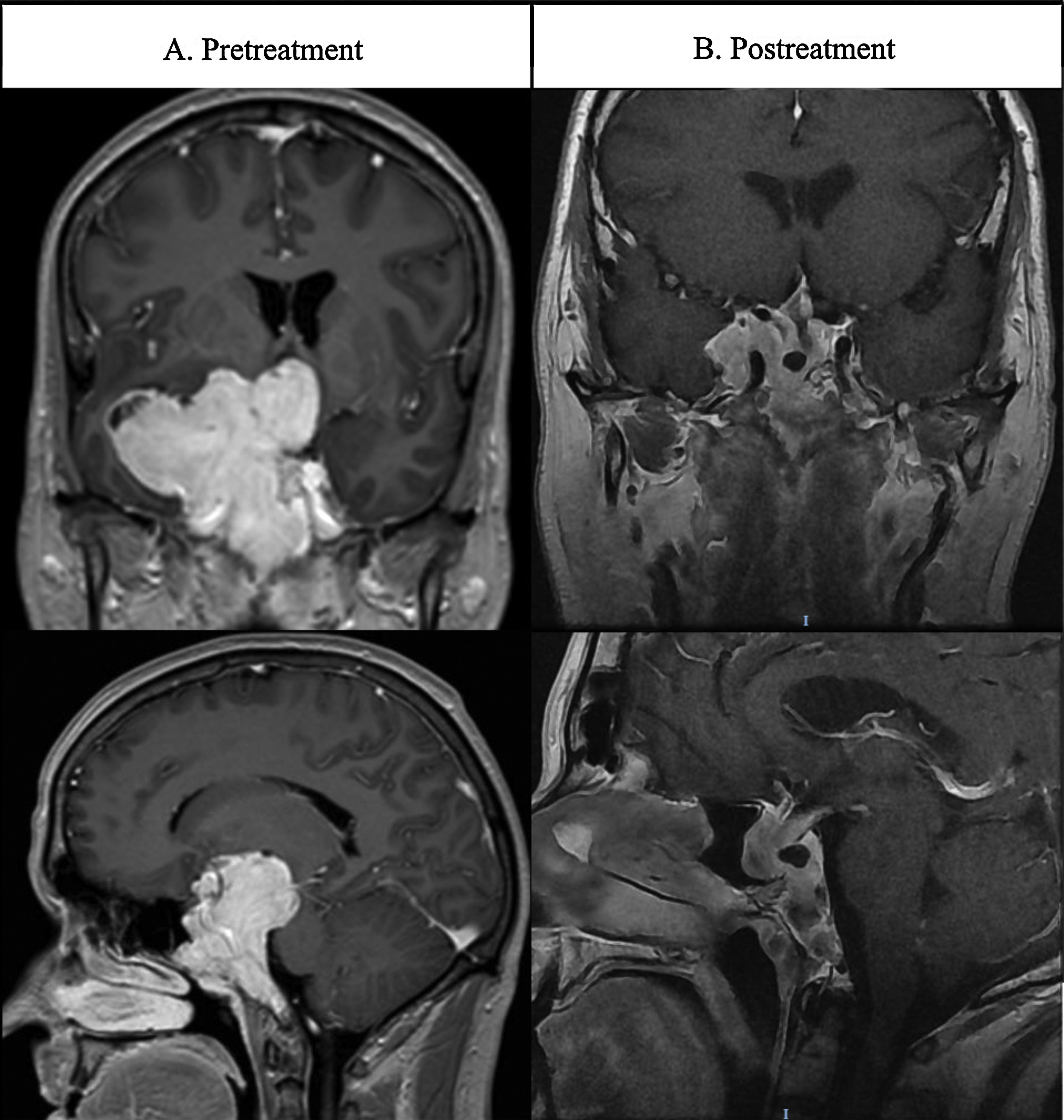
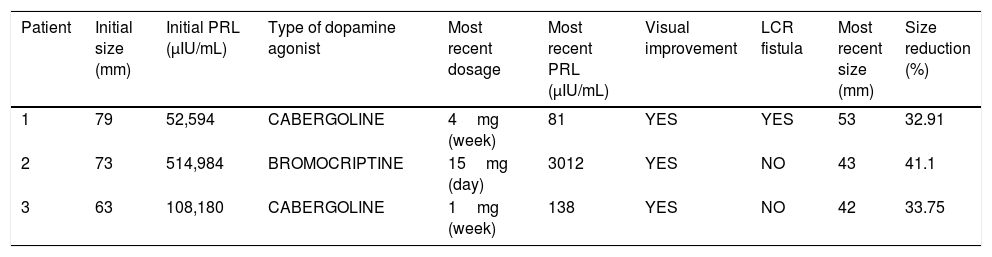
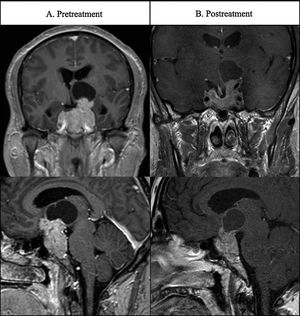
ResultsCase 1A 39-year-old man presented at the emergency room with absence seizures. He referred a medical history of erectile dysfunction, decreased libido and gradual loss of vision over the last 10 years. Cerebral magnetic resonance imaging (MRI) showed a solid mass with a maximal diameter of 80mm with cystic areas located in clivus with extension through sella, sphenoidal sinus, right temporal lobe, compromising ipsilateral optic nerve, mesencephalon and left lateral ventricle (Fig. 1A). Ophthalmological examination revealed bitemporal hemianopsia. Panhypopituitarism and very high serum prolactin levels (52,594μIU/mL) was observed in laboratory evaluation. A diagnosis of giant pituitary prolactinoma was made. Treatment with cabergoline was initiated gradually increasing each two weeks to 3mg/week. At discharge from hospitalization, the patient was under treatment with levetiracetam (1000mg twice a day), hydrocortisone (30mg a day, 2/3 in the morning and 1/3 in the afternoon), levothyroxine (50 mcg once a day). Twenty weeks later PRL levels normalized and visual