Associating Liver Partition and Portal vein ligation for Staged hepatectomy (ALPPS) is a novel surgical technique that provides fast and effective growth of liver remnant volume, allowing surgical resection of hepatic lesions initially considered unresectable.
Short and long-term results and the convenience of carrying out this technique are issues that still remain under debate while waiting for the final outcomes of the multicenter registries with larger number of cases.
The aim of this paper is to describe, from a critical point of view, the outcomes of the cases performed at our centre (n=8).
On the other hand, it is possible to leave only one hepatic segment as a liver remnant and we illustrate this new surgical procedure (ALPPS monosegment) performed in one patient.
La hepatectomía secuencial, descrita en la literatura anglosajona con el acrónimo ALPPS (Associating Liver Partition and Portal vein ligation for Staged hepatectomy) es una técnica novedosa que ofrece un crecimiento rápido y efectivo del volumen remanente hepático, y que permite la resección quirúrgica de lesiones hepáticas consideradas inicialmente irresecables.
Los resultados a corto y largo plazo y la conveniencia de realizar esta técnica son cuestiones que permanecen en discusión a la espera de los resultados finales de los registros multicéntricos.
El objetivo del presente trabajo es la revisión crítica de los resultados de la serie de casos realizados en nuestro centro (n=8).
Por otra parte, es posible con esta técnica dejar un único segmento hepático como remanente y realizamos una descripción de esta variante técnica novedosa (ALPPS monosegmento), llevada a cabo en uno de los casos.
The curative treatment of most primary or metastatic malignant hepatic tumours requires surgical resection.
In the case of hepatic metastases of colorectal carcinoma (CRC), the definition of resection involves the fulfilment of 2 conditions: complete oncologic resection with free margins (R0) and sufficient functional liver remnant to prevent postoperative hepatic failure.1 This last aspect is more important in patients subject to preoperative chemotherapy or underlying liver disease.
Two-stage surgical resection and preoperative portal embolisation (PPE) are broadly developed strategies which help to increase the rate of resection of hepatic lesions. Moreover, a new surgical technique, initially described by Schnitzbauer et al.,2 known as ALPPS (Associating Liver Partition and Portal vein ligation of Staged hepatectomy), has been developed. This consists of conducting, during the first stage, the portal occlusion of the hemi-liver which is to be resected, together with a hepatic parenchymal transection by associating, if necessary, the resection of contralateral lesions and completing, during the second stage, the resection of the embolised liver.
The most evident advantage of this technique is its demonstrated capacity for achieving rapid and effective hepatic regeneration within a short period of time. On the other hand, its main disadvantage is a high morbidity and mortality rate, with figures usually higher than those published in hepatic resection series over recent years. For this reason some authors do not recommend the use of this technique.
The objective of this study is to publish the results obtained in a series of patients who underwent this procedure in the Hepato-Bilio-Pancreatic Surgery and Transplantation (HPB) Unit of our centre (n=8) and to show, by means of a clinical case, a more recent variant of this technique.
Material and MethodA series of 8 patients were operated on in the HPB Surgery Unit using the ALPPS technique between January 2012 and April 2013. In the first case (January 2012), an original technical variant was implemented, and only one segment was left as liver remnant.
Quantitative variables were described as mean and standard deviation values or median and range values. For categorical variables, percentages were used.
Postoperative complications were described using the Clavien-Dindo classification.3 The International Study Group of Liver Surgery (ISGLS)4 definition of postoperative hepatic failure was used.
ResultsOf the 8 operated cases, CRC hepatic metastases were diagnosed in 7 (87.5%), and healthy-liver fibrolamellar hepatocarcinoma was diagnosed in one case (12.5%).
The median age was 59 years (23–67), and there were 5 male patients and 3 female patients. The surgical risk scale assessed using the classification of the American Society of Anaesthesiology (ASA) was ≤2 in all cases. The most relevant characteristics of the series are shown in Table 1. About 85.7% (6/7) of patients with hepatic metastases were treated with neoadjuvant chemotherapy, except for the case with third recurrence in which, due to the low tumour load (only one lesion) and previously administered chemotherapy, the multidisciplinary committee decided to administer adjuvant chemotherapy after the surgical resection.
Characteristics of the Patients From the Series.
