The aim of this study is to present our patients with lung cancer and synchronous adrenal metastases treated with a reversal approach: starting with adrenalectomy and doing the lung resection second.
MethodsA total of 108 laparoscopic adrenalectomies were performed, and we analyze a consecutive series of 10 patients with isolated adrenal synchronous metastases from the lung, surgically treated in a sequential way. All patients underwent staging mediastinoscopy, and patients with positive lymph nodes were primary treated with chemotherapy. We analyze: postoperative morbidity, length of stay, time between the 2 surgeries, survival free progression and global survival. Survival analysis was performed by the Kaplan–Meier method.
ResultsMean age: 56.8 (41–73) years old. Of the total, 8 patients were surgically performed by laparoscopy. Metastases average size: 5.9 (3–10)cm. Days between the 2 surgeries were 28 (12–35) days. No morbidity after adrenalectomy. Length of stay was 4.3 (3–5) days. Disease-free survival at 2 years was 60%, the 5-year overall survival was 30%, with a median survival of 41.5 (0–98) months.
ConclusionsAdrenalectomy involves no significant morbidity and can be performed safely without delaying lung surgery, and allows us to operate the primary lung tumor successfully as long as we ensure complete resection of the adrenal gland. A multidisciplinary oncology committee must individualize all cases and consider this therapeutic approach in all patients with resectable primary tumor and resectable adrenal metastases.
El objetivo de este estudio es presentar nuestra serie de pacientes con carcinoma pulmonar y metástasis única suprarrenal sincrónica tratados de manera secuencial inversa: primero adrenalectomía y después resección pulmonar.
MétodosDel total de 108 adrenalectomías laparoscópicas transperitoneales realizadas se analiza de manera retrospectiva una serie consecutiva de 10 pacientes diagnosticados de tumor primario de pulmón con metástasis suprarrenal sincrónica tratados de manera secuencial inversa. A todos se les realizó mediastinoscopia de estadificación; aquellos con metástasis ganglionares recibieron inducción. Las variables analizadas fueron: morbilidad tras adrenalectomía y tras resección pulmonar, estancia hospitalaria, tiempo entre ambas intervenciones, intervalo libre de enfermedad y supervivencia global. La supervivencia se analizó según el método de Kaplan–Meier.
ResultadosEdad media: 56,8 años (rango: 41–73). Del total, 8 casos se intervinieron por laparoscopia. Tamaño medio de la metástasis: 5,9cm (rango: 3–10). Tiempo medio entre ambas intervenciones: 28 días (rango: 12–35). No hubo complicaciones tras la adrenalectomía. Estancia media: 4,3 días (rango: 3–5). La supervivencia libre de enfermedad a los 2 años fue del 50% y la supervivencia global a los 5 años fue del 30%, con una supervivencia global mediana de 41,5 meses (rango: 0–98).
ConclusionesLa adrenalectomía para metástasis de carcinoma pulmonar tiene baja morbilidad, no retrasa la resección del tumor primario y permite realizar la resección pulmonar una vez asegurada la resección completa de la metástasis. Por tanto, a falta de ensayos clínicos, un comité multidisciplinar debe considerar de forma individualizada esta opción terapéutica para todos aquellos pacientes en quienes la estadificación clínica de su carcinoma indique que tanto el tumor primario como la metástasis pueden extirparse de forma completa.
In recent years, it has been demonstrated that local curative treatment of localized metastases provides better survival than systemic treatment with chemotherapy.1,2
Several types of tumor metastasize to the adrenal glands, and in some 40% of cases the origin is non-small-cell lung carcinoma. However, the isolated presentation of adrenal metastasis represents only 1%–4%, and these are the cases that will obtain a greater benefit from surgical resection.3
Proper preoperative diagnostic studies are necessary because the finding of an adrenal mass in patients diagnosed with lung cancer does not necessarily represent a metastasis. Some 5% of the general population have benign adrenal adenomas.4 Taking the definition of pulmonary synchronic metastases as those diagnosed at the same time as primary pulmonary tumors, we present our series of patients treated in inverted sequence: first the metastasis, and then the primary tumor.