defects greatly improved. Follow-up MRI at 24 weeks after treatment showed a substantial reduction of the tumor size (12.5%). The most recent (192 weeks after the diagnosis) MRI images (Fig. 1B) showed further reduction in tumor size (32.91%). After the reduction of tumor size, the patient developed a cerebrospinal fluid (CSF) leak which was exacerbated during Valsalva maneuvers. High β2 transferring levels (7.7mg/L; normal levels <1mg/L) in the fluid confirmed the CSF fistula. The patient remains under cabergoline treatment with no seizures (under antiepileptic treatment) and confirmed visual improvement. His antiepileptic treatment includes levetiracetam and valpromide. Once the tumor size reduction is stable surgical repair of CSF leak will be performed.
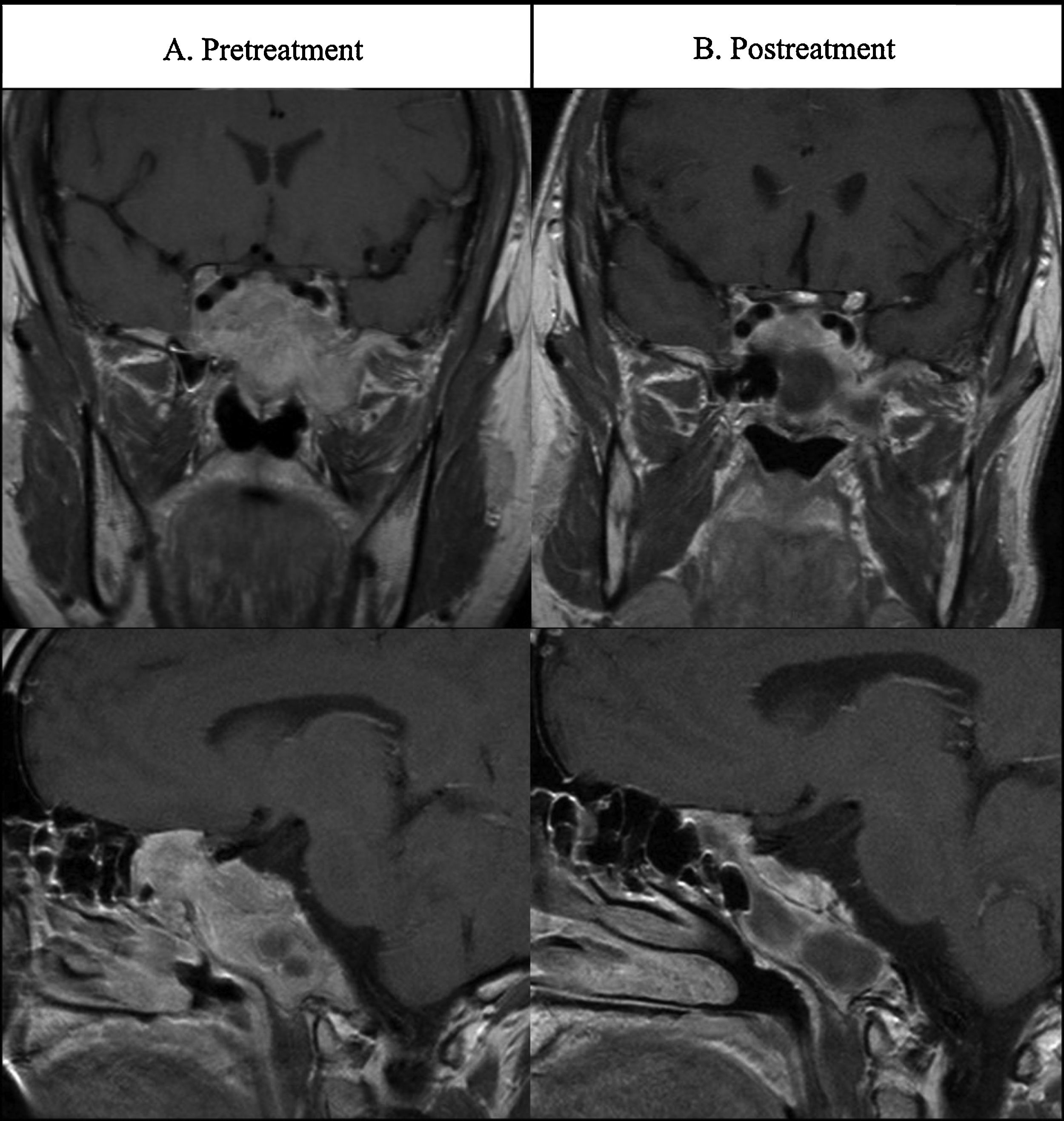
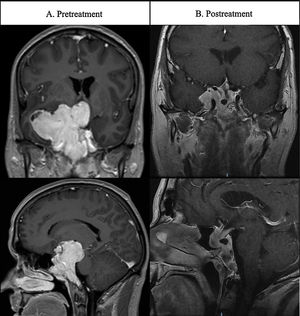
Case 2A 34-year old man presented anterior and posterior epistaxis. Nasal endoscopy showed a bloody polypoidal mass in both choanas. MRI revealed a large pituitary mass extending into the sphenoidal sinus, clivus, sellar region, superior and inferior orbital fissure and growing throught left choana (Fig. 2A). The tentative diagnosis was nasopharingeal angiofibroma but high serum PRL levels (814,984μIU/mL) led to the diagnosis of giant invasive prolactinoma. Hormonal evaluation revealed central hypogonadism due to hyperprolactinemia but no other pituitary deficits. Ophthalmological examination revealed bitemporal hemianopsia. Cabergoline treatment was initiated at 0.5mg/week. With this dosage a great drop of prolactin levels and a significant shrinkage of the prolactinoma was achieve, after 10 months of treatment. Cabergoline was increased to 0.75mg per week and after 6 months more to 1mg per week. However, cabergoline treatment was not well tolerated after titration to 1mg/week and bromocriptine treatment was initiated at 5mg/day and increased to 20mg/day (with dose titration each two-four months). Fifteen weeks later, a 50% reduction in PRL levels was observed. At the last follow-up visit (4 years and 3 months after treatment) PRL levels were still elevated. MRI at 24 weeks after treatment did not show a reduction in tumor size. However, the most recent MRI images (Fig. 2B), 185 weeks after the diagnosis, showed a tumor size of 43mm (58.9% decrease). With medical treatment the patient showed an entire recovery of his visual commitment.