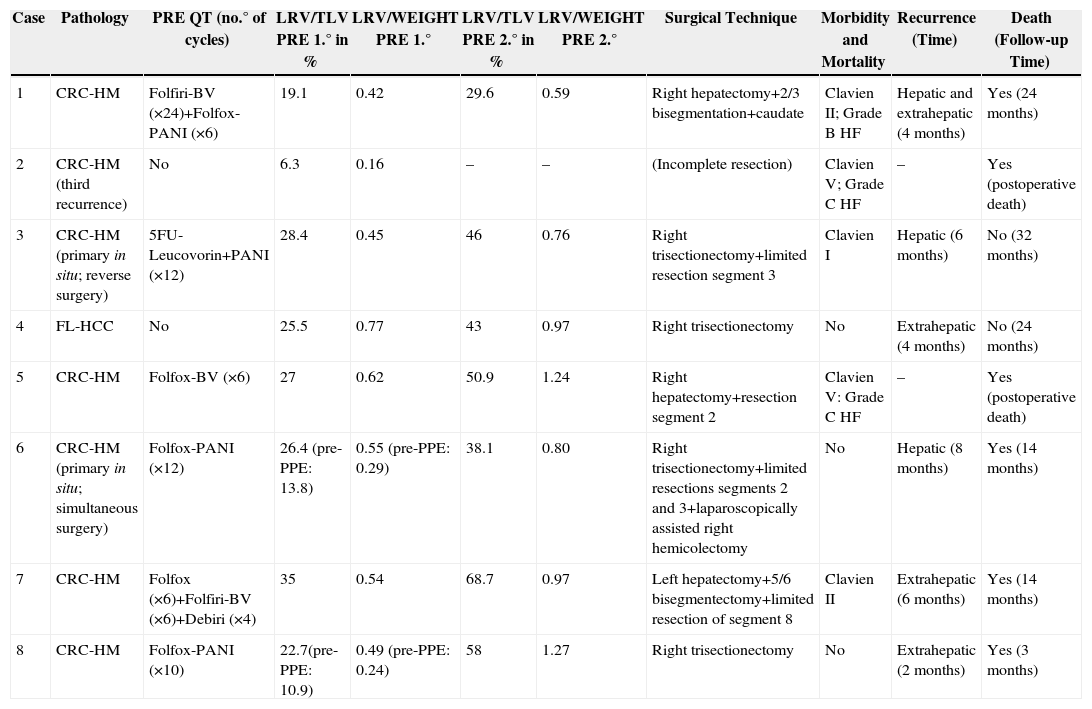
| Case | Pathology | PRE QT (no.° of cycles) | LRV/TLV PRE 1.° in % | LRV/WEIGHT PRE 1.° | LRV/TLV PRE 2.° in % | LRV/WEIGHT PRE 2.° | Surgical Technique | Morbidity and Mortality | Recurrence (Time) | Death (Follow-up Time) |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | CRC-HM | Folfiri-BV (×24)+Folfox-PANI (×6) | 19.1 | 0.42 | 29.6 | 0.59 | Right hepatectomy+2/3 bisegmentation+caudate | Clavien II; Grade B HF | Hepatic and extrahepatic (4 months) | Yes (24 months) |
| 2 | CRC-HM (third recurrence) | No | 6.3 | 0.16 | – | – | (Incomplete resection) | Clavien V; Grade C HF | – | Yes (postoperative death) |
| 3 | CRC-HM (primary in situ; reverse surgery) | 5FU-Leucovorin+PANI (×12) | 28.4 | 0.45 | 46 | 0.76 | Right trisectionectomy+limited resection segment 3 | Clavien I | Hepatic (6 months) | No (32 months) |
| 4 | FL-HCC | No | 25.5 | 0.77 | 43 | 0.97 | Right trisectionectomy | No | Extrahepatic (4 months) | No (24 months) |
| 5 | CRC-HM | Folfox-BV (×6) | 27 | 0.62 | 50.9 | 1.24 | Right hepatectomy+resection segment 2 | Clavien V: Grade C HF | – | Yes (postoperative death) |
| 6 | CRC-HM (primary in situ; simultaneous surgery) | Folfox-PANI (×12) | 26.4 (pre-PPE: 13.8) | 0.55 (pre-PPE: 0.29) | 38.1 | 0.80 | Right trisectionectomy+limited resections segments 2 and 3+laparoscopically assisted right hemicolectomy | No | Hepatic (8 months) | Yes (14 months) |
| 7 | CRC-HM | Folfox (×6)+Folfiri-BV (×6)+Debiri (×4) | 35 | 0.54 | 68.7 | 0.97 | Left hepatectomy+5/6 bisegmentectomy+limited resection of segment 8 | Clavien II | Extrahepatic (6 months) | Yes (14 months) |
| 8 | CRC-HM | Folfox-PANI (×10) | 22.7(pre-PPE: 10.9) | 0.49 (pre-PPE: 0.24) | 58 | 1.27 | Right trisectionectomy | No | Extrahepatic (2 months) | Yes (3 months) |
BV, bevacizumab; FL-HCC, fibrolamellar hepatocarcinoma; HF, hepatic failure; CRC-HM, colorectal carcinoma hepatic metastases; PANI, panitumumab; WEIGHT, patient weight (kg); PRE 1.°, before the first intervention; PRE 2.°, before the second intervention; pre-PPE, before the percutaneous portal embolisation; PRE QT, preoperative chemotherapy; TLV, total liver volume (cc); LRV, liver remnant volume (cc).
The median liver remnant percentage measured before surgery amounted to 25.9% (6.3–35). The residual volume percentage in the 2 patients with preoperative portal embolisation was 22.7% and 26.4%. The median residual volume ratio related to patient weight during the preoperative period was 0.52 (0.16–0.77), which amounted to 0.49 and 0.55 in patients with preoperative portal embolisation.
The most common surgical technique was right trisectionectomy (n=4; 50%), with or without limited contralateral resection. In 5 cases (62.5%), hepatic resection of liver remnant lesions was carried out during the first stage.
