Ossifying fibromyxoid tumors are a type of myxoid soft-tissue tumors, which are a heterogeneous group of lesions that produce an extracellular matrix rich in mucopolysaccharides. These tumors are rare (less than 300 reported cases) and have slow growth with great capacity for local recurrence and metastasis.
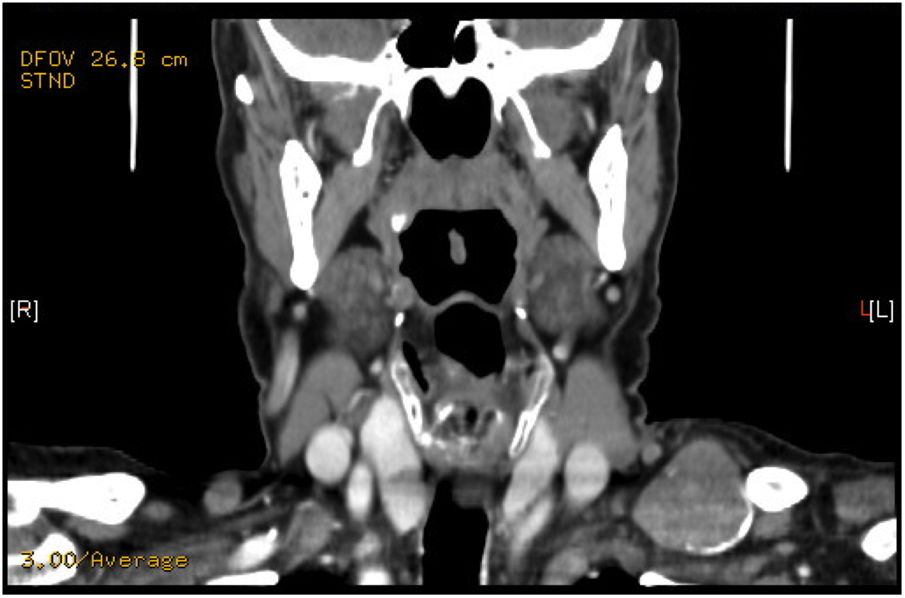
We present the case of a 74-year-old male who presented with a supraclavicular tumor that had recently appeared, measuring 4 cm in diameter. During examination, the lesion was painless, hard and partially mobile. On ultrasound, a solid-cystic nodule measuring 32 mm appeared to be in contact with the clavicle. FNA biopsy results reported a neoplasm that was probably neuroendocrine. The hormone study was within normal ranges. CT scan revealed a supraclavicular lesion measuring 3 cm in the left posterior cervical triangle (Fig. 1), and the SPECT-CT showed a focal deposit with moderate-to-high density in the left supraclavicular fossa. During surgery, we observed a mass measuring 4 cm in diameter in the supraclavicular space, extending behind the clavicle and hard in consistency, with areas of calcification. Surgical excision was performed. The patient evolved favorably and was discharged 24 h later.
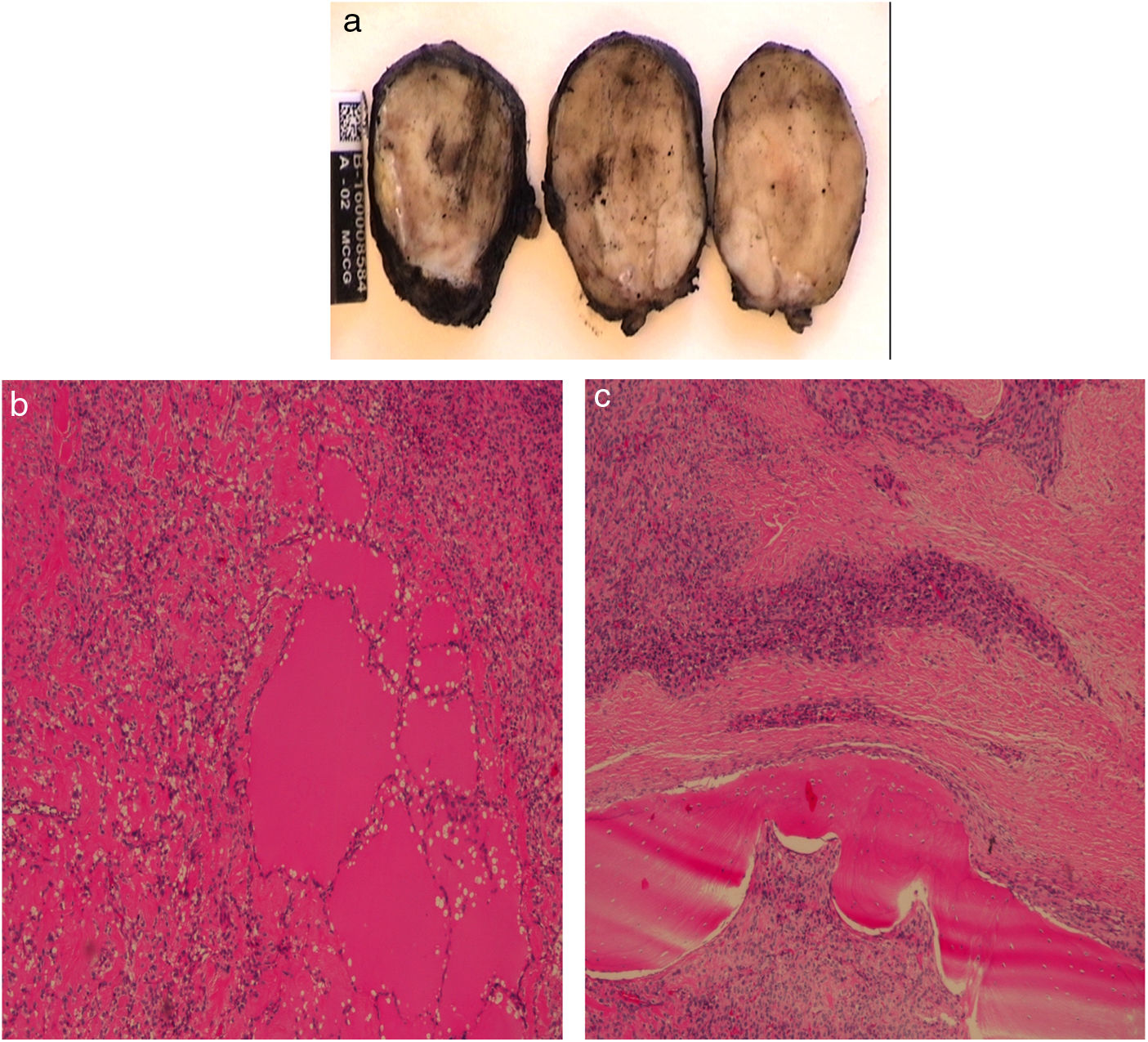
The pathology results reported an ovoid mass measuring 4 × 4 × 2.5 cm, corresponding with a pseudo-encapsulated multinodular mesenchymal tumor (Fig. 2A). The lesion was hypercellular with collagenized areas, epithelioid habit, moderate eosinophilic cytoplasm and irregular nuclei. It presented 5 mitoses/50 HPF (reaching 2–3/10 HPF in certain areas) and areas of ossification in concentric rings. Myxoid-looking foci were identified, which were compatible with malignant calcifying fibromyxoid tumor (Fig. 2B and C). The tumor committee decided to administer adjuvant radiotherapy, and the patient remains disease-free after 2 years of follow-up.
Ossifying fibromyxoid tumors occur in adults of any age, especially in men around the age of 50.1 Affecting the subcutaneous soft tissue or skeletal muscles, 70% appear in the extremities (more proximal than distal), with cases described in the trunk, head, neck, mediastinum, retroperitoneum and breasts. These tumors usually present as small nodules, with an average size of 4−5 cm1,2; they are well circumscribed, painless and deep, attached to the underlying tendons, fascia or skeletal muscles. They usually show growth from months to years of duration (mean 4 years).1
Based on 59 cases at the Armed Forces Institute of Pathology over the course of 25 years, Enzinger et al.3,4 described this entity for the first time in 1989 as a well-circumscribed nodular mass in the subcutaneous tissue or muscles, calling it ossifying fibromyxoid tumor of soft parts.
