Central nervous system germ cell tumors need to be adequately diagnosed because their treatment is usually effective and they do not always require surgery. The study objectives are to describe the endocrine manifestations of these tumors and to compare the time of their onset to that of the occurrence of neurological and visual changes.
Patients and methodsThe medical histories of patients under 14 years of age seen at a pediatric endocrinology unit between 2000 and 2018 were reviewed. Wilcoxon and Fisher statistical tests were performed.
ResultsWe found 12 patients (10 females) with an age at diagnosis of 9.4±1.7 years and a follow-up time of 5.5±3.0 years, 10 with tumors in the sellar region, and each one with a pineal gland and a bifocal tumor. Clinical changes leading to diagnosis were neurological and/or visual in nine patients and hormonal in three. Seven patients diagnosed on the basis of neurological or visual symptoms had previously reported hormonal changes, giving us a total of 10 children at diagnosis (the most common diagnosis was central diabetes insipidus, found in 8). Endocrine symptoms had been present before diagnosis for 25.0±26.2 months, considerably longer than neuro-ophthalmological complaints (2.0±2.1 months, p=0.012).
ConclusionsAlmost all intracranial germ cell tumors have associated endocrine manifestations at diagnosis, with central diabetes insipidus the most common. Hormonal symptoms usually appear long before neuro-ophthalmological manifestations. Adequate clinical and endocrinological assessment may allow for an earlier diagnosis of these tumors.
Los tumores germinales del sistema nervioso central deben ser correctamente diagnosticados pues su tratamiento suele ser eficaz y no siempre requieren cirugía. Los objetivos del estudio son describir las manifestaciones endocrinas de estas neoplasias y comparar el momento de aparición de las mismas con el de las alteraciones neurológicas y visuales.
Pacientes y métodosRevisión de historias de pacientes menores de 14 años atendidos en una unidad de endocrinología pediátrica desde 2000 hasta 2018. Pruebas estadísticas: Wilcoxon y Fisher.
ResultadosSe estudió a 12 pacientes (10 mujeres) con una edad al diagnóstico de 9,4±1,7 años y un tiempo de seguimiento de 5,5±3,0 años; 10 presentaban tumores de la region selar, uno pineal y uno bifocal. Las alteraciones clínicas que llevaron al diagnóstico eran neurológicas o visuales en 9 casos y hormonales en 3. De los que consultaron por síntomas neurológicos o visuales, 7 refirieron previamente alteraciones hormonales, luego, estas estaban presentes en 10 de los niños al diagnóstico; la más frecuente fue la diabetes insípida central (8 casos). El periodo medio de presencia de síntomas endocrinológicos previos al diagnóstico fue de 25,0±26,2 meses, mucho más largo que el de los neurooftalmológicos, de 2,0±2,1 meses (p=0,012).
ConclusionesCasi todos los tumores germinales intracraneales presentan al diagnóstico manifestaciones endocrinas, siendo la más frecuente la diabetes insípida central. Los síntomas hormonales suelen presentarse bastante antes que los neuro y oftalmológicos. La correcta valoración clínica y endocrinológica puede adelantar el diagnóstico de estos tumores.
Germ cell tumors account for 3–15% of all intracranial primary tumors in childhood, and are considered to be the most curable central nervous system (CNS) malignancy. These tumors are usually located in the pineal gland, in the sellar region, in both locations at the same time or, more rarely, in other midline structures (the fourth ventricle, basal ganglia, thalamus, cerebral hemispheres, bulbar region and cerebellum). Leptomeningeal spread occurs in 10% of the cases, but hematogenous metastases are exceptional. Those lesions that secrete the tumor markers alpha-fetoprotein (αFP) or beta-chorionic gonadotropin (βHCG) are more aggressive and have a poorer prognosis.1–5
In the case of tumors located in the sellar region (hypothalamic-pituitary-chiasmatic lesions), a differential diagnosis must be established with lesions that are heterogeneous in terms of their nature and therapeutic approach.6,7 While craniopharyngioma - the most common neoplasm in this location in the first two decades of life - usually requires surgery, we should always first rule out other nonsurgical lesions such as prolactinoma, pituitary hyperplasia secondary to chronic primary hypothyroidism, and, in particular, germ cell tumors, the first choice treatment of which is chemo- and radiotherapy.8–10 Thus, the measurements of αFP and βHCG, as well as pituitary hormone assessment, are priority elements in the initial differential diagnosis of a sellar mass, especially where surgery may be indicated.
The present study describes the endocrine manifestations of germ cell tumors of the CNS in children and adolescents, and compares their time of occurrence with that of the neurological and visual alterations and radiological detection of the tumor.