MethodsWe present a descriptive study of a consecutive series of 10 patients with synchronous adrenal metastases of pulmonary origin as the only metastatic site. The patients were treated surgically in reverse sequential order at a single hospital from 1991 to 2010. Our initial experience in 5 patients was previously published.5
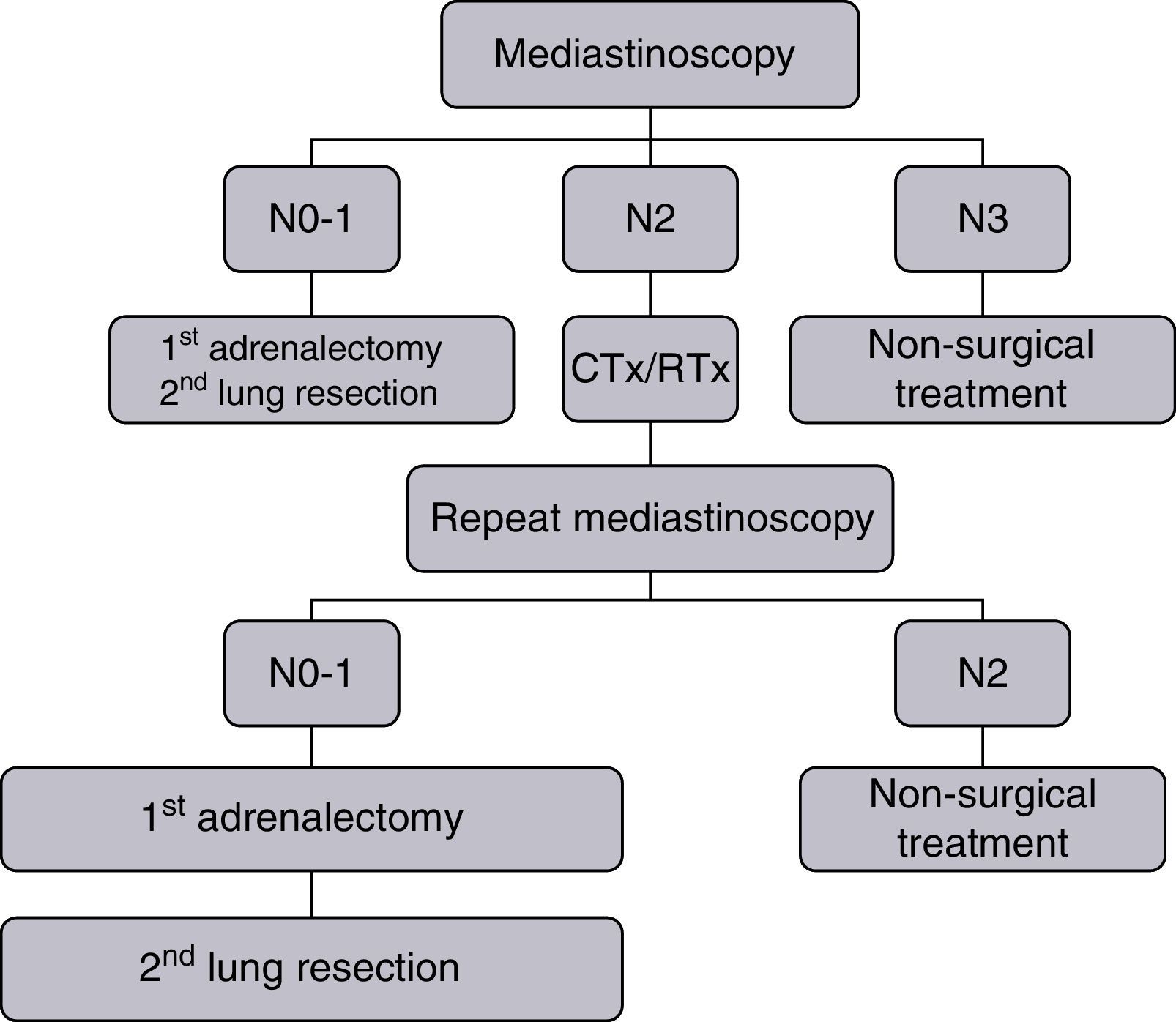
Clinical staging was performed with posterior–anterior and lateral chest radiographs, blood count and blood biochemistry (including at least glycemia, total bilirubin, liver enzymes, calcium, creatinine and alkaline phosphatase), fiberoptic bronchoscopy, and computed tomography (CT) scans of the chest and upper abdomen, both with contrast. From 1991 to 2000, bone scans were performed systematically. As of 2001, these were replaced by systematic positron emission tomography (PET) scans. All patients with right lung carcinomas underwent staging mediastinoscopy, with left parasternal mediastinotomy or extended cervical mediastinoscopy if the lung carcinoma was on the left. Patients with mediastinal lymph node metastases received induction therapy, after which they were reassessed with a second mediastinoscopy to establish or rule out the definitive surgical indication, as shown in Fig. 1.