In 7 patients (87.5%), both surgical stages were completed; the median interval of days between the 2 interventions was 10 days (9–27).
The control CT scan was performed in 5 cases 7 days after the first procedure (6–10).° The median remnant volume percentage (n=7) amounted to 46% (29.6–68.7), while the median ratio of remnant volume over patient weight amounted to 0.97 (0.59–1.27). Therefore, the median increase in the liver residual volume was 250cc (105–581), which amounted to 68.6% (44.3–155.5) compared to the preoperative remnant volume, and the median difference ratio of liver remnant volume over patient weight was 0.4 (0.2–0.8).
The mean surgical time was 356.3min (SD 46.6) for the first procedure (n=8) and 101.4min (SD 42.3) for the second procedure (n=7).
In 4 cases (50%), postoperative morbidity was observed. All of these cases suffered from hepatic failure. Two patients (25%) died: one patient died after the first stage (patient with third hepatic recurrence) and another patient died due to intraoperative cardiorespiratory arrest during the second surgical stage. In the 2 deceased patients, who suffered from grade C hepatic failure, the anatomopathological study showed severe steatohepatitis, with grade 2/4 fibrosis in one of them. These histological findings were not observed in any of the other 6 patients of the series.
The median hospital stay was 15.5 days (4–28).
The median follow-up time in patients who completed both surgical stages (n=7) was 444 days (1–798). All patients had recurrence of the disease, with a median disease-free time of 155 days (43–226).
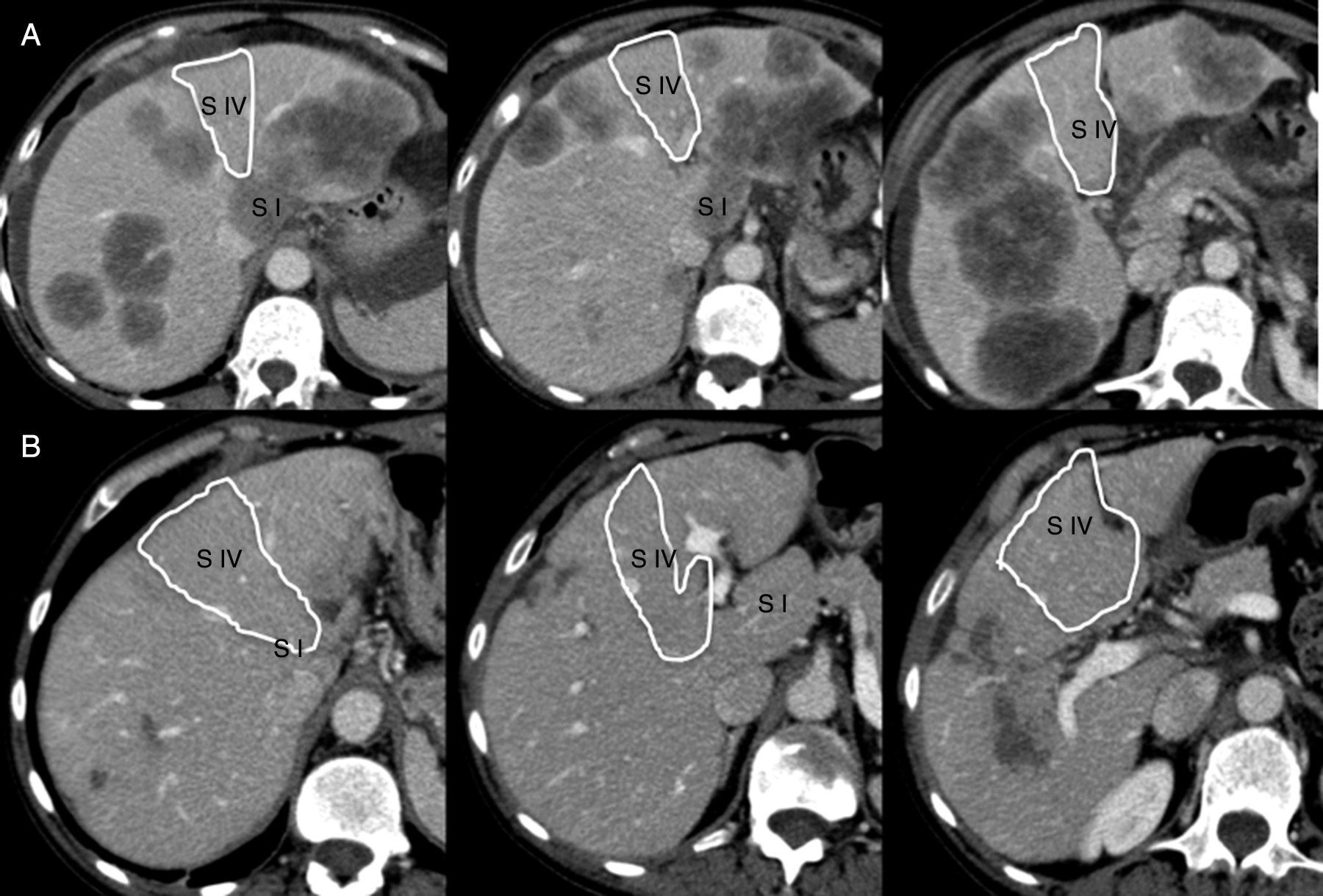
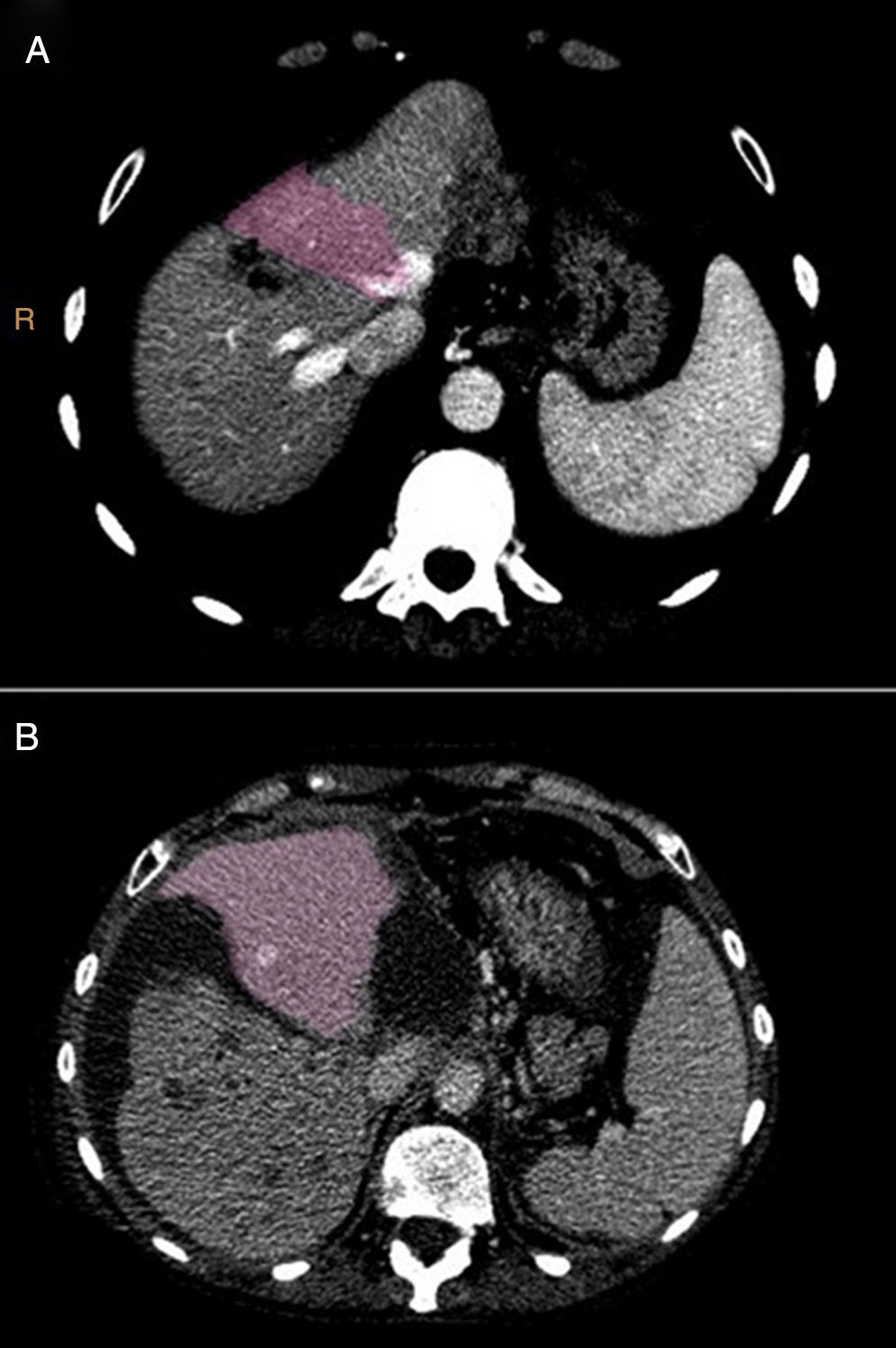
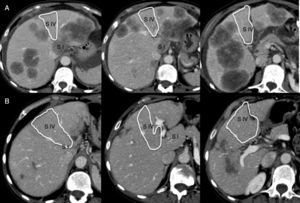
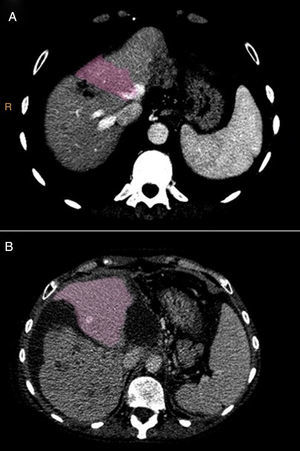
Technical Variant of ALPPS (Monosegment)One of the cases involved a 65-year old female patient (68kg), diagnosed with synchronous hepatic metastases of sigmoid adenocarcinoma, initially irresectable, with partial radiological response to neoadjuvant chemotherapy (FOLFIRI scheme+bevacizumab×30) (Fig. 1). The volumetry showed a total liver volume of 1309cc, with segment 4 and partial segment 1 volume (liver remnant) amounting to 275cc (19.1% of the total, and ratio of liver remnant [cc] over weight of the patient [kg]=0.42).