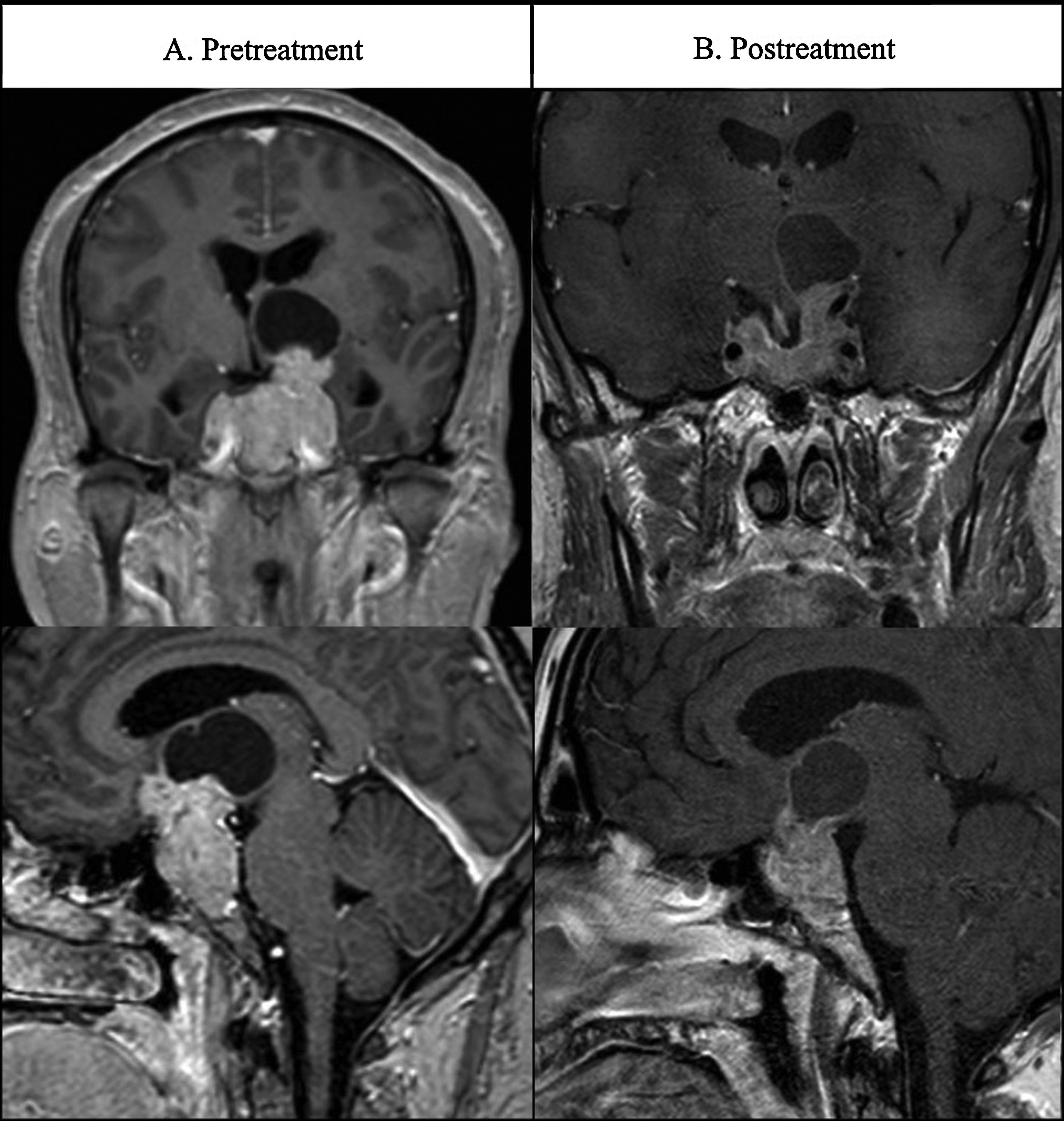
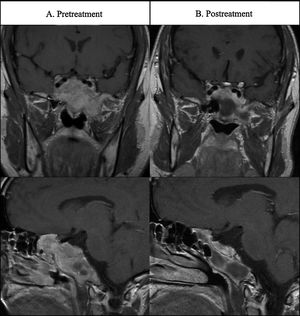
Case 3A 42-year-old man presented at the emergency room with a 10-minute episode of dysphasia. The patients reported headache and gradual loss of vision over the last 4 months. Cerebral MRI revealed a solid mass with cystic areas with 63mm of maximal diameter. Suprasellar invasion lead to compression of left basal ganglia and lateral ventricle dilatation (Fig. 3A). Hormonal evaluation revealed elevated serum PRL levels (108,180μIU/mL) and hypogonadotropic hypogonadism but no other pituitary deficits. Ophthalmological examination revealed bitemporal hemianopsia. Treatment with cabergoline was initiated at 0.5mg/week increasing to 1mg/week after 4 months. PRL levels were normalized at week 12. MRI at 12 weeks after treatment (Fig. 3B) showed a substantial reduction of the tumor size to 42mm (34% decrease). Even though PRL levels were normalized, hypogonadism persisted and testosterone therapy was initiated (Testosterone cypionate 250mg each 21 days). The patient has continued in cabergoline treatment with no clinical symptoms.
