To assess the prevalence of oropharyngeal dysphagia (OD) using the Eating Assessment Tool (EAT-10) and its association with malnutrition and long-term mortality.
Material and methodsA retrospective cohort study of patients admitted to the general internal medicine ward. In the first 48h after hospital admission, OD was assessed using the EAT-10, and presence of malnutrition with the Mini Nutritional Assessment-Short Form (MNA-SF). Association of OD to malnutrition and long-term mortality was analyzed.
ResultsNinety patients with a mean age of 83 (SD: 11.8) years were enrolled. Of these, 56.7% were at risk of OD according to EAT-10. This group of patients had greater prevalence rates of malnutrition (88.2% vs. 48.7%; p=0.001) and mortality (70% vs. 35.9%; p=0.001). During follow-up for 872.71 (SD: 642.89) days, risk of DO according to EAT-10 was an independent predictor of mortality factor in a multivariate analysis (HR: 2.8; 95%CI: 1.49–5.28; p=0.001).
ConclusionsThe EAT-10 is a useful tool for screening OD. Adequate screening for OD is important because of its associated risks of malnutrition and long-term mortality.
Evaluar la prevalencia de disfagia orofaríngea (DO) mediante el Eating Assessment Tool-10 (EAT-10) y su asociación con desnutrición y mortalidad a largo plazo.
Material y métodosEstudio de cohortes retrospectivo de pacientes hospitalizados en medicina interna. En las primeras 48h del ingreso de los pacientes se evaluó la DO mediante el test EAT-10 y la presencia de desnutrición con el Mini Nutritional Assessment-Short Form (MNA-SF). Se estudió la asociación de la presencia de DO con la desnutrición y la mortalidad a largo plazo.
ResultadosSe incluyeron 90 pacientes con una edad media de 83 (DE: 11,74) años. El 56,7% (n=51) presentaron riesgo de DO según EAT-10. Este grupo de pacientes presentó mayores prevalencias de desnutrición (88,2% vs. 48,7%; p=0,001) y mortalidad (70% vs 35,9%; p=0,001). Durante un seguimiento de 872,71 (DE: 642,89) días el riesgo de DO según EAT-10 fue un factor predictivo independiente de mortalidad en análisis multivariante (HR: 2,8; IC95%: 1,49-5,28; p=0,001).
ConclusionesEl test EAT-10 es una herramienta útil en el cribado de la DO. Es importante realizar un cribado adecuado de DO debido a los riesgos asociados de desnutrición y mortalidad a largo plazo que conlleva.
Oropharyngeal dysphagia (OD) is a common problem in the elderly population. It has been estimated that up to 47% of all hospitalized elderly patients and up to 75% of all institutionalized elderly patients may have clinical signs of OD.1,2
The identification of patients at risk of developing OD is crucial. The presence of OD is significantly associated with two clinically relevant complications: decreased swallowing efficiency that can lead to malnutrition and dehydration; and impaired swallowing safety that may result in aspiration and aspiration pneumonia.2 Thus, OD is clearly associated with prolonged hospital stay and with increased mortality both in hospital and in the twelve months following hospital discharge.3 Because of its prevalence and associated clinical consequences, OD is currently considered a geriatric syndrome that should be systematically evaluated in the elderly population in order that its main complications can be treated and prevented.4
Videofluoroscopy is the gold standard for the diagnosis of OD.5 This is a dynamic radiological technique in which sequences of the different swallowing stages are obtained after the administration of radiological contrast media possessing different viscosities. The main disadvantage of videofluoroscopy is that it requires medical specialization and technical resources that are not readily available at most healthcare levels and in most healthcare centers. In addition, it requires a significant degree of patient cooperation that is not always possible in many cases, particularly when dealing with chronic patients and patients with multiple diseases.5
The objective of screening in OD therefore should be to identify subjects at risk of bronchoaspiration and/or malnutrition, with a view to referring them for a more comprehensive study of OD, such as that provided by videofluoroscopy.6 In daily clinical practice, simpler methods than videofluoroscopy have been developed to adequately screen individuals at risk of developing OD.6,7 The Volume-Viscosity Swallow Test (V-VST) evaluates the occurrence of clinical signs of OD through the administration of boluses of different viscosity in subjects at risk of suffering OD. It offers adequate sensitivity in detecting OD (83.7%) and aspiration (100%).6,8 Interobserver agreement is moreover also adequate.8 However, the technique requires adequate training, which precludes its widespread use.3 Another employed screening tool is the Eating Assessment Tool-10 (EAT-10).7 The EAT-10 is a verbal, unidimensional, self-administered, direct-scored analog instrument for the evaluation of specific symptoms of dysphagia that has demonstrated excellent internal consistency and reproducibility. Furthermore, it has been validated in the Spanish population9 and has demonstrated its ability to predict bronchoaspiration.8,10
The primary objective of our study was to determine whether the score obtained with the EAT-10 test, performed 48h after hospital admission, is associated with an increase in long-term mortality in a cohort of elderly patients hospitalized due to acute disease. The secondary objectives were to estimate the prevalence of OD and its relation to bronchoaspiration and malnutrition at the time of hospital admission.