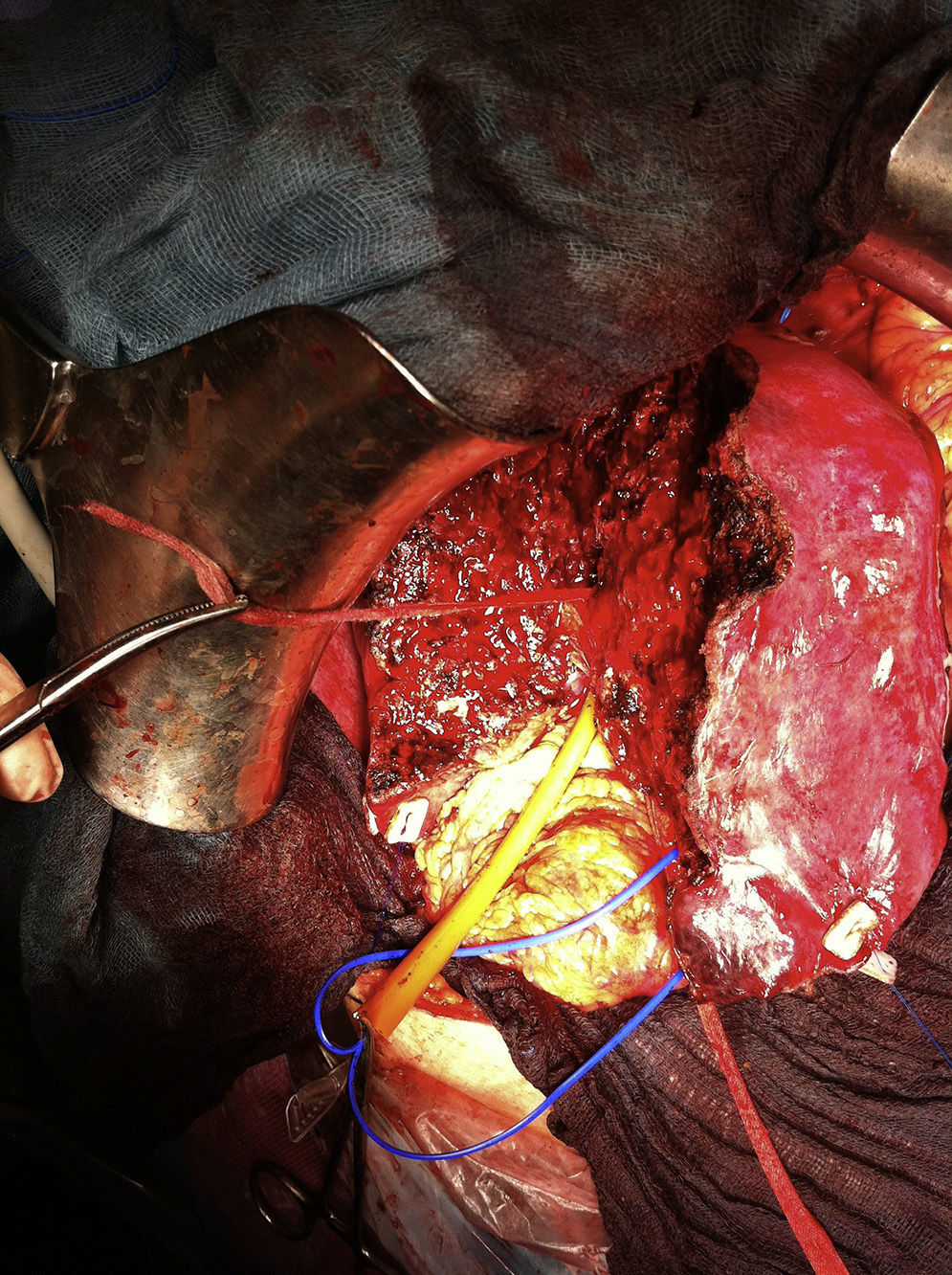
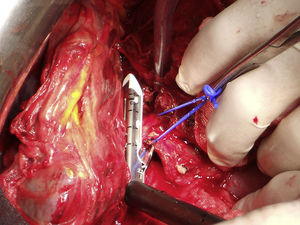
For the scheduled hepatic resection (left lateral sectionometry with caudate segment during the first stage and right hepatectomy during the second stage), an intraglissonian dissection of the vascular elements of the hepatic pedicle was performed. After achieving the complete cleavage of the liver by sectioning direct drainage veins of the vena cava caudate, a cloth tape was used to secure the right (RSV) and middle (MSV) suprahepatic veins for the liver suspension manoeuvre. The tape was replaced on the left of the MSV to facilitate the left lateral sectionectomy and partial caudate resection. The transection was conducted using a CUSA® ultrasonic scalpel and LigaSure® clamp of 5mm, and 3/0 silk suture for the ligature of vascular and bile ducts larger than 2mm in diameter. After resection, the tape was placed on the right side of the MSV and left portal pedicle to align the transection line with the Cantlie line (Fig. 2). For this step, a selective vascular occlusion was conducted with a tourniquet for the right portal pedicle and with a bulldog clamp for the right hepatic artery. If there is risk of damaging the MSV (only drainage vein of the remnant segment) during the parenchymal transection or if the R0 resection cannot be ensured until the end, it is recommended not to section the portal vein until this step is completed, so as to be able to stop the procedure at any time without having conducted an irreversible manoeuvre. Upon proving the viability of segment 4, the right portal branch was proximally bound by distally injecting 15ml of 100% alcohol with low pressure and, finally, the portal vein was sectioned. The right intrahepatic bile duct (not sectioned) and the right hepatic artery were marked with vessel loop. The final left portal flow amounted to 280ml/min.