Patients and methodsA retrospective study was made of patients under 14 years of age referred to the Pediatric Endocrinology Unit of a tertiary hospital due to the presence of a germ cell tumor of the CNS during a 19-year period (2000–2018). The lesions were diagnosed by magnetic resonance imaging (MRI) and biopsy (in non-secretory lesions) or by high levels of βHCG or αFP in plasma or cerebrospinal fluid (in the case of secretory lesions).
The following data were recorded from the clinical records: gender, age, symptoms and signs of clinical presentation (and their duration), long axis measurement of the mass in the MRI study at the time of tumor diagnosis, primary treatment and the occurrence of endocrine diseases over the follow-up period.
The symptoms and signs of a clinical presentation were defined as the first lesion-related manifestations leading to the diagnosis, though we also recorded those manifestations reported or observed earlier. Different forms of presentation were established: neurological (headache, signs of cranial hypertension, seizures, focal signs), ophthalmological (decreased visual acuity or field, strabismus, nystagmus) or endocrinological. The prior clinical period was defined as the time from the onset of symptoms attributable to the lesion (even if not leading to medical consultation) until the diagnosis.
Endocrine diseases included disorders of hypothalamic origin (new onset obesity, eating or thirst disorders, sleep disturbances or temperature alterations) and pituitary problems secondary to deficiencies of any of the anterior (growth hormone, corticotropic hormone, thyrotropic hormone, luteinizing hormone and follicle-stimulating hormone) or posterior pituitary hormones (central diabetes insipidus [CDI]), or hormone hyperproduction (hyperprolactinemia, early puberty, gigantism). The clinical and laboratory test assessments were those regarded as standard in the care protocol of these patients, and were made at diagnosis of the lesion and then at least every 6 months.
Quantitative variables were reported as the mean±standard deviation (SD), and were compared using the Wilcoxon test. Qualitative variables were reported as proportions and were compared using the Fisher test.
ResultsTwelve patients (of whom 10 were females) between 7–12 years of age (mean 9.4±1.7 years) and with a mean follow-up of 5.5±3.0 years were included in the study. Ten of the tumors were located in the sellar region, one in the pineal gland, and one was bifocal (in both locations). In 11 cases the long axis of the lesion in the first MRI study measured >10mm, though in one of them the first MRI scans were normal, and in one girl the initial lesion consisted of a slight widening of the pituitary stalk. The specific diagnosis was established from the elevation of plasma βHCG and αFP in 6 cases (“secretory” tumors) and from the biopsy findings in the other 6 cases (“non-secretory” tumors, with no such markers in either plasma or cerebrospinal fluid). All the children received chemo- and radiotherapy, and three also underwent surgical resection of the lesion (those in whom chemotherapy did not achieve remission).
The clinical signs leading to the diagnosis of the tumor were neurological or visual in 9 cases and endocrinological in three cases (CDI in all three). In addition, 7 of the 9 patients diagnosed on the basis of neurological or visual disturbances reported other endocrinological disorders starting months and even several years before (polyuric syndrome in 5 children; early puberty at 7 years of age in one case; and eating disorder in another) that had not been diagnosed, except for two cases of CDI, which were already being monitored in our clinic. Of these two cases, one was an “idiopathic” condition starting 5 years before and thus classified because it had proved negative in previous MRI studies, while the other consisted of pituitary stalk widening starting three years before. Overall, 10 of the 12 patients had endocrine manifestations at baseline, of which 8 corresponded to CDI. The mean duration of the hormonal symptoms prior to diagnosis was 25.0±26.2 months, this being much longer than that of the neurological or visual symptoms, with 2.0±2.1 months (p=0.012).