Material and methodsStudy designAn analytical, retrospective cohort study was carried out.
PatientsThe study comprised adult male and female patients admitted to the Internal Medicine ward of a tertiary general hospital with 802 beds during the months of January, February and March 2012, with subsequent follow-up until death or the study closure date in April 2016. Follow-up was performed by reviewing the electronic case history. We excluded patients unable to cooperate in the EAT-10 test due to advanced dementia or severe neurological disease, in the absence of an accompanying relative who could be interviewed instead of the patient.
Clinical variablesDemographic variables were collected, together with patient origin and reason for admission. Cognitive status was assessed by asking the subject simple questions referring to orientation in time and space, and person. Patients with previous dementia diagnostic reports were included in the cognitive impairment group. Body weight (kg) and height (cm) were recorded for anthropometric evaluation. The body mass index (BMI) was calculated by dividing weight by height squared (kg/m2). In situations where patient weight and/or height could not be measured, these variables were obtained using estimation methods. Height was estimated based on forearm length, using the tables proposed by the British Association for Parenteral and Enteral Nutrition (BAPEN).11 Weight in turn was estimated using the Chumlea equation,12 and BMI was estimated using arm circumference and values proposed by BAPEN.11
In the first 48h after admission, nutritional and OD screenings were performed. Nutritional screening was performed using the Mini-Nutritional Assessment-Short Form (MNA-SF).13 The MNA-SF is the test recommended by the European Society for Clinical Nutrition and Metabolism to detect malnutrition and malnutrition risk in the elderly population at outpatient and hospital level.14 A score between 8 and 11 represents a risk of malnutrition, while malnutrition is defined as a score of ≤7. Screening for OD was performed using the Spanish version of the EAT-10 questionnaire.9 In the EAT-10 test, the patient or relative answers 10 questions related to the occurrence and severity of symptoms of OD. Each question is scored on a scale from 0 (no problem) to 4 (severe problem). Data from different studies define a score of ≥3 as indicating OD risk.10
The Charlson Comorbidity Index (CCI) was used as a summary measure of the presence of patient comorbidities.15
Clinical endpointThe clinical endpoint was mortality during follow-up after hospitalization.
Statistical analysisQuantitative variables were reported as the mean and standard deviation (SD) or as the median and interquartile range (IQR), while qualitative variables were reported as percentages and frequencies.
Quantitative variables were compared between two groups using the Student t-test or nonparametric Mann–Whitney U-test, while proportions were compared using the chi-squared test. A linear trend test was applied for variables with ordered categories.
The difference in patient survival according to the EAT-10 score was evaluated based on Kaplan–Meier curves and the log rank test. The mortality risk of the variables was established using univariate and multivariate Cox regression models, estimating the hazard ratios (HRs) with their 95% confidence interval (95%CI). The multivariate models were generated entering age, gender and other variables yielding p<0.1 in the univariate analysis. A sequential exclusion procedure was then applied to select the best model. Associations with p<0.05 were regarded as significant.
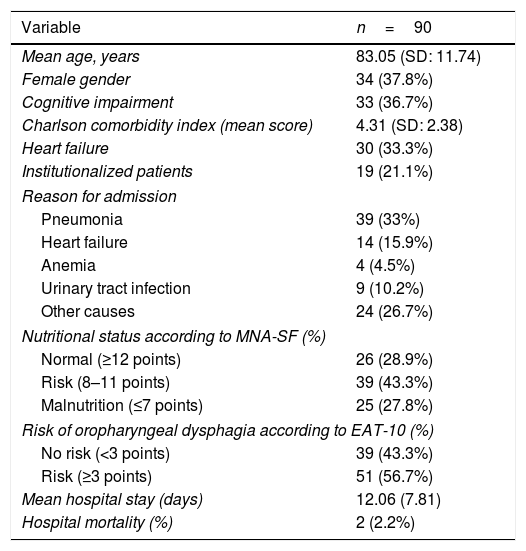
ResultsA total of 90 patients (34 women and 56 men) with a mean age of 83 years (SD: 11.8) were initially evaluated. The mean hospital stay was 12.1 days (SD: 7.8). Table 1 shows the most relevant data of the initial sample. Follow-up was carried out in 89 patients (one of the patients was lost to follow-up and his vital status could not be confirmed). The median duration of follow-up was 1187 days (IQR: 1342), with a mean of 872 days (SD: 68). Forty-nine patients died (54.4%).