An intraoperative bolus of somatostatin (500μg i.v.) was administered, as well as continuous perfusion of somatostatin (250μg/h) during the first 5 days.
During the postoperative period, the patient presented grade B hepatic failure.
On the 7th° postoperative day, a dynamic hepatic CT scan with vascular study and volumetry was performed (Fig. 3). The liver remnant volume was 388.1cc (29.6% of the total volume), which represented a 40% increase compared to the initial volume and a ratio of liver volume (cc) over patient weight (kg) of 0.59.
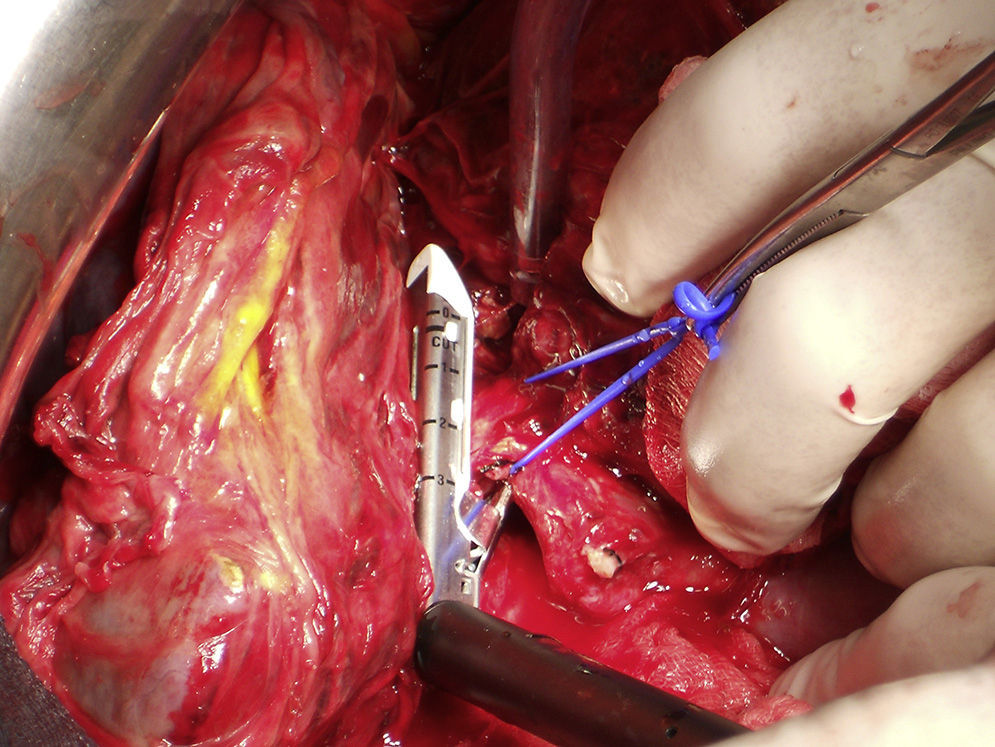
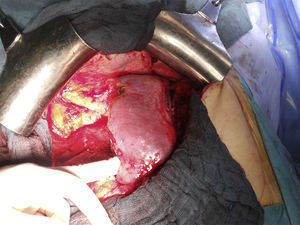
The second laparotomy conducted on the 10th day lasted 110min, in which the right hepatectomy was completed after the ligature of the right hepatic artery and the section of the right biliary pedicle and the RSV using an endo-GIA stapler™ (Figs. 4 and 5).
During the postoperative period of the second intervention, the patient was diagnosed with grade B hepatic failure. The admission lasted 22 days in total.
On the 28th° postoperative day, a control CT scan was performed which showed a liver remnant volume of 595cc, which meant a 53% increase compared to the volume at the time the second surgical stage was performed and a 116% increase compared to the remnant volume calculated at the beginning.
DiscussionHepatic resection surgery has improved considerably over recent decades due to the improvements in surgical material, and anaesthetic and resuscitation techniques, as well as due to the greater understanding of the anatomy and physiology of the liver.
The 2-stage hepatectomy procedure should not be considered simply as a combination of 2 hepatic resections conducted in a sequential manner. Instead, it should be recognised as another example of the multidisciplinary management of these diseases, given that it must be co-ordinated with systemic chemotherapy and portal embolisation.