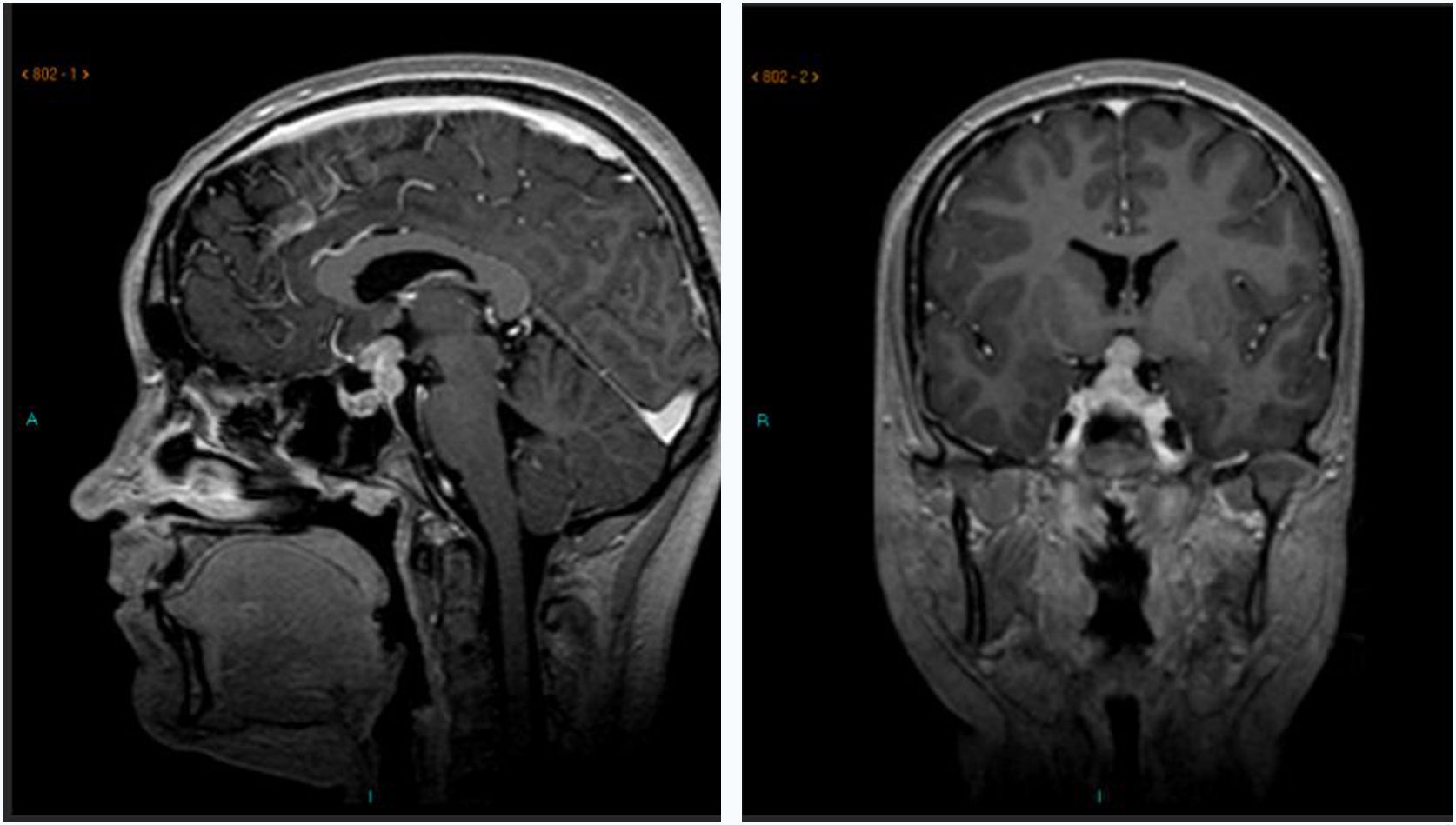
All the sellar tumor cases developed panhypopituitarism. Two of them did so in a sequential manner: the patient with pituitary stalk widening after consulting due to polyuria in the context of isolated CDI developed anterior pituitary deficiency three years later, coinciding with tumor growth (Fig. 1), thus indicating biopsy since there had been no tumor marker secretion; and the patient with “idiopathic” CDI who four years later likewise developed “idiopathic” growth hormone deficiency, the MRI findings remaining normal. In this latter case the neurological manifestations led months later to the diagnosis of the neoplasm. The other patients with sellar tumors developed panhypopituitarism after the start of chemotherapy. Other endocrine complications seen over follow-up were obesity in four cases and thirst mechanism disorders in two cases, both following surgery.
In addition to the acute neurological deficit leading to diagnosis, the only patient with pineal tumor in our series also experienced early puberty starting months before, though there had been no medical consultation due to this condition. Following treatment and remission, the testosterone concentrations returned to prepubertal levels with the disappearance of the secreted βHCG, thus confirming that the tumor had been the cause of early puberty.
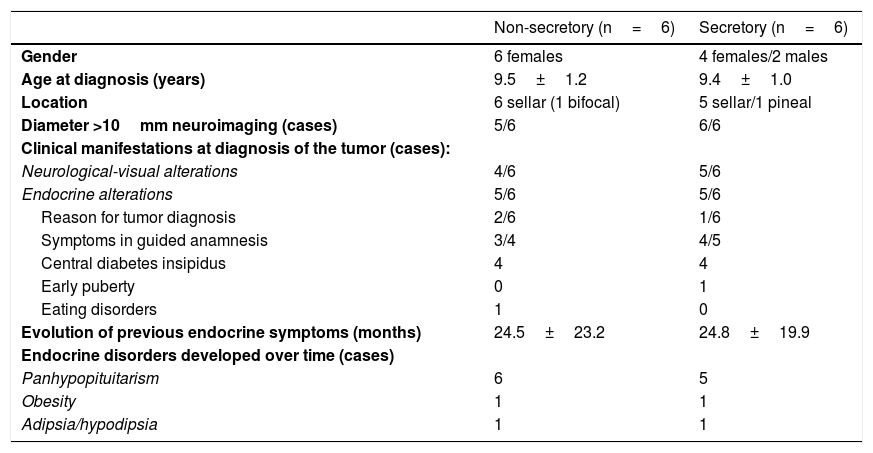
Table 1 shows the study variables in the two patient subgroups with secretory and non-secretory tumors; no significant differences between them were observed.