Demographic and clinical characteristics of the patients.
| Variable | n=90 |
|---|---|
| Mean age, years | 83.05 (SD: 11.74) |
| Female gender | 34 (37.8%) |
| Cognitive impairment | 33 (36.7%) |
| Charlson comorbidity index (mean score) | 4.31 (SD: 2.38) |
| Heart failure | 30 (33.3%) |
| Institutionalized patients | 19 (21.1%) |
| Reason for admission | |
| Pneumonia | 39 (33%) |
| Heart failure | 14 (15.9%) |
| Anemia | 4 (4.5%) |
| Urinary tract infection | 9 (10.2%) |
| Other causes | 24 (26.7%) |
| Nutritional status according to MNA-SF (%) | |
| Normal (≥12 points) | 26 (28.9%) |
| Risk (8–11 points) | 39 (43.3%) |
| Malnutrition (≤7 points) | 25 (27.8%) |
| Risk of oropharyngeal dysphagia according to EAT-10 (%) | |
| No risk (<3 points) | 39 (43.3%) |
| Risk (≥3 points) | 51 (56.7%) |
| Mean hospital stay (days) | 12.06 (7.81) |
| Hospital mortality (%) | 2 (2.2%) |
SD: standard deviation; EAT-10: Eating Assessment Tool-10; MNA-SF: Mini-Nutritional Assessment-Short Form; NRS-2002: Nutrition Risk Screening-2002.
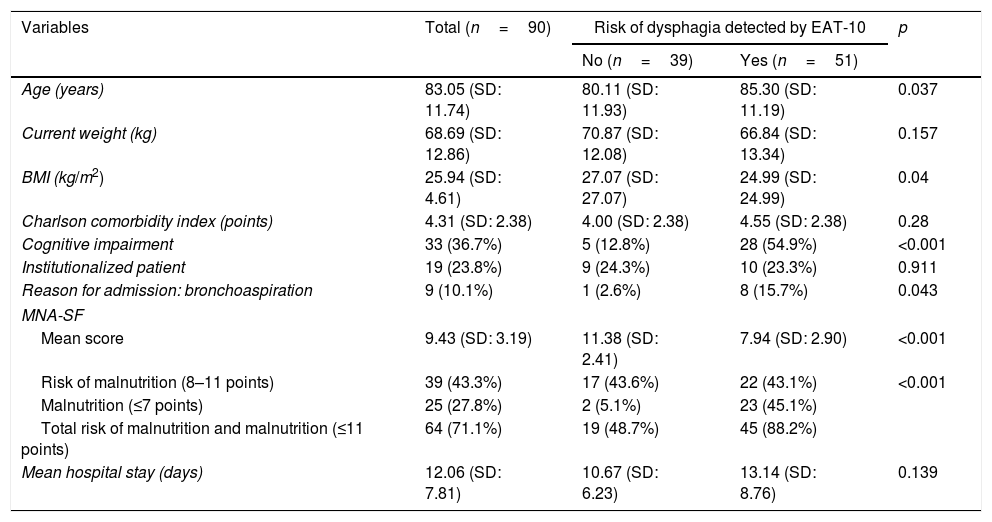
The median score of the EAT-10 test was 4, with scores of 0, 4 and 14 points corresponding to percentiles 25, 50 and 75, respectively. The patients were classified into two groups according to their risk of developing OD as determined by the EAT-10 test: patients at risk of OD (n=51; 56.7%) and patients at no risk of OD (n=39; 43.3%). Patients at risk of OD were older, had a lower BMI, a poorer MNA-SF score, and a greater incidence of cognitive impairment (Table 2). Mortality over follow-up was higher among the patients at risk of suffering OD (70% vs. 35.9%; p<0.001). Likewise, in the group at risk of OD, 8 patients (15.7%) had a diagnosis of bronchoaspiration pneumonia upon admission versus one of the patients without OD (2.6%) (p=0.043).