Operability was assessed with anamnesis and physical examination, electrocardiogram, spirometry and diffusing capacity of the lung for carbon monoxide (DLCO). Those patients with a forced expiratory volume in the first second (FEV1) or DLCO below 80% were studied with a 6-min walk test or by climbing stairs; if results were suboptimal, the study was completed with ergometer testing. Quantitative lung perfusion scintigraphy was performed in those patients with predicted postoperative FEV1 or DLCO of less than 35%.
Adrenalectomy was performed using a transperitoneal laparoscopic approach in all patients, except for the first two patients of the series who underwent surgery in 1991 and 1997. Patients were placed in lateral decubitus position 90° to the surgical table, and 3 or 4 trocars were used according to whether the adrenalectomy was left or right.
Lung resection was conducted by posterolateral thoracotomy and accompanied by systematic lymph node dissection.
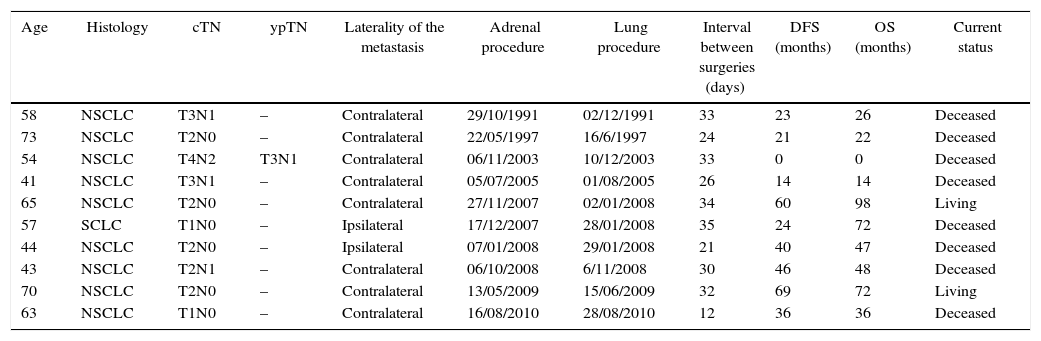
The variables analyzed were postoperative morbidity after adrenalectomy and lung surgery, hospital stay, time between surgeries, disease-free interval and overall survival (Table 1).