DiscussionTherapeutic managing of giant prolactinomas has considerably changed during the last few decades. Surgery was the mainstream treatment for this type of tumors, but older as well as most recent series showed high rates of morbidity (from 3.3 to 31.2%).4 In addition, several surgical and comparative series have demonstrated that surgical removal might not be sufficient to control tumor size and hormone levels, generally due to their large size and invasiveness. Yu et al. evaluated the effectiveness of both first-line treatments (surgery and medical therapy) showing better clinical outcomes in medical therapy group including a larger tumor volume reduction (91.3% vs 76.5%) and improvement in visual function (50% vs 11.1%).5 Due to the clinical evidence of better outcomes and lower complication rates, treatment with dopamine agonists is currently considered the first-line treatment for these invasive tumors. Surgery is reserved for patients with acute symptoms due to tumor mass (e.g. CSF leak, intracranial hypertension) and/or resistance to medical therapy.1 However, summary of evidence in this type of tumors is based in studies in which tumors larger than 60mm are underrepresented.1 The large giant tumors usually present a more aggressive behavior, with invasion of brain structures farther from sella and occasionally showing life-threatening conditions at clinical presentation.6 To the best of our knowledge, only another study of prolactinomas larger than 60mm has been previously published. Shimon et al. presented a cohort of 18 patients with giant prolactinomas bigger than 60mm from five pituitary centers in four different countries.3
We included 3 patients with prolactinomas larger than 60mm of a series of 283 patients with prolactinomas treated in our center from 2012 to 2017. Median age at diagnosis was 39 [34–42] years (Table 1). All of them were men. Despite of microprolactinomas are more frequent in women, macroprolactinomas are more frequently observed in men with a male/female ratio of 3:1.7 This gender bias in giant prolactinoma series is even more pronounced, close to 9:1,8 and it is also observed in giant prolactinomas larger than 60mm. Published series have shown that in men giant prolactinomas have not different natural history than non-giant ones.9 Nevertheless, giant prolactinomas in men are more aggressive and resistant (30% Vs 5%) to bromocriptine than in women.10,11 In contrast with regular prolactinomas, giant prolactinomas as well as large giant prolactinomas (larger than 60mm) had atypical symptoms at presentation. All patients in our series (100%) showed hypogonadism and visual dysfunction. Galactorrhea was not observed in our series. Our patients showed a higher rate of male hypogonadism compared to giant prolactinomas. In fact, Maiter et al. reported a rate of male hypogonadism of 61% (43/70) in giant prolactinomas.1 However, Shimon et al. observed a 100% rate (16/16) in large giant prolactinomas.3 Thus, prolactinomas larger than 60mm appear to cause male hypogonadism more frequently. Pituitary function has not been reported in many published series. Hypocortisolism was present in 22.2%, hypothyroidism in 38.89% and low IGF-1 in 16.67% of patients from Shimon et al. series.3 In our cases, other than hypogonadism, pituitary function was preserved in 2/3 patients. At least in 1 case (case 1) these symptoms were present years before the diagnosis probably reflecting a low tumor growth rate.