Study variables in the two subgroups of germ cell tumors (secretory and non-secretory).
| Non-secretory (n=6) | Secretory (n=6) | |
|---|---|---|
| Gender | 6 females | 4 females/2 males |
| Age at diagnosis (years) | 9.5±1.2 | 9.4±1.0 |
| Location | 6 sellar (1 bifocal) | 5 sellar/1 pineal |
| Diameter >10mm neuroimaging (cases) | 5/6 | 6/6 |
| Clinical manifestations at diagnosis of the tumor (cases): | ||
| Neurological-visual alterations | 4/6 | 5/6 |
| Endocrine alterations | 5/6 | 5/6 |
| Reason for tumor diagnosis | 2/6 | 1/6 |
| Symptoms in guided anamnesis | 3/4 | 4/5 |
| Central diabetes insipidus | 4 | 4 |
| Early puberty | 0 | 1 |
| Eating disorders | 1 | 0 |
| Evolution of previous endocrine symptoms (months) | 24.5±23.2 | 24.8±19.9 |
| Endocrine disorders developed over time (cases) | ||
| Panhypopituitarism | 6 | 5 |
| Obesity | 1 | 1 |
| Adipsia/hypodipsia | 1 | 1 |
Our data demonstrate the importance of the early identification of symptoms and signs of endocrine disorders in childhood and adolescence, since they sometimes constitute the first manifestation of a neoplasm, particularly CDI. As seen in our series, most sellar lesions are associated with endocrine clinical manifestations at diagnosis, many of them already long-standing. In a large series of 176 pediatric patients with sella turcica tumors, two-thirds presented hormonal alterations over a mean interval of 6months before onset, with space-occupying symptoms.10 Of the different sellar neoplasms, germ cell tumors are those which cause hypopituitarism most often and earliest. These cases usually start with CDI and later develop anterior pituitary gland deficiencies. The largest literature review to date comprised 97 pediatric cases of germ cell tumors of the CNS, and found that over 90% presented endocrine symptoms at baseline. Of these, the earliest and most frequent (over 80% of the cases) was CDI.11 Those cases that do not present panhypopituitarism at diagnosis develop it after the start of oncological treatment.11–14
Of note in our series is the presence of several cases with very long-standing CDI (between 1–5years) that had not been referred for the study of polyuria. Other authors also report this delay as being usual. When longer than one year, the delay may be associated with a risk of tumor spread.15
The presentation of forms with normal or atypical radiographic findings poses another challenge. As we have seen, germ cell tumors may begin with a widening of the pituitary stalk. In such cases, due clinical and radiological follow-up is required, and a biopsy is required in the event of the progression of hormone deficiencies or a radiologically manifest increase in size.16–18 Likewise, a periodic clinical and radiological re-evaluation of “idiopathic” pituitary deficiencies (i.e., those with normal MRI findings) is also very important, since the pathological images sometimes take years to appear.19,20
With regard to tumors of pineal origin, these lesions usually manifest with neurological symptoms, but we must bear in mind that the production of βHCG can cause early puberty, particularly in males (very rarely in females), and this biochemical parameter should be included in the study.21
In conclusion, it is essential to recognize the symptoms and signs of endocrinopathy in childhood and adolescence in order to establish an early diagnosis of germ cell tumors of the CNS and thus reduce patient morbidity and mortality, since many patients present such manifestations well before any neurological or visual symptoms and, sometimes, before any radiological alterations are observed. Central diabetes insipidus, the most common endocrinopathy, is usually diagnosed late. Attempts should always be made to establish the underlying cause and, in “idiopathic” presentations or cases with poorly defined radiological findings, such as a widening of the pituitary stalk, we should persist in the investigation of a possible germ cell tumor.
Conflicts of interestThe authors declare that they have no conflicts of interest.
Please cite this article as: García García E, Gómez Gila AL, Merchante E, Rivero Garvia M, Venegas Moreno E, Soto Moreno A, et al. Manifestaciones endocrinas de los tumores germinales del sistema nervioso central en niños. Endocrinol Diabetes Nutr. 2020;67:540–544.