Characteristics of Patients, Tumors, Survival and Current Status.
| Age | Histology | cTN | ypTN | Laterality of the metastasis | Adrenal procedure | Lung procedure | Interval between surgeries (days) | DFS (months) | OS (months) | Current status |
|---|---|---|---|---|---|---|---|---|---|---|
| 58 | NSCLC | T3N1 | – | Contralateral | 29/10/1991 | 02/12/1991 | 33 | 23 | 26 | Deceased |
| 73 | NSCLC | T2N0 | – | Contralateral | 22/05/1997 | 16/6/1997 | 24 | 21 | 22 | Deceased |
| 54 | NSCLC | T4N2 | T3N1 | Contralateral | 06/11/2003 | 10/12/2003 | 33 | 0 | 0 | Deceased |
| 41 | NSCLC | T3N1 | – | Contralateral | 05/07/2005 | 01/08/2005 | 26 | 14 | 14 | Deceased |
| 65 | NSCLC | T2N0 | – | Contralateral | 27/11/2007 | 02/01/2008 | 34 | 60 | 98 | Living |
| 57 | SCLC | T1N0 | – | Ipsilateral | 17/12/2007 | 28/01/2008 | 35 | 24 | 72 | Deceased |
| 44 | NSCLC | T2N0 | – | Ipsilateral | 07/01/2008 | 29/01/2008 | 21 | 40 | 47 | Deceased |
| 43 | NSCLC | T2N1 | – | Contralateral | 06/10/2008 | 6/11/2008 | 30 | 46 | 48 | Deceased |
| 70 | NSCLC | T2N0 | – | Contralateral | 13/05/2009 | 15/06/2009 | 32 | 69 | 72 | Living |
| 63 | NSCLC | T1N0 | – | Contralateral | 16/08/2010 | 28/08/2010 | 12 | 36 | 36 | Deceased |
NSCLC: non-small-cell lung cancer; OS: overall survival; SCLC: small-cell lung cancer; DFS: disease-free survival.
This is a longitudinal descriptive study analyzed retrospectively. The survival analysis was calculated with the Kaplan–Meier method (SPSS v.15).
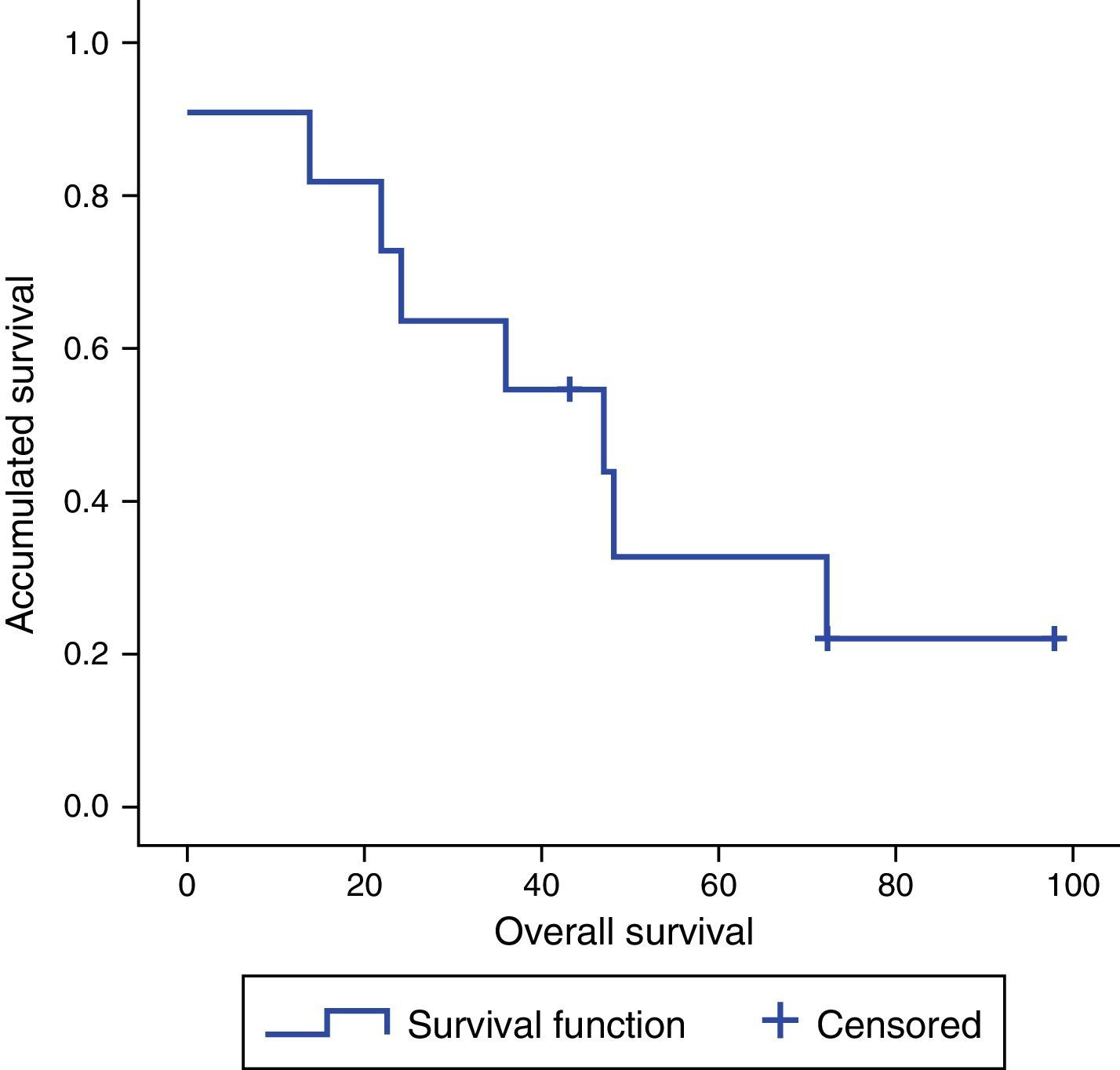
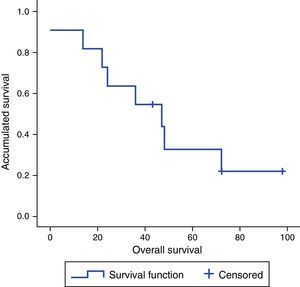
