Intraoperative peripheral nerve injury during colorectal surgery procedures is a potentially serious complication that is often underestimated.
The Trendelenburg position, use of inappropriately padded armboards and excessive shoulder abduction may encourage the development of brachial plexopathy during laparoscopic procedures.
In open colorectal surgery, nerve injuries are less common. It usually involves the femoral plexus associated with lithotomy position and self-retaining retractor systems.
Although in most cases the recovery is mostly complete, treatment consists of physical therapy to prevent muscular atrophy, protection of hypoesthetic skin areas, and analgesics for neuropathic pain. The aim of the present study is to review the incidence, prevention and management of intraoperative peripheral nerve injury.
La lesión de nervio periférico durante procedimientos de cirugía colorrectal constituye una complicación potencialmente grave a menudo infravalorada durante el postoperatorio.
La posición de Trendelenburg, la colocación de topes y las abducciones de los brazos han demostrado favorecer el desarrollo de plexopatía braquial durante los procedimientos laparoscópicos.
En cirugía colorrectal abierta las lesiones nerviosas son menos frecuentes, afectan preferentemente al plexo femoral y se asocian a la posición de litotomía y al uso de autorretractores.
Aunque en la mayoría de los casos la recuperación es completa, el tratamiento consiste en fisioterapia para prevenir la atrofia muscular, protección de las zonas con hipoestesia y analgésicos frente al dolor neuropático. El objetivo del presente artículo es realizar una revisión de la literatura existente sobre incidencia, prevención y manejo de la lesión intraoperatoria de nervio periférico.
Intraoperative peripheral nerve injury (IPNI) is an important complication that has not been mentioned very often in the context of perioperative morbidity. Although some papers cite an incidence of from 0.3%1 to 1.5%,2 its actual incidence is unknown due, on the one hand, to the fact that it may vary depending on its position and the surgical speciality, and on the other, that it is rarely mentioned in the literature, where it is limited to a heterogeneous series of cases. In spite of this, it is a potentially serious complication with the risk of chronic neurological damage1,3–5 and major medical-legal costs.2,5–12 In colorectal surgery the range of approaches and surgical positions mean that knowledge and publication of the mechanisms that cause neurological damage is especially important. The purpose of this paper is to revise the current evidence for IPNI in colorectal surgery using an open or laparoscopic approach. To this end we analyse the factors associated with IPNI as described in the literature, as well as its diagnosis, and evolution.
MethodThe literature was searched in MEDLINE, PubMed, and EMBASE using the following keywords: Peripheral Nerve Injury, Positioning Colorectal Surgery, Laparoscopic Colorectal Surgery, Brachial Plexus Injury, and Femoral Neuropathy. Papers in English and Spanish were selected which included adult patients operated on using open or laparoscopic colorectal surgery from 1993 to 2014. Of a total of 78 papers, 19 were selected for this revision. These include 3 systematic revisions, 12 case series, and 4 original papers. Case data and aspects in connection with their diagnosis, treatment, and prevention were extracted from all of these papers. 59 studies were excluded because they covered patients operated on for other specialities, had different subjects or dealt with direct injuries to the nerves during surgery.