The vast majority of these lesions are histologically benign and have a favorable prognosis.5 However, there are cases with atypical cytoarchitecture characteristics and histological features of malignancy (high cellularity, nuclear atypia and high mitotic activity >2/50 HPF), which present greater recurrence and capacity to metastasize (60% malignant).4,6 The existence of small white cell ‘cords’ with a matrix rich in mucopolysaccharides and frequent parietal hyalinization is characteristic.3 These tumors may express bone tissue in 60%-90% of cases and necrosis in up to 20%.7
There is no universally accepted risk classification. Folpe and Weiss2 have described 70 cases, divided into 3 types. “Typical” tumors are those with low cellularity and nuclear grade, lack of necrosis or vascular invasion, with mitotic activity <2/50 HPF. “Atypical” lesions differ from the typical, but show no criteria for malignancy. “Malignant” tumors have a recurrence and metastasis rate of 60%. In their published series, 8 patients out of 70 had distant metastases (mainly lung).2,7
Regarding immunohistochemistry, these lesions express S100 protein, especially in “typical” tumors.5,8 Antonescu et al.8 observed S100 expression in 60% of cases and positivity for EAAT4, MUC4, NFP, CD56 and CD10 in “atypical” tumors. Graham et al.9 described a characteristic mosaic pattern of loss of INI1/SMARCB1. In a small number of cases, the INI1 gene is absent, corresponding with the loss of nuclear expression.7 Studies with FISH have found a recurrent rearrangement of the PHF1 gene (located on chromosome 6p21) in 49%-79% of cases.
Radiologically it behaves like a soft tissue nodular mass with an incomplete ossification ring. The CT scan shows a peripheral “bone shell” in 60%-70% of cases.1,2,5 Technetium tests demonstrate the presence of intratumoral formations of mature bone.1 On MRI, the tumor is usually isointense to the muscles in T1 and shows intermediate-high intensity in T2.10
Treatment usually involves complete resection of the lesion with free margins, which is curative in most cases.4 However, since the malignant variant has a risk of recurrence and metastasis, adjuvant radiotherapy of the tumor bed is added.
Malignant ossifying fibromyxoid tumors are very rare. Due to the high rate of recurrence and metastasis, their diagnosis by histological, immunohistochemical and FISH methods is very important. A better knowledge of these tumors is necessary in order to avoid errors in their diagnosis and to contribute to their correct management, thereby increasing survival.
Please cite this article as: Jiménez Gómez M, Caballero Díaz Y, Gutierrez Giner I, Camacho MC, Hernández Hernández JR. Tumor fibromixoide osificante maligno supraclavicular. Cir Esp. 2020;98:107–109.