Characteristics of patients, presentation features.
| Case 1 | Case 2 | Case 3 | |
|---|---|---|---|
| Gender (M/F) | M | M | M |
| Age (years) | 39 | 34 | 42 |
| Neurological symptoms | YES | NO | YES |
| Visual impairment | YES | NO | NO |
| Testosterone pretreatment (9.9–27.8nmol/L) | 6.5 | 1 | 0.6 |
| Testosterone post-treatment (9.9–27.8nmol/L) | 6.4 | 11.9 | 5.1 |
| Panhypopituitarism | YES | NO | NO |
| Cavernous sinus invasion (Knosp score) | YES (4) | YES (4) | YES (4) |
Depending on the surrounding areas invaded by the tumor, different atypical symptoms may be found. Seizures, dementia, behavior abnormalities, or epistaxis are clinical findings which are rare but can be observed in giant prolactinomas.1 Two of our patients presented neurological symptoms. Case 1 presented absence seizures caused by extension of the tumor into the temporal lobe. Antiepileptic treatment and reduction of tumor size made these neurological symptoms subside and the patient has not had any seizure within the last year. Case 3 presented a variety of neurological symptoms due to the compression of basal ganglia and ventricular dilation. However, the neurological symptoms quickly improved after treatment with dopamine agonists and no surgical treatment was performed. In this case, surgical approach could be performed due to ventricle dilatation but a quick prolactin determination and dopamine agonist treatment led to symptomatic relief. At the present time no surgical intervention has been needed and significant tumor shrinkage was promptly achieved. Several case reports have shown safe and rapid relief in hydrocephalus and intracranial hypertension with dopamine agonists.6,12 Frontal lobe affectation leads to behavioral abnormalities and frontal dementia that can also be reversed with medical treatment.13
Caudal invasion might cause nasopharyngeal extension. In these cases, diagnosis is challenging and patients usually manifest respiratory problems or even epistaxis. In our case (patient 2), orbital fissures were also compromised and the diagnosis of angiofibroma was suggested. However, the determination of prolactin levels definitely stablished the diagnosis and permitted prompt initiation of dopamine agonist treatment.
First-line treatment for giant prolactinomas has been established based on different series, case reports and reviews, summarizing enough evidence to support medical treatment as a safe and effective first-line treatment for giant prolactinomas.1 However, prolactinomas larger than 60mm are extremely rare and they are underrepresented in published series.3 Their enormous size, invasiveness of surrounding structures and clinical presentation could determine the initial approach and follow-up. The large giant prolactinoma series by Shimon et al. showed a significant higher surgery rate compared to other published giant prolactinoma series. Surgery before medical treatment in giant prolactinomas is rarely performed given the proven efficacy of dopamine agonists even in life-threatening situations.6 Espinosa et al. reported a rate of first line surgery of 4.3% (2/47) in giant prolactinomas due to intracranial hypertension and pituitary apoplexy.8 In contrast, Shimon et al. showed a 22.2% (4/18) rate of first-line surgical treatment in large giant prolactinomas.3 None of our three cases needed an initial surgical approach, 2/3 patients showed neurological commitment but those symptoms were promptly reverted by medical treatment. Despite of the small size of our series, dopamine agonist seems to be an effective first line treatment, even in urgent situations. Rapid relief of neurological symptoms was achieved in two cases allowing to postpone or even avoid surgical intervention.
Overall, the reported pituitary surgery rate in large giant prolactinomas is slightly higher than in regular giant prolactinomas. In giant prolactinomas, a surgery rate of 14% has been reported1 in contrast to 50% reported in large giant prolactinomas.3 CSF leak, intracranial hypertension, pituitary apoplexy or non-response to medical treatment are the main causes of surgery after initiation of medical treatment. One of our patients (patient 1) developed CSF leak after initiation of dopamine agonist. CSF leak is generally presented in prolactinomas as a complication of medical therapy and tumor shrinkage. Lam et al. reviewed medically induced CSF leak in pituitary adenomas.14 CSF leak was observed in 52 patients of which 80.8% (42) were prolactinomas. 85.7% (36) of prolactinomas presented CSF leak after initiation of medical treatment. Meningitis was reported in 19.4% of medically induced CSF leak in prolactinomas. Recognition and treatment of CSF leak is necessary in order to reduce morbidity and mortality in these patient.14 Glucose and beta-2 transferrin levels determination in the nasal liquid can easily determine the presence of CSF rhinorrhea.15 In our case, CSF leak is being tightly monitored until we observe tumor size stabilization. If reconstructive surgery is made despite of continuous reduction in tumor size, CSK leak could recur.