Clinical and demographic characteristics of the patients according to the risk of oropharyngeal dysphagia as determined by the EAT-10 test.
| Variables | Total (n=90) | Risk of dysphagia detected by EAT-10 | p | |
|---|---|---|---|---|
| No (n=39) | Yes (n=51) | |||
| Age (years) | 83.05 (SD: 11.74) | 80.11 (SD: 11.93) | 85.30 (SD: 11.19) | 0.037 |
| Current weight (kg) | 68.69 (SD: 12.86) | 70.87 (SD: 12.08) | 66.84 (SD: 13.34) | 0.157 |
| BMI (kg/m2) | 25.94 (SD: 4.61) | 27.07 (SD: 27.07) | 24.99 (SD: 24.99) | 0.04 |
| Charlson comorbidity index (points) | 4.31 (SD: 2.38) | 4.00 (SD: 2.38) | 4.55 (SD: 2.38) | 0.28 |
| Cognitive impairment | 33 (36.7%) | 5 (12.8%) | 28 (54.9%) | <0.001 |
| Institutionalized patient | 19 (23.8%) | 9 (24.3%) | 10 (23.3%) | 0.911 |
| Reason for admission: bronchoaspiration | 9 (10.1%) | 1 (2.6%) | 8 (15.7%) | 0.043 |
| MNA-SF | ||||
| Mean score | 9.43 (SD: 3.19) | 11.38 (SD: 2.41) | 7.94 (SD: 2.90) | <0.001 |
| Risk of malnutrition (8–11 points) | 39 (43.3%) | 17 (43.6%) | 22 (43.1%) | <0.001 |
| Malnutrition (≤7 points) | 25 (27.8%) | 2 (5.1%) | 23 (45.1%) | |
| Total risk of malnutrition and malnutrition (≤11 points) | 64 (71.1%) | 19 (48.7%) | 45 (88.2%) | |
| Mean hospital stay (days) | 12.06 (SD: 7.81) | 10.67 (SD: 6.23) | 13.14 (SD: 8.76) | 0.139 |
SD: standard deviation; EAT-10: Eating Assessment Tool-10; MNA-SF: Mini-Nutritional Assessment-Short Form.
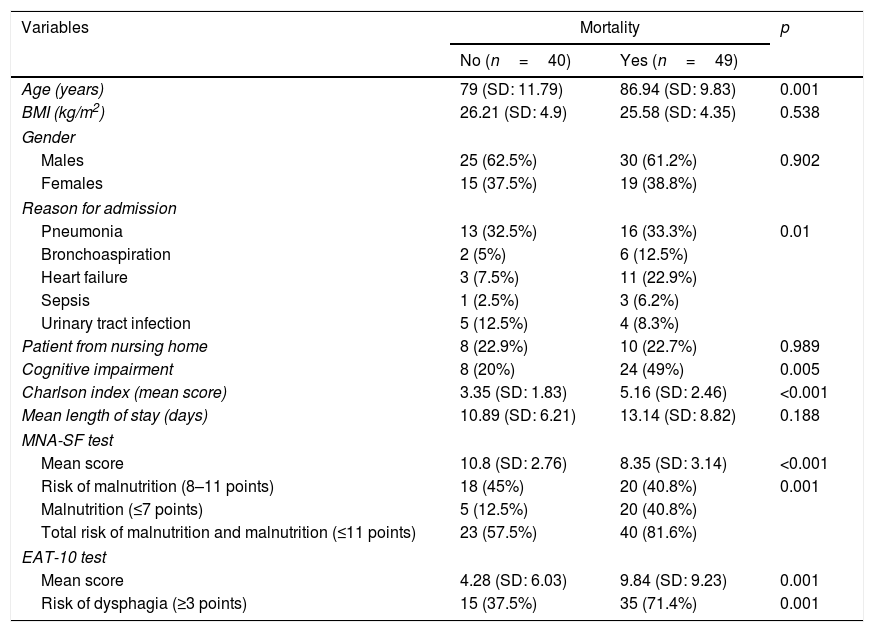
The patients were also divided into two groups according to the occurrence of death during the follow-up period (Table 3). The median EAT-10 score was 7 in the group of patients who died versus 1 in those who did not. The patients who died were older and had a higher CCI score, a poorer MNA-SF score, and a higher prevalence of cognitive impairment (Table 3). Gender was not found to influence mortality risk (Table 4).