Most of the candidates for hepatic resection have received preoperative chemotherapy, and the accepted liver remnant volume in these cases must be at least 25% of the total liver volume, with a ratio of liver volume (cc) to patient weight (kg)>0.5,5 increasing by 40% in cirrhotic patients. The remnant volume in patients with cholestasis also ranges between these values. Volumetry does not take into account the hepatic function, which may be altered by the above factors.
In 2007, Dr Hans Schlitt discovered by chance that it was possible to achieve a larger-than-usual liver hypertrophy in a short period of time by performing a parenchymal transection, leaving both hemi-livers in situ and placing the portal ligature on the side that will be removed during the second surgical stage.2 In 2011,6 those results were first published, and in 2012 Santibañes and Clavien suggested the term ALPPS for this procedure, which was adopted in subsequent publications.7
Different variants of the technique have been described,8–10 although all of these respond to the same principle of portal ligature and in situ hepatic bipartition, thus keeping the “auxiliary” liver during the short time in which the liver hypertrophy is performed. However, no evidence suggests that the variant suggested in this work, which could be referred to as monosegment ALPPS, was published. The implementation of this procedure variant, in which only segment 4 is left, requires an exclusive treatment of the inflow and outflow of the liver remnant, preserving the MSV throughout and assessing the final appearance of the parenchyma before deciding on the section of the right portal branch. The volume of segment 4 may be very small in some patients, and the advantage of ALPPS over PPE is the rapid and effective hypertrophy achieved, with the additional possibility of having resected the lesions from the left lateral segment. Therefore, it is a feasible technique, with liver remnant hypertrophy amounting to 40.8% during a 7-day period, in spite of the prolonged treatment (30 cycles) of preoperative chemotherapy.
ALPPS is probably the surgical technique that has caused the greatest controversy in surgical forums in recent years, with almost as many opinions in favour as those against.
The advantages described with this technique are the following:
- 1.
Sufficient hypertrophy of the contralateral liver remnant achieved within a short time. This growth is variable and ranges from 40% to 80% in 7–13 days,11,12 with a mean of 74% in 9 days,2 similar to our records (68.6% in 7 days).
- 2.
Precise knowledge of the extension of the disease, which may be assessed from the beginning by conducting an exploratory laparotomy and an intraoperative ultrasound scan, with the subsequent reduction in the percentage of false negative results obtained by means of preoperative imaging techniques, which makes it possible to modify the treatment strategy for the patient's benefit. In our series, in one patient (12.5%), the surgical technique was modified based on intraoperative findings (case 6).
- 3.
The possibility of performing an extemporaneous liver biopsy for the assessment of the parenchymal affection, which will facilitate the resection decision. In our series, there seems to be a relation between the steatohepatitis finding and postoperative mortality, although the small number of patients does not make it possible to establish an association between these 2 variables. Therefore, these findings may help to reject hepatic resection in these patients.
- 4.
With PPE, the recommended period of time to achieve sufficient liver remnant volume usually ranges from 4 to 8 weeks and, in a percentage of cases (18%–40%),13 resection may not be completed before disease progression. The ALPPS reduces the waiting time and shortens the interval between the 2 surgical resections, so that, in theory, disease progression would be reduced. In our experience, the interval between the 2 interventions was ≤10 days in 71.4% (4/7) of the cases.
- 5.
The portal branches of segment 4 should be embolised in patients who require right trisectionectomy, but it is technically more complicated and not always feasible. In some reports, the role of the ALPPS8 is outlined as a rescue technique when the PPE has not achieved sufficient hypertrophy. In our series, it was necessary to resort to ALPPS in 2 patients with percutaneous PPE (cases 6 and 8). Besides, in cases (such as the one described in this work) where segment 4 is disease-free and, thus, embolisation of only the right portal branch is indicated, the ALPPS seems to be more adequate to achieve sufficient hypertrophy and at the same time resect lesions of the left lateral segment.
Our criteria for using this surgical technique are based on the above advantages. We consider that the ratio of liver remnant (cc) to patient weight (kg) >0.5 is a more reliable value for the identification of patients at risk of severe postoperative hepatic failure than the liver remnant percentage compared to the total liver, particularly in patients with long periods of preoperative chemotherapy (cases 3 and 7). In cases of bilateral hepatic metastases, the location of remnant lesions in relation to portal branches or suprahepatic veins may be explored with surgery, which allows for rejection in cases of surgical unresectability and avoidance of an unnecessary step, such as PPE (case 5). In some cases, the PPE may be insufficient, particularly if new lesions are discovered during laparotomy. Their resection leads to a decrease in the estimated remnant volume in the preoperative study (case 6).
Moreover, this technique also presents multiple disadvantages:
- 1.
The main disadvantage of this technique is its high morbidity (16%–54%)2,14 and mortality rate (0%–23%).15,16 These figures are significantly high, particularly when compared with the improvements achieved over the last years. It is evident that this technique is indicated for a group of patients with doubtfully resectable lesions and, therefore, results should be compared among similar populations. In our series, the mortality rate amounted to 25% (2/8), a figure considered too high, although it should be noted that this series had very few cases, including patients with doubtfully resectable lesions.