Treatment of giant prolactinomas with dopamine agonists seems to be effective in reducing tumor size although the different methods used make it hard to compare the outcomes among the published series.1 In our case, we followed the RECIST criteria in which a reduction of at least 30% in maximum tumor diameter is considered partial response to treatment. All our patients (3/3) exhibited a significant reduction in tumor size with a median of 33.33% (Table 2). Shimon et al. series showed 83.3% (15/18) of significant tumor size reduction and 53.3% (8/18) with size reduction above 90%. Differences in size reduction between both series may be explained due to shorter follow-up in our patients (2.48±1.94 years vs 7.8±5.1 years). Medical treatment is also effective in normalization of PRL levels. 2/3 of our patients achieve a complete PRL normalization. One of the patients (case 2) exhibited intolerance to cabergoline that had to be withdrawn. Bromocriptine treatment in this patient achieved a 99.6% reduction of PRL levels but without total normalization (last PRL determination was 3012μIU/mL). Even though PRL elevation significant size reduction has been observed in this patient with a relative size reduction of 41.1%. This trend has been also reported in giant prolactinomas, Maiter et al. showed a significant size reduction among patients without normalization of PRL levels of 65% (22/34).1 Currently there are no standardized guidelines for cabergoline titration in giant prolactinomas. Nevertheless, the possibility of pituitary tumor apoplexy16 and the evidence of substantial tumor shrinkage with low doses of cabergoline suggests that starting with low doses and increasing as needed, based on prolactin levels, is a safer option.17 In all our three cases, low doses of cabergoline (0.5mg per week) achieved rapid relief from both urgent neurological symptoms and visual impairment.
Type and response to treatment.
| Patient | Initial size (mm) | Initial PRL (μIU/mL) | Type of dopamine agonist | Most recent dosage | Most recent PRL (μIU/mL) | Visual improvement | LCR fistula | Most recent size (mm) | Size reduction (%) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 79 | 52,594 | CABERGOLINE | 4mg (week) | 81 | YES | YES | 53 | 32.91 |
| 2 | 73 | 514,984 | BROMOCRIPTINE | 15mg (day) | 3012 | YES | NO | 43 | 41.1 |
| 3 | 63 | 108,180 | CABERGOLINE | 1mg (week) | 138 | YES | NO | 42 | 33.75 |
Our three patients presented with visual impairment. Treatment with dopamine agonists also improved these visual defects even though the tumor was still present and indeed, MRI imaging showed compression of the optic chiasm. These observations are in line with previous studies reporting that dopamine agonist treatment in giant prolactinomas resulted in significant improvement of visual defects in 96% of patients,1 and 85.7% in large giant prolactinomas.3
ConclusionsDopamine agonists are considered the first line of treatment for giant prolactinomas. The report of several case series indicates that pharmacological treatment is safe and effective while surgery must be reserved for patients with acute symptoms due to tumor mass and/or resistance to dopamine agonists. For prolactinomas larger than 60mm, however, data about the efficacy of these treatments is limited. Our series of three patients shows a good response to dopamine agonists, normalizing PRL levels in most cases, achieving substantial tumor shrinkage and marked improvement of clinical symptoms without major complications.
Medical treatment is also effective under life-threatening situations which usually occurs at presentation of this subset of prolactinomas. Rapid relief of neurological symptoms is achieved after initiation of medications allowing to postpone or avoiding surgery, without significant morbidity and mortality.
More studies and long-term follow-ups are necessary to determine the nature and behavior of large giant prolactinomas. In particular, it is important to predictive marker of medical response to develop better strategies of tumor control.
Author declarationThe authors confirm that the manuscript, is original research that has not been submitted and is not under consideration elsewhere. All the author have contributed in a substantial way to the described work and have participated in the preparation of the manuscript.
Funding statementThis research received funding from the ISCIII-Subdirección General de Evaluación y Fomento de la Investigación with FEDER founds, Spain (grant numbers: PI16/00175 to A.S-M. and D.A.C.) and Junta de Andalucía, Spain (grant numbers: A-0003-2016, A-0006-2017 to A.S-M., C-0015-2014 and RC-006-2018 to D.A.C.).
Compliance with ethical standardsAll procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of interestThe authors report no conflict of interest.