Clinical characteristics of the patients according to mortality occurrence during follow-up.
| Variables | Mortality | p | |
|---|---|---|---|
| No (n=40) | Yes (n=49) | ||
| Age (years) | 79 (SD: 11.79) | 86.94 (SD: 9.83) | 0.001 |
| BMI (kg/m2) | 26.21 (SD: 4.9) | 25.58 (SD: 4.35) | 0.538 |
| Gender | |||
| Males | 25 (62.5%) | 30 (61.2%) | 0.902 |
| Females | 15 (37.5%) | 19 (38.8%) | |
| Reason for admission | |||
| Pneumonia | 13 (32.5%) | 16 (33.3%) | 0.01 |
| Bronchoaspiration | 2 (5%) | 6 (12.5%) | |
| Heart failure | 3 (7.5%) | 11 (22.9%) | |
| Sepsis | 1 (2.5%) | 3 (6.2%) | |
| Urinary tract infection | 5 (12.5%) | 4 (8.3%) | |
| Patient from nursing home | 8 (22.9%) | 10 (22.7%) | 0.989 |
| Cognitive impairment | 8 (20%) | 24 (49%) | 0.005 |
| Charlson index (mean score) | 3.35 (SD: 1.83) | 5.16 (SD: 2.46) | <0.001 |
| Mean length of stay (days) | 10.89 (SD: 6.21) | 13.14 (SD: 8.82) | 0.188 |
| MNA-SF test | |||
| Mean score | 10.8 (SD: 2.76) | 8.35 (SD: 3.14) | <0.001 |
| Risk of malnutrition (8–11 points) | 18 (45%) | 20 (40.8%) | 0.001 |
| Malnutrition (≤7 points) | 5 (12.5%) | 20 (40.8%) | |
| Total risk of malnutrition and malnutrition (≤11 points) | 23 (57.5%) | 40 (81.6%) | |
| EAT-10 test | |||
| Mean score | 4.28 (SD: 6.03) | 9.84 (SD: 9.23) | 0.001 |
| Risk of dysphagia (≥3 points) | 15 (37.5%) | 35 (71.4%) | 0.001 |
SD: standard deviation; EAT-10: Eating Assessment Tool-10; BMI: body mass index; MNA-SF: Mini-Nutritional Assessment-Short Form.
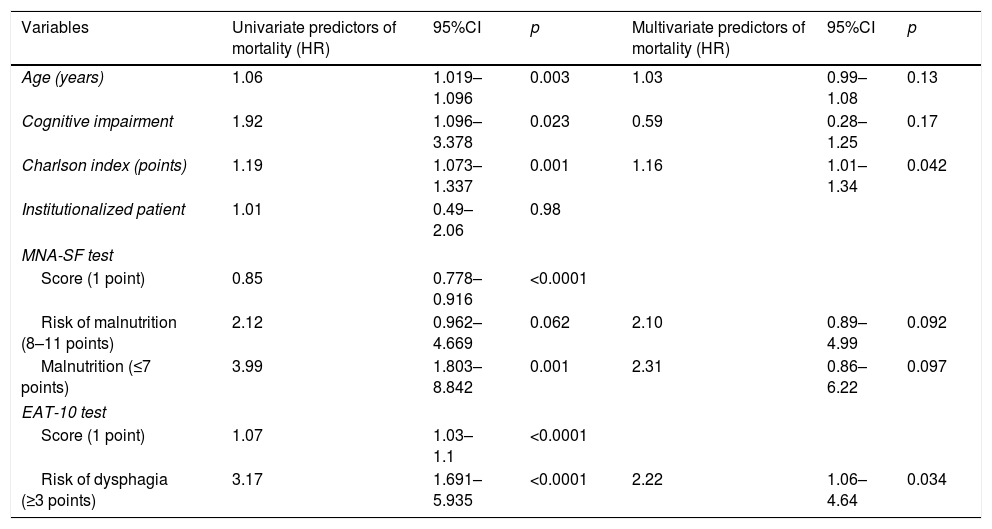
Mortality risk factors in the univariate and multivariate analyses. The HRs of the variables found to be significant in the univariate analysis and also in the fully adjusted multivariate analysis are reported.