ResultsAetiologySince Herbert Seddon systematised damage to the peripheral nerves in 1942,13,14 these injuries have been classified into 3 types according to their morphological characteristics. Neuropraxy is the blockage of the nerve connection without structural damage, and it usually recovers in 4–6 weeks. Axonotmesis consists of the breakage of the axon, while the perineuronal conjunctive tissue remains intact. Although in the majority of cases spontaneous regeneration occurs within a few weeks, surgical treatment may be required.15 Neurotmesis involves the complete sectioning of the axon and elements of conjunctive tissue, preventing it from regenerating spontaneously. These patients lose the nerve function (motor or sensitive) and require surgical repair for their recovery.16,17
The chief mechanisms which cause IPNI are the position of the patient and the duration of the surgery.17,18 The duration of surgery is a time during which muscle relaxation and the absence of pain facilitate injuries due to stretching and slackening.19 Although the peripheral nerves are elastic20,21 and can withstand stretching to 6%–10% of their length,22–25 greater traction causes dysfunction due to blockage of axon transmission, reduction of the intraneural blood flow, and histological damage.22,26–30 Nevertheless, IPNI may even occur in patients who have been subjected to the necessary postural measures during the intervention. Previous diabetic neuropathy, peripheral vascular disease11,17,31,32 and malnutrition with the absence of subcutaneous fat cushioning are preoperative factors that make patients more liable to IPNI due to compression or stretching.31,33 Other contributing factors are nicotinism, alcoholism, and vitamin B12 deficit,4,11,34 or intraoperative factors such as hypotension, hypothermia,11,31 and heparinization.1,35