ResultsThe 10 patients included 9 men and one woman, with a mean age of 56.8 years (range: 41–73); 8 were treated by laparoscopy and 2 by laparotomy (the first 2 of the series). In 8 cases, the metastasis was contralateral and, in 2, ipsilateral to the lung tumor. One patient had isolated adrenal metastasis with N2 mediastinal lymph node involvement (cT4N2) at the time of diagnosis; after neoadjuvant CTx/RTx and correct downstaging (T3N1), surgery was performed. None of the patients presented complications in the immediate postoperative period after adrenalectomy. Mean hospital stay was 4.3 days (range: 3–5). The mean size of the metastases was 5.9cm (range: 3–10), and the mean time interval between the two surgeries was 28 days (range: 12–35). All patients were operated on for both the metastasis and primary tumor within 6 weeks. Resections of the lung tumors and adrenal metastases were complete. Histology studies showed non-small cell lung carcinoma in 9 patients (5 adenocarcinomas, 2 large-cell, one epidermoid and one sarcomatoid) and one patient diagnosed with a carcinoid tumor. There was no postoperative mortality following adrenalectomy. One patient died after lung surgery when nosocomial pneumonia did not respond to antibiotic treatment, requiring ICU admission with orotracheal intubation; this finally led to septic shock and death. As for survival, if all patients in the series are included, we obtain a median disease-free survival of 30 months (range: 0–98), an overall 3-year survival of 60% and 5-year survival of 30%, with a median survival of 41.5 months (range: 0–98). If we exclude from the analysis the patient diagnosed with a carcinoid tumor, we obtain a median disease-free survival of 36 months (range: 0–98), an overall 3-year survival of 60% and 5-year survival of 20%, with a median survival rate of 36 months (range: 0–98) (Fig. 2).