| Variables | Univariate predictors of mortality (HR) | 95%CI | p | Multivariate predictors of mortality (HR) | 95%CI | p |
|---|---|---|---|---|---|---|
| Age (years) | 1.06 | 1.019–1.096 | 0.003 | 1.03 | 0.99–1.08 | 0.13 |
| Cognitive impairment | 1.92 | 1.096–3.378 | 0.023 | 0.59 | 0.28–1.25 | 0.17 |
| Charlson index (points) | 1.19 | 1.073–1.337 | 0.001 | 1.16 | 1.01–1.34 | 0.042 |
| Institutionalized patient | 1.01 | 0.49–2.06 | 0.98 | |||
| MNA-SF test | ||||||
| Score (1 point) | 0.85 | 0.778–0.916 | <0.0001 | |||
| Risk of malnutrition (8–11 points) | 2.12 | 0.962–4.669 | 0.062 | 2.10 | 0.89–4.99 | 0.092 |
| Malnutrition (≤7 points) | 3.99 | 1.803–8.842 | 0.001 | 2.31 | 0.86–6.22 | 0.097 |
| EAT-10 test | ||||||
| Score (1 point) | 1.07 | 1.03–1.1 | <0.0001 | |||
| Risk of dysphagia (≥3 points) | 3.17 | 1.691–5.935 | <0.0001 | 2.22 | 1.06–4.64 | 0.034 |
EAT-10: Eating Assessment Tool-10; HR: hazard ratio; MNA-SF: Mini-Nutritional Assessment-Short Form.
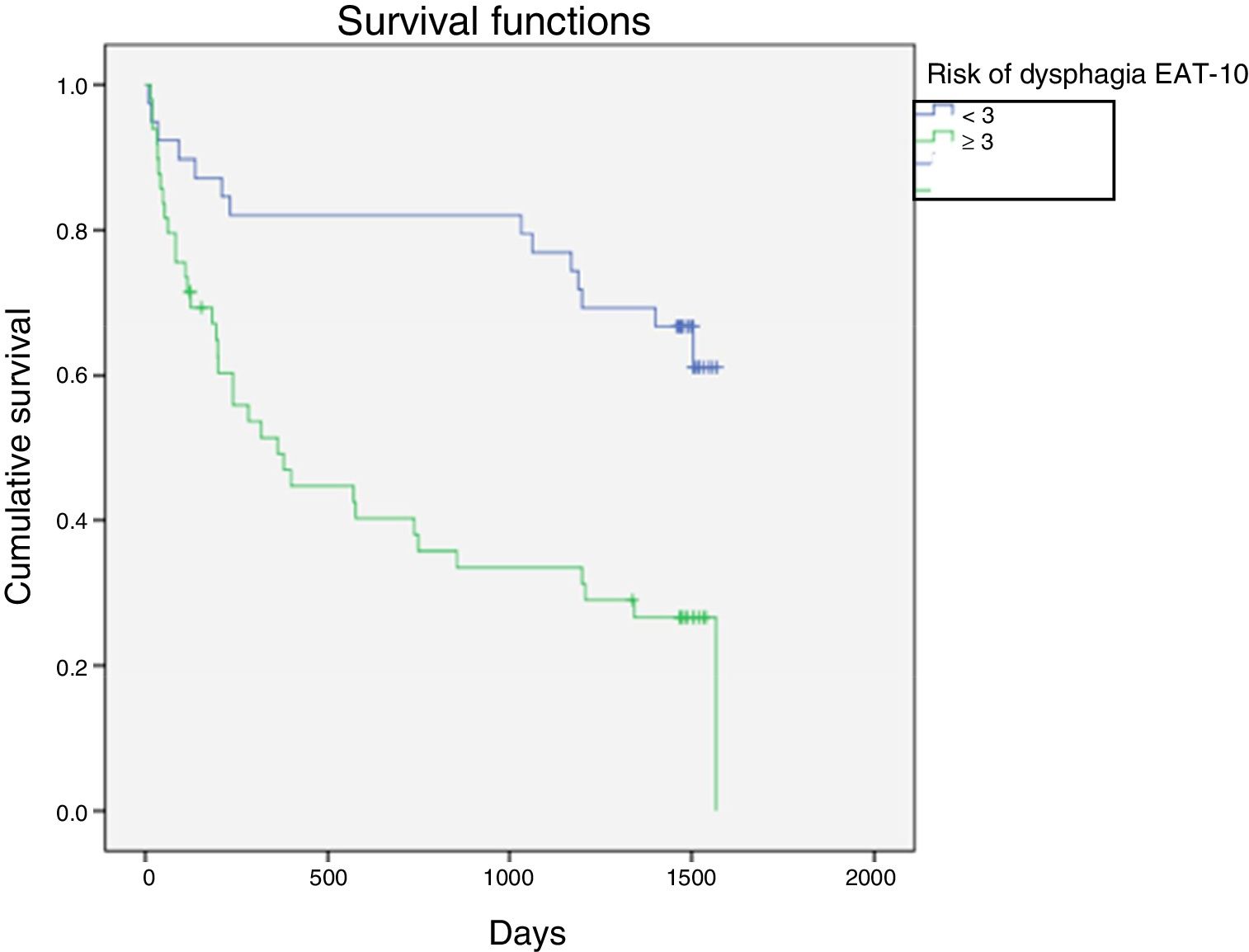
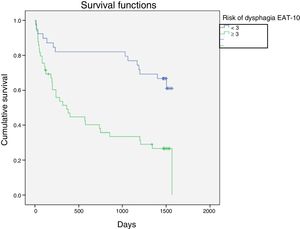
Kaplan–Meier curves were constructed to assess patient survival stratified by the EAT-10 score. The statistical significance of the differences between the curves of each group was analyzed using the log-rank test (Fig. 1). Survival was significantly reduced in those at risk of OD according to the EAT-10 test (mean survival in days=666 vs. 1242; p<0.001).