Laparoscopic SurgeryLaparoscopic colorectal surgery has been associated with a higher risk of IPNI than open surgery (3.2% vs 0.2%).1 The main reason is that the use of laparoscopy in inframesocolic surgery often requires the Trendelenburg position during a long time to facilitate the descent of the small bowel loops into the supramesocolic compartment.
In left colon and rectal surgery the patient is placed with the hips semi-extended, the knees flexed at about 45° and with the calf on leg supports to prevent compression of the popliteal fossa.35 Due to the lateral inclination and the Trendelenburg position, head and side restraints are required at shoulder level to prevent the patient from descending down the operating table. Some published cases refer to the injurious effect due to compression of the restraint on the brachial plexus.36 The arms are held to the trunk by cloth bands on the right side and in abduction on the left side. During the perineal time of an abdominoperineal amputation, this position is modified by flexing the hips in lithotomy (the Lloyd-Davies position)37 to permit the perineal approach to the rectum.
In right colon surgery the patient is placed in a supine position with the legs separated and slightly flexed on leg supports, left lateral anti-Trendelenburg inclination. The left arm is held to the trunk while the right arm is placed on an arm support in abduction. Injuries to the nerve branches have been described with abductions greater than 90° or with the neck turned contralaterally.38 Lateral restraints must be put into place at shoulder level to prevent excessive compression; the leg supports function as the caudal stop.
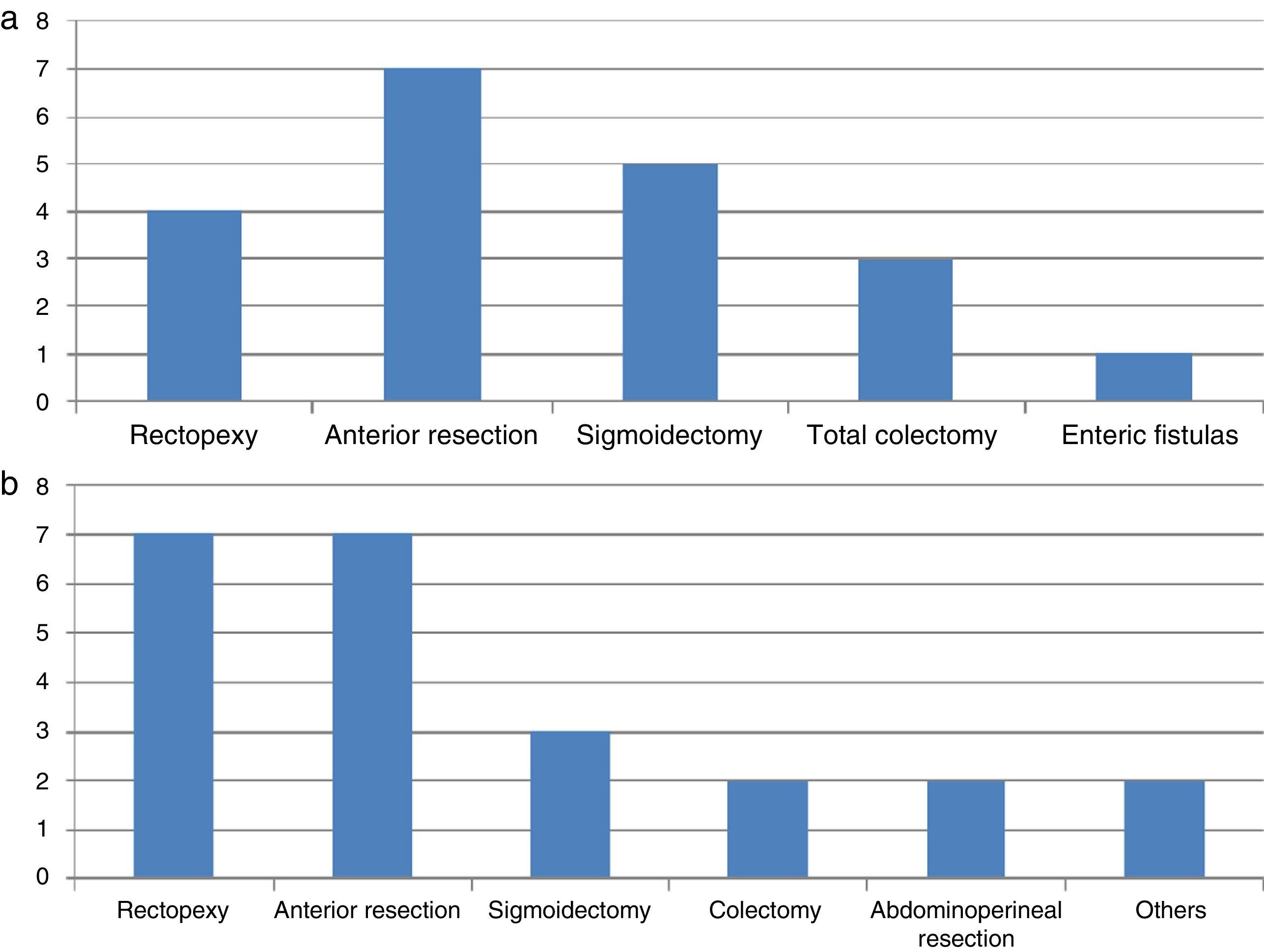
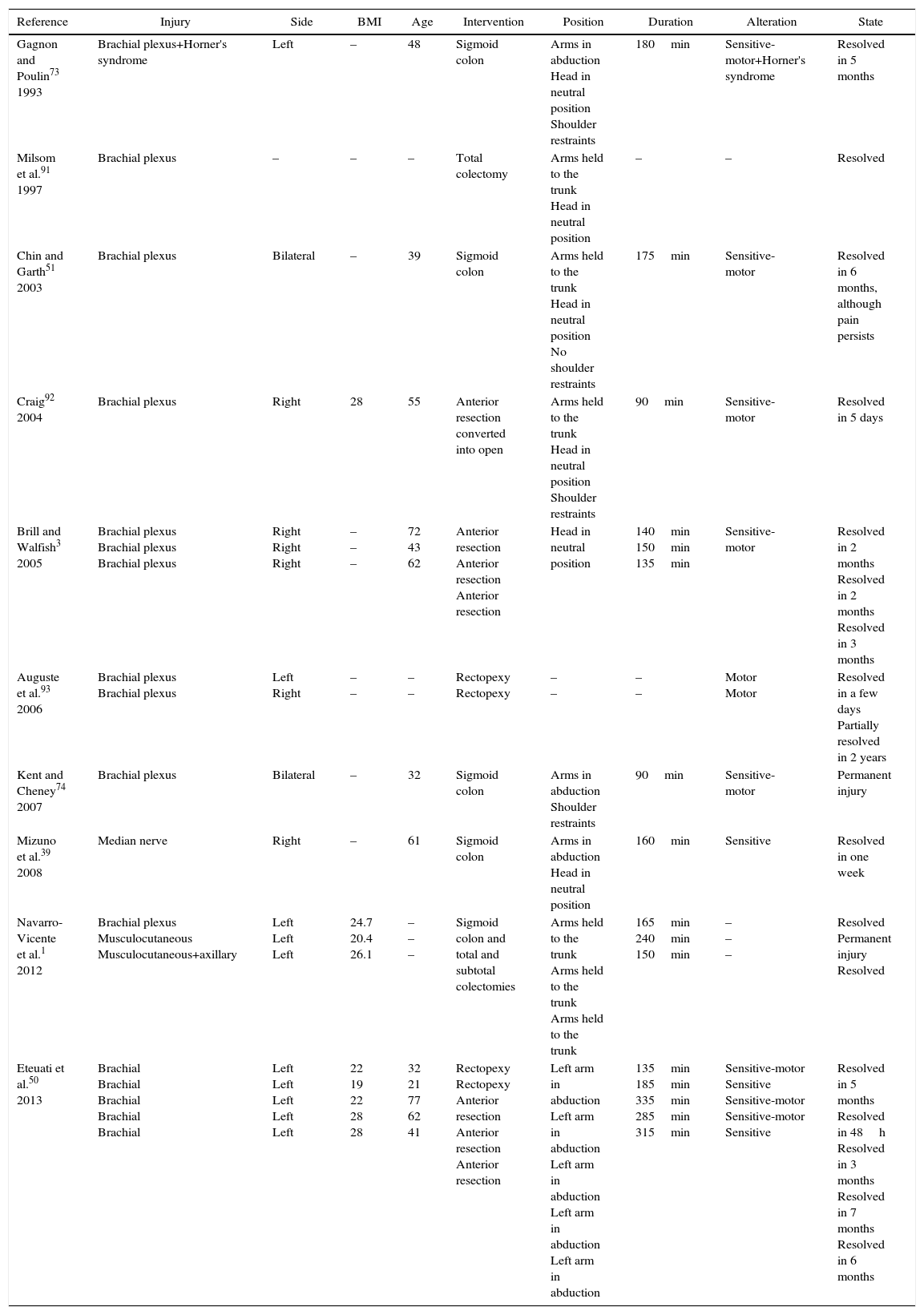
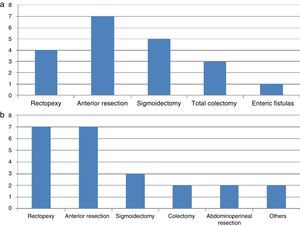