- 2.
The surgical procedure and perioperative care are complex and highly demanding. Thus, it is advisable to conduct them in centres with broad experience in hepatic resections, with consolidated multidisciplinary teams and, therefore, this is not a feasible procedure for all hospitals.
- 3.
The 2-stage standard hepatic resection has achieved a 5-year survival rate in patients ranging from 32% to 64%.17 ALPPS does not yet have enough history to allow for the assessment of long-term survival, but preliminary results indicate high early recurrence rates.12 In patients with CRC hepatic metastases and high tumour load or with multiple factors of bad prognosis, and thus high chances of recurrence, the role of extensive surgical resection is considered.18 Portal embolisation has been identified as one of the factors involved in tumour progression19 and, particularly, in the ALPPS procedure; the short period of time between the 2 interventions would stop the progression from occurring before completing the resection, and it would start after its completion. In our series, all cases presented recurrence during the first year, although there was a response to preoperative chemotherapy. This is a cause of concern and it has led to greater caution when indicating this technique if there are other surgical options available.
Lastly, some of the aspects of the surgical technique should be analysed. The contribution of the ligature and the section of the ipsilateral bile duct to the liver remnant hypertrophy in these cases is unknown. In the initial descriptions of ALPPS, the bile duct was sectioned.20 The bile duct was not ligated in any case in our series, and an adequate hypertrophy was obtained nonetheless. We consider that the risk of having postoperative complications (bile leaks, bilomas and cholangitis) secondary to biliary ligature is high and would complicate the second surgical stage.
The lymphadenectomy of the hepatic pedicle is useful not only for oncological reasons, but also to identify vascular and biliary elements of the hilum. This is not recommended as a routine procedure in the case of hepatic metastases, as it occurs with other lesions, such as cholangiocarcinoma. ALPPS with lymphadenectomy conducted during the first stage could make us consider that this technique is a good treatment option for these patients. In the case of Klatskin tumours or gall bladder tumours, the idea of pedicle lymphadenectomy during the first stage could be inconsistent with the “non-touch” oncological principle, but some solutions applicable to these cases have already been published.21
The risk of portal hyperflow should always be taken into account when conducting extensive hepatic resections. To buffer its consequences and the endothelial lesion of the remnant parenchyma, we administered in all cases a somatostatin bolus at the time of the portal ligature and agreed on its continuous perfusion during the immediate postoperative period. The efficiency of this treatment has not been verified by scientific evidence, but its effects have actually been demonstrated by the reduction of the increased portal flow in the liver remnant.
The additional portal embolisation conducted after portal ligature is a surgical step derived from surgical portal embolisations without transection. This technical detail is not included in previous descriptions of the technique and it is probably unnecessary due to the associated parenchymal transection. We consider that it does not lead to new risks in the procedure and it could contribute to the hypertrophy due to the distal occlusion of portal branches, according to some physiopathological theories on liver hypertrophy in connection with the macrophage interception phenomenon in the embolised hemi-liver due to the foreign body reaction.22
One advantage of ALPPS that will have to be demonstrated in a larger series is the “easiness” with which the second surgical stage is performed. In our series, the mean time of the second procedure was 110min (SD 42.3), similar to the mean time of 152min, with a median of 117min (64–364), described in other series.2 The success of this second resection depends on the first resection. It is essential to achieve complete cleavage of the liver, which should only be bound by vascular pedicles and the bile duct.
Therefore, this surgical technique has led to considerable discussion among surgeons before its results were known, which were based only on information from series with few patients and heterogeneous in both indications and technique. Common sense indicates that the indications and contraindications of this technique should be determined, as it is demanding in terms of surgical skill and perioperative care, its short-term and medium-term results should be understood, and the aspects that could be improved should be analysed.
Early recurrence, both hepatic and extrahepatic, is an additional problem in the results of the technique. Rapid liver hypertrophy, which allows for a complete resection of the disease in a short time, prevents the selection of patients who could benefit from it. There have been some attempts to compare techniques based on their results, but it seems evident that these conclusions lack scientific rigour because they are based on methodologically incorrect analyses. The comparative analysis of oncological results should be carried out based on intention-to-treat, taking into account patients who do not complete both surgical stages after PPE due to the progression of the disease or any other reason (27.8%),23 which in our series amounted to 12.5% (case 2).
Prospective and randomised studies seem difficult to perform because of the great variability in the clinical presentation of patients and several factors involved in their management, which can affect their prognosis. Therefore, the analysis of active multicentre records should provide results arranged by groups of diseases and guide the standardisation of the technique.
Conflicts of InterestThe authors declare that there are no conflicts of interest.
Please cite this article as: Montalvá Orón EM, Maupoey Ibáñez J, Bañuelos Carrillo R, Boscà Robledo A, Orbis Castellanos JF, Moya Herraiz Á, et al. ALPPS monosegmento: una nueva variante de las técnicas de regeneración hepática rápida. Revisión crítica de los resultados iniciales de nuestra serie. Cir Esp. 2015;93:436–443.