Based on Cox regression analysis, the factors predictive of increased mortality risk in the univariate analysis were seen to be age, the CCI, the EAT-10, the MNA-SF and the presence of cognitive impairment. In the multivariate analysis, after applying the sequential exclusion procedure, the only variables found to be predictive of mortality risk were the CCI (HR: 1.17; 95%CI: 1.04–1.32; p=0.011) and the risk of OD according to the EAT-10 questionnaire (HR: 2.8; 95%CI: 1.49–5.28; p=0.001).
DiscussionThe present study recorded a 56.7% prevalence of OD risk as detected by the EAT-10 questionnaire in elderly patients admitted to a tertiary hospital due to acute disease. Likewise, the study showed the risk of OD, as assessed by the EAT-10 test, to be an independent predictor of long-term mortality. In turn, it was associated with the presence of bronchoaspiration and malnutrition as assessed by the MNA-SF test.
Oropharyngeal dysphagia is a frequent condition in elderly patients. It can affect up to 40% of all subjects over 65 years of age and 60% of all institutionalized elderly people.16 Among elderly patients admitted to hospital due to acute disease, the prevalence of OD can reach 47.4%.1 This prevalence may further increase to 75% when OD is assessed by videofluoroscopy in patients admitted due to pneumonia.17 Our recorded prevalence was 56.7%. The prevalence of OD in our study is probably similar to that found in previous studies, because the EAT-10 test, as a screening tool, detects a greater number of cases of OD than when videofluoroscopy, which is the confirmatory test, is used.5 Galán Sánchez-Heredero et al.18 found that 88.9% of the patients with positive EAT-10 screening for OD finally presented the disorder according to the V-VST test. Considering the above data, the prevalence of OD in our study was probably around 45.6%.
Oropharyngeal dysphagia is a serious condition in the elderly population, being associated with risk factors such as advanced age and high comorbidity.1,18 Age is identified as a clear risk factor for OD in the vast majority of studies conducted in patients with advanced chronic diseases.1,2,19,20 We found most patients admitted to the Internal Medicine unit of our hospital to have a high CCI score (mean score=4.3; SD: 2.38). The CCI score was identified as a predictor of mortality risk in the multivariate analysis (HR: 1.17; 95%CI: 1.04–1.32). Other studies19 have shown that the CCI score indeed influences mortality in patients with OD, increasing the risk 5-fold (ORCharlson>5=5.16; 95%CI: 1.32–20.07). The prognostic implications of the presence of OD in hospital are significant, as the condition is associated with patient functional decline and malnutrition, as well as with prolonged hospital stay and increased healthcare costs.2,4,15,19–21
Oropharyngeal dysphagia is one of the most underdiagnosed problems among elderly patients, and with the greatest impact upon malnutrition.2,4,17,19 A total of 88.2% of the patients at risk of developing OD as identified by the EAT-10 test had malnutrition or a risk of malnutrition according to the MNA-SF in our study. This finding agrees with the data reported in other studies, which estimated the prevalence of nutritional problems to be over 70% in patients with OD.18,22 Likewise, the presence of OD has been shown to be an independent predictor of malnutrition, increasing the risk of nutritional problems up to 8-fold in the study published by Galán Sánchez-Heredero et al.18
Different studies have shown that malnutrition, in addition to having serious implications for patient recovery from disease, is associated with increased hospital stay and greater long-term mortality.1,23 In our study the group of patients with malnutrition according to the MNA-SF suffered increased mortality over follow-up, though statistical significance was lost in the multivariate analysis adjusted for the EAT-10. Other studies, such as that published by Carrión et al.,1 showed malnutrition according to the Mini-Nutritional Assessment test (score <17 points) to be an independent risk factor for mortality one year after hospital discharge (OR: 1.7; 95%CI: 1.2–2.4). Likewise, the authors of this study were able to establish a relationship between the presence of OD and mortality at one year of follow-up (OR: 1.68; 95%CI: 1.2–2.3).1 Other studies have shown that the presence of OD is indeed an independent predictor of mortality risk.3 In our study we found the presence of OD risk according to the EAT-10 test to be an independent predictor of long-term mortality in the multivariate analysis adjusted for the CCI score, age, the MNA-SF and cognitive impairment.