A revision of the bibliography from 1993 to date shows 20 described cases of neural injuries in colorectal laparoscopic surgery (Table 1). Of all these, 17 occurred in the brachial plexus4,7 while the others refer to specific damage in its terminal nerves: the median, musculocutaneous and axillary nerves.1,39 No IPNI in the lower limbs has been described during a laparoscopic approach. The average age of the patients is 50 years (range 21–77 years) with an average BMI of 24.2kg/m2 (range 19–28). The duration of the operation for development of plexopathy varied from 90min to 335min. The different colorectal operations in which this is described are shown in Fig. 1. In all cases the patient was kept in the Trendelenburg position; with the arms in abduction at 80° in 7 cases and held to the trunk in 3 cases. The injuries are usually to the left plexus, coinciding with the arm in abduction.
List of the Laparoscopic Colorectal Operations Associated With Injury to the Peripheral Nerves.
| Reference | Injury | Side | BMI | Age | Intervention | Position | Duration | Alteration | State |
|---|---|---|---|---|---|---|---|---|---|
| Gagnon and Poulin73 1993 | Brachial plexus+Horner's syndrome | Left | – | 48 | Sigmoid colon | Arms in abduction Head in neutral position Shoulder restraints | 180min | Sensitive-motor+Horner's syndrome | Resolved in 5 months |
| Milsom et al.91 1997 | Brachial plexus | – | – | – | Total colectomy | Arms held to the trunk Head in neutral position | – | – | Resolved |
| Chin and Garth51 2003 | Brachial plexus | Bilateral | – | 39 | Sigmoid colon | Arms held to the trunk Head in neutral position No shoulder restraints | 175min | Sensitive-motor | Resolved in 6 months, although pain persists |
| Craig92 2004 | Brachial plexus | Right | 28 | 55 | Anterior resection converted into open | Arms held to the trunk Head in neutral position Shoulder restraints | 90min | Sensitive-motor | Resolved in 5 days |
| Brill and Walfish3 2005 | Brachial plexus Brachial plexus Brachial plexus | Right Right Right | – – – | 72 43 62 | Anterior resection Anterior resection Anterior resection | Head in neutral position | 140min 150min 135min | Sensitive-motor | Resolved in 2 months Resolved in 2 months Resolved in 3 months |
| Auguste et al.93 2006 | Brachial plexus Brachial plexus | Left Right | – – | – – | Rectopexy Rectopexy | – – | – – | Motor Motor | Resolved in a few days Partially resolved in 2 years |
| Kent and Cheney74 2007 | Brachial plexus | Bilateral | – | 32 | Sigmoid colon | Arms in abduction Shoulder restraints | 90min | Sensitive-motor | Permanent injury |
| Mizuno et al.39 2008 | Median nerve | Right | – | 61 | Sigmoid colon | Arms in abduction Head in neutral position | 160min | Sensitive | Resolved in one week |
| Navarro-Vicente et al.1 2012 | Brachial plexus Musculocutaneous Musculocutaneous+axillary | Left Left Left | 24.7 20.4 26.1 | – – – | Sigmoid colon and total and subtotal colectomies | Arms held to the trunk Arms held to the trunk Arms held to the trunk | 165min 240min 150min | – – – | Resolved Permanent injury Resolved |
| Eteuati et al.50 2013 | Brachial Brachial Brachial Brachial Brachial | Left Left Left Left Left | 22 19 22 28 28 | 32 21 77 62 41 | Rectopexy Rectopexy Anterior resection Anterior resection Anterior resection | Left arm in abduction Left arm in abduction Left arm in abduction Left arm in abduction Left arm in abduction | 135min 185min 335min 285min 315min | Sensitive-motor Sensitive Sensitive-motor Sensitive-motor Sensitive | Resolved in 5 months Resolved in 48h Resolved in 3 months Resolved in 7 months Resolved in 6 months |
The incidence of IPNI after laparotomy varies according to different studies from 0.2%1 to 0.17%.35 Although neuropathy of the brachial plexus may occur, the associated injuries usually affect the lower limbs, and they derive from the use of self-retracting instruments and the lithotomy position.
Self-retaining separators are essential instruments for facilitating exposure in open colorectal surgery. They are composed of a stable frame on which interchangeable valves are affixed that retracts the wall and viscera. The positioning of the side valves of the self-retaining separator may injure the femoral nerve,40–43 causing ischemia44 by compressing the psoas muscle on the nerve.45,46 Goldman et al.41 state that the self-retaining separator increases the incidence of postoperative femoral neuropathy by more than 10 times.
The lithotomy position facilitates the exposure of the perineum, so that it is commonly used in proctology. It is useful in access to the rear face of the rectum during endorectal and combined approaches in abdominoperineal amputation.35 This position is a recognised cause of neuropathy of the lower limbs,47 due to stretching of the nerves with the abduction and forced external rotation of the hips or compression at the level of the inguinal ligament.48
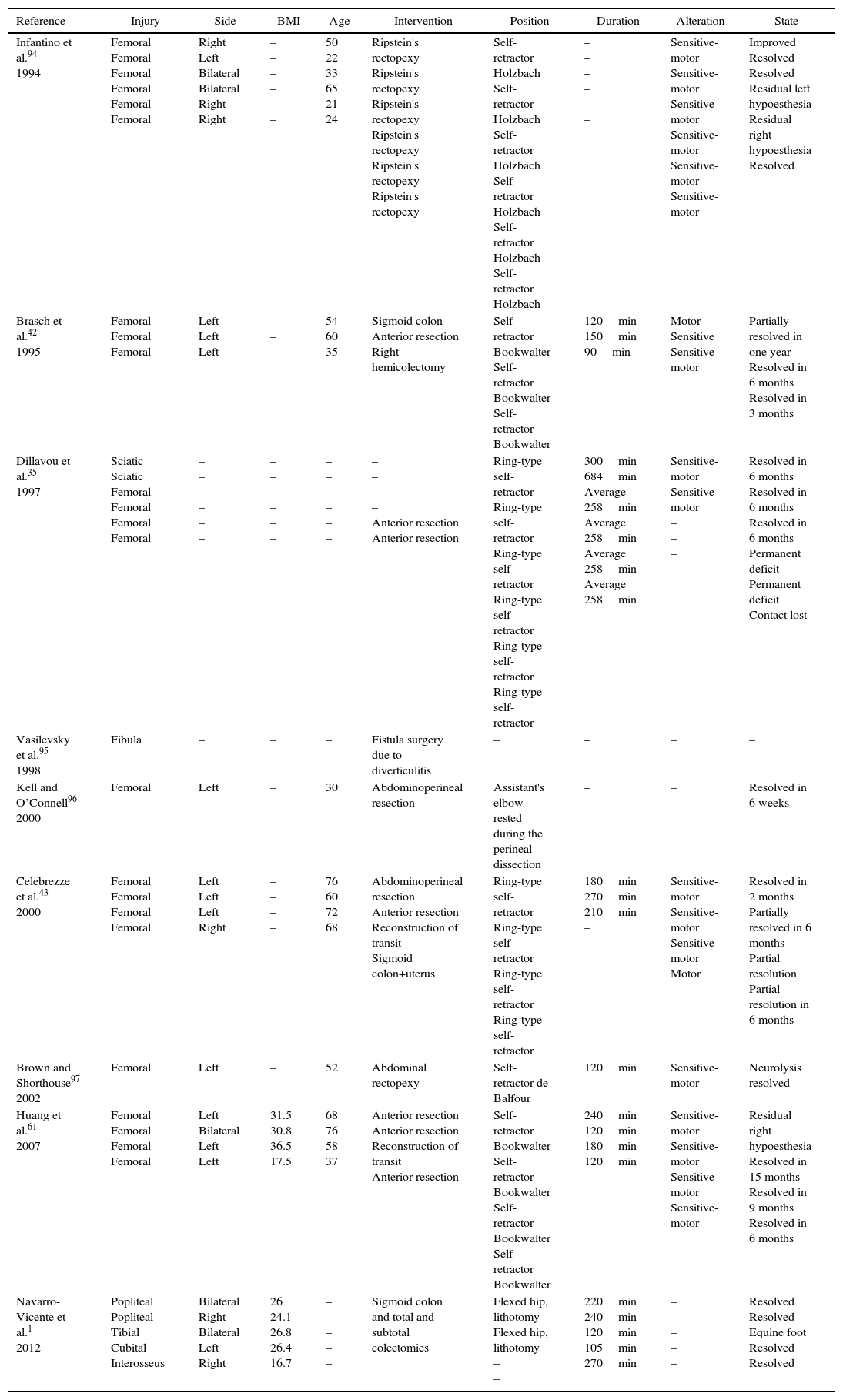
Although it is a controversial question, bodily constitution, the duration of surgery and transversal incisions45,49 have all been said to be possible factors associated with IPNI in open surgery. Table 2 shows a revision of the cases published since 1994. It can be seen that of the 31 cases described, 29 refer to lower limb neuropathy, of which 23 are of the femoral plexus and the others of the sciatic, popliteal, peroneal and tibial nerves. Average patient age is 50 years old (range 21–76 years old) with an average BMI of 26.2kg/m2 (range 16.7–36.5kg/m2). The duration of the operation for the development of plexopathy varies from 90min to 684min. The different colorectal interventions described are shown in Fig. 1.
List of the Open Colorectal Operations Associated With Injury to the Peripheral Nerves.
| Reference | Injury | Side | BMI | Age | Intervention | Position | Duration | Alteration | State |
|---|---|---|---|---|---|---|---|---|---|
| Infantino et al.94 1994 | Femoral Femoral Femoral Femoral Femoral Femoral | Right Left Bilateral Bilateral Right Right | – – – – – – | 50 22 33 65 21 24 | Ripstein's rectopexy Ripstein's rectopexy Ripstein's rectopexy Ripstein's rectopexy Ripstein's rectopexy Ripstein's rectopexy | Self-retractor Holzbach Self-retractor Holzbach Self-retractor Holzbach Self-retractor Holzbach Self-retractor Holzbach Self-retractor Holzbach | – – – – – – | Sensitive-motor Sensitive-motor Sensitive-motor Sensitive-motor Sensitive-motor Sensitive-motor | Improved Resolved Resolved Residual left hypoesthesia Residual right hypoesthesia Resolved |
| Brasch et al.42 1995 | Femoral Femoral Femoral | Left Left Left | – – – | 54 60 35 | Sigmoid colon Anterior resection Right hemicolectomy | Self-retractor Bookwalter Self-retractor Bookwalter Self-retractor Bookwalter | 120min 150min 90min | Motor Sensitive Sensitive-motor | Partially resolved in one year Resolved in 6 months Resolved in 3 months |
| Dillavou et al.35 1997 | Sciatic Sciatic Femoral Femoral Femoral Femoral | – – – – – – | – – – – – – | – – – – – – | – – – – Anterior resection Anterior resection | Ring-type self-retractor Ring-type self-retractor Ring-type self-retractor Ring-type self-retractor Ring-type self-retractor Ring-type self-retractor | 300min 684min Average 258min Average 258min Average 258min Average 258min | Sensitive-motor Sensitive-motor – – – – | Resolved in 6 months Resolved in 6 months Resolved in 6 months Permanent deficit Permanent deficit Contact lost |
| Vasilevsky et al.95 1998 | Fibula | – | – | – | Fistula surgery due to diverticulitis | – | – | – | – |
| Kell and O’Connell96 2000 | Femoral | Left | – | 30 | Abdominoperineal resection | Assistant's elbow rested during the perineal dissection | – | – | Resolved in 6 weeks |
| Celebrezze et al.43 2000 | Femoral Femoral Femoral Femoral | Left Left Left Right | – – – – | 76 60 72 68 | Abdominoperineal resection Anterior resection Reconstruction of transit Sigmoid colon+uterus | Ring-type self-retractor Ring-type self-retractor Ring-type self-retractor Ring-type self-retractor | 180min 270min 210min – | Sensitive-motor Sensitive-motor Sensitive-motor Motor | Resolved in 2 months Partially resolved in 6 months Partial resolution Partial resolution in 6 months |
| Brown and Shorthouse97 2002 | Femoral | Left | – | 52 | Abdominal rectopexy | Self-retractor de Balfour | 120min | Sensitive-motor | Neurolysis resolved |
| Huang et al.61 2007 | Femoral Femoral Femoral Femoral | Left Bilateral Left Left | 31.5 30.8 36.5 17.5 | 68 76 58 37 | Anterior resection Anterior resection Reconstruction of transit Anterior resection | Self-retractor Bookwalter Self-retractor Bookwalter Self-retractor Bookwalter Self-retractor Bookwalter | 240min 120min 180min 120min | Sensitive-motor Sensitive-motor Sensitive-motor Sensitive-motor | Residual right hypoesthesia Resolved in 15 months Resolved in 9 months Resolved in 6 months |
| Navarro-Vicente et al.1 2012 | Popliteal Popliteal Tibial Cubital Interosseus | Bilateral Right Bilateral Left Right | 26 24.1 26.8 26.4 16.7 | – – – – – | Sigmoid colon and total and subtotal colectomies | Flexed hip, lithotomy Flexed hip, lithotomy – – | 220min 240min 120min 105min 270min | – – – – – | Resolved Resolved Equine foot Resolved Resolved |
Neuropathy of the brachial plexus C5-D1 has a published incidence that varies from 0.9%50 to 6.7% according to Brill and Walfisch,3 and it is the second most common perioperative nerve injury.8,9
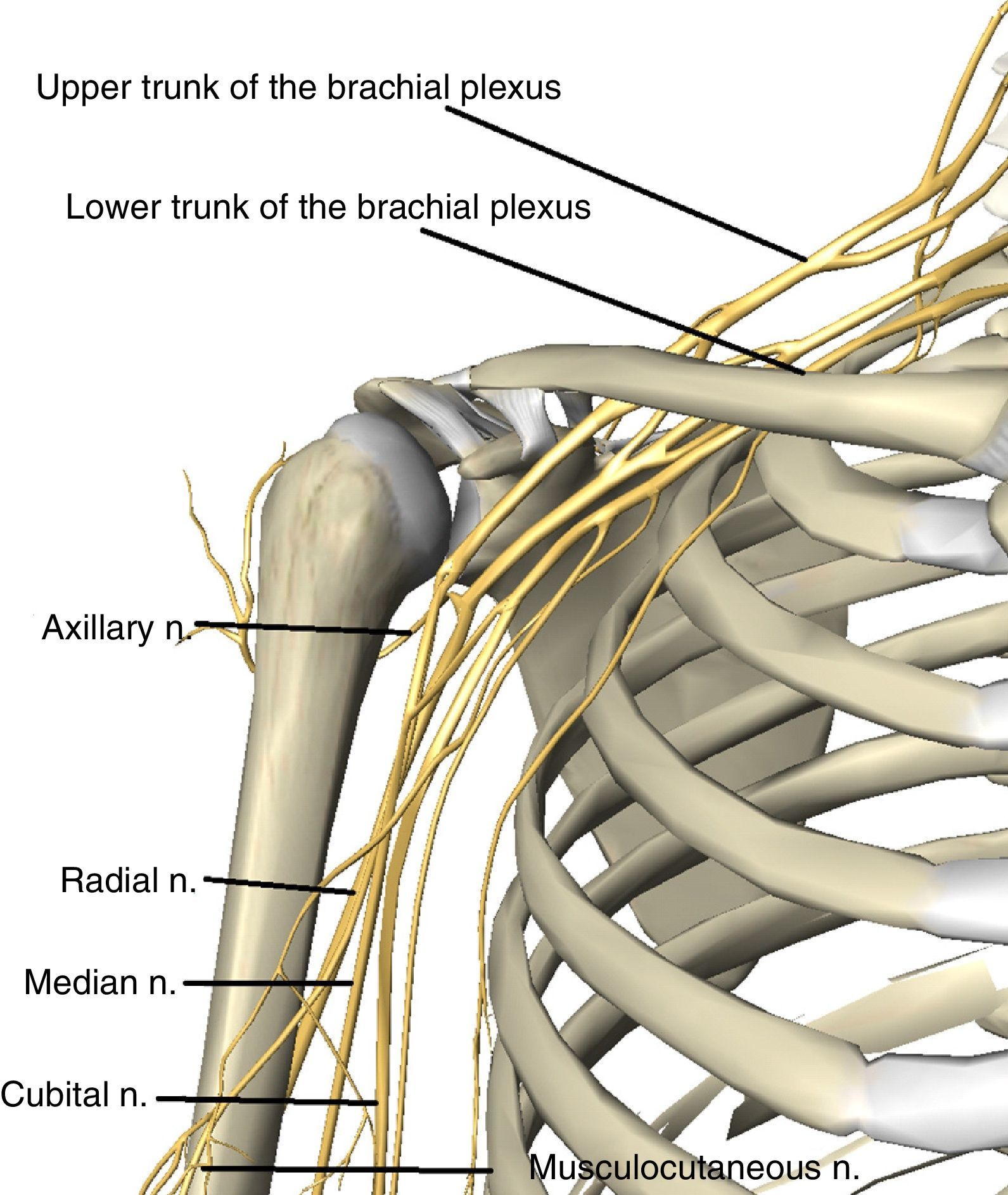
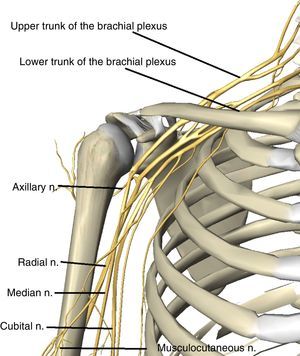
The long trajectory of the plexus from the neck to the arm, as well as its position proximal to the cervical vertebra and distal to the axillary fascia, make it especially vulnerable to traction and stretching injuries.16–18,51–53 From its origin the plexus passes through the supraclavicular fossa between the middle and anterior scalene muscles, then it passes under the clavicle to reach the subclavicular fossa where the 3 trunks into which the plexus is divided accompany the subclavicular vascular bundle (Fig. 2).