DiscussionClassically, patients diagnosed with primary lung tumors and distant disease were treated systemically, with median survivals that did not exceed 11 months.6 However, it has been demonstrated that those patients with resectable extrathoracic disease present longer survivals when treated locally than when treated systemically.7,8
Although the concept of oligometastases has aroused interest in recent times, it was described more than 20 years ago by Hellman and Weichselbaum, who suggested that the number of metastases was a reflection of the biological behavior of the tumor and determined the possibility of a cure with surgical treatment.9 Despite the increase in published articles, the majority are retrospective studies with heterogeneous definitions for the number of metastases (from a single lesion up to 5) and therapeutic management. Thus, in the absence of a homogeneous definition, an update of the M category has recently been published with a view to the upcoming 8th edition of the TNM lung classification, in which patients with a single metastatic lesion in a single organ (brain, liver, bone, lymph nodes, skin, peritoneum and adrenal gland) are considered M1b.10
If we focus on the adrenal location, surgical treatment of isolated adrenal metastases has gained popularity in recent years, which confirms a change in therapeutic strategy, even though it was first described in 1996 by Luketich and Burt.11
The incidence of isolated adrenal metastasis in patients with resectable lung tumors is low and it is these patients who will most benefit in the long term from surgical treatment. Numerous articles have shown this, with longer survivals when adrenalectomy is performed.12–19
At our hospital, we advocate this surgical line of treatment, but when the adrenal metastasis is synchronous, we are in favor of inverting the surgical order by first performing adrenalectomy and then lung resection. In the literature, there have been no reports of the standardized practice of this inverse sequential treatment that we propose.
On the one hand, we believe that not achieving complete resection in the extrathoracic disease limits the pulmonary resection, as the patient would not benefit from primary lung tumor resection in terms of survival. And, on the other hand, the morbidity of lung surgery is superior to the morbidity of adrenalectomy, which could delay or even impede the surgery of the metastasis if the patient becomes inoperable due to altered lung function. Thus, as demonstrated in our series, all patients were operated on for both the adrenal metastasis and the primary tumor, with a mean time interval between both surgeries of less than 30 days.
We believe that preoperative staging is essential when determining the therapeutic strategy. Mediastinal staging can involve either non-invasive (CT/PET) or invasive tests (fiberoptic bronchoscopy, mediastinoscopy, endobronchial ultrasound). At our hospital, we performed mediastinoscopy for both staging and re-staging after induction chemotherapy (cisplatin+etoposide or cisplatin+vinorelbine) and concomitant radiotherapy, since the persistence of mediastinal lymph node involvement after induction would contraindicate surgical resection of any kind. In our series, there was one patient who, at the time of diagnosis, had N2 mediastinal lymph node involvement and who, after chemoradiotherapy and with correct downstaging, could be operated on.
On the other hand, not all adrenal masses are necessarily metastases, so complete extension studies are necessary. Imaging techniques such as nuclear magnetic resonance or PET both have high sensitivity and specificity, which make a correct diagnosis possible without the need for biopsy of the adrenal mass.
Median overall survival published in this type of patients is 12 months; however, in our series, we obtained a median survival of 41.5 months (range: 0–98), and 36 months (range: 0–98) if we exclude the carcinoid tumor from the series and only analyze patients with non-small-cell lung cancer. Despite the good results, the low number of patients should make us interpret these survival data with caution and compare them to future studies with larger patient samples.
From this small series, we can conclude that adrenalectomy for synchronous metastasis of lung carcinoma presents, firstly, low morbidity without delaying the resection of the primary tumor. Second of all, it allows the pulmonary resection to be performed once complete resection of the metastasis has been achieved. Therefore, those patients in whom both the primary tumor as well as the metastasis can be completely removed will be able to benefit from this therapeutic strategy.
Conflict of InterestThe authors have no conflict of interest to declare.
Please cite this article as: Pardo Aranda F, Larrañaga Blanc I, Rivero Déniz J, Trujillo JC, Rada Palomino A, García-Olivares E, et al. Tratamiento quirúrgico de metástasis suprarrenal sincrónica de cáncer de pulmón: adrenalectomía primero. Cir Esp. 2017;95:97–101.