Oropharyngeal dysphagia has been recognized as a geriatric syndrome by different European medical bodies.4 A geriatric syndrome is a clinical condition highly prevalent in the elderly population, associated with multiple comorbidities, and implying clearly reduced survival. A multidisciplinary approach is required for the management of OD.4 The early detection of OD has been shown to improve the nutritional status and functional capacity of subjects with OD, as well as reducing the incidence of pneumonia, hospital costs, and both in- and out-hospital mortality.24 For the correct identification of the disorder, it is important to introduce a protocolized program for the detection and management of OD in a setting such as ours, where the condition is highly prevalent. Some authors propose screening for OD in elderly patients admitted due to acute disease, particularly in those with suspected community-acquired pneumonia, where inadvertent aspirations may be present.17 Aspirations are often silent and may not be obvious when the patient is evaluated during the swallowing process.5 Screening for OD therefore should be rapid, inexpensive, and easy to apply in order that most of the individuals with this disorder can be detected. Videofluoroscopy is the gold standard for the diagnosis of OD, but its use is not feasible in all clinical settings, since it requires specific equipment and is only found in some tertiary hospitals.4,5 The EAT-10 test is an adequate and simple tool that is useful for detecting swallowing disorders. Its universal application would help in detecting those individuals at risk of malnutrition and increased long-term mortality, as evidenced in our study. Since its development by Belafsky et al.7 in 2008, a number of publications have questioned certain aspects of the EAT-10 test.8,25 Although a cut-off score of 3 was initially established by Belafsky et al.7 as indicating a risk of OD, authors such as Rofes et al.8 have suggested that a score of 2 would be a more adequate cut-off point, after contrasting the results of the EAT-10 test with those obtained by videofluoroscopy. In this way the sensitivity of the test appears to increase by 5%, with a corresponding increase in its negative predictive value, and with no impact upon its specificity.8 In our study the patients were found to be at risk of OD if they had an EAT-10 score of 3 or higher, in concordance with Belafsky et al.7 Our study was not intended to assess the ability of the EAT-10 test to detect OD risk. However, we found the EAT-10 test to be an independent predictor of mortality in the multivariate analysis. It would be interesting to establish whether the application of a score of 2 to discern individuals at risk of OD would also be associated with increased mortality. On the other hand, although the EAT-10 test appears to have adequate internal consistency and good reproducibility,7,10 some authors25 have proposed a modification of the evaluated items due to the impact current items may have on internal consistency and the ability to specifically differentiate groups of subjects with different OD scales.
The limitations of our study include its small number of patients and our not having compiled other variables that could have acted as confounding factors, such as performance status measured by the Barthel index and laboratory parameters. We likewise could not establish the true prevalence of OD in the Internal Medicine ward of our hospital, since screening was not performed systematically in all new admissions. The retrospective nature of the study limited the possibility of analyzing the occurrence of new bronchoaspiration episodes in the presence of positive screening with the EAT-10, as well as adjustments for laboratory variables. The causes of mortality likewise could not be ascertained. However, the advantages of the study are its prolonged follow-up, with only a single loss, and the fact that we were able to adjust for the main confounders, such as malnutrition and the CCI score.
In conclusion, early screening for OD risk using the EAT-10 test in elderly patients hospitalized due to acute disease contributes to the detection of a population at risk of denutrition related to OD and increased long-term mortality. We recommend systematic use of the EAT-10 questionnaire as a useful tool for the screening of OD.
Conflicts of interestThere are no conflicts of interest.
Please cite this article as: Mañas-Martínez AB, Bucar-Barjud M, Campos-Fernández J, Gimeno-Orna JA, Pérez-Calvo J, Ocón-Bretón J. Asociación de un cribado positivo para disfagia con el estado nutricional y la mortalidad a largo plazo en pacientes ancianos hospitalizados. Endocrinol Diabetes Nutr. 2018;65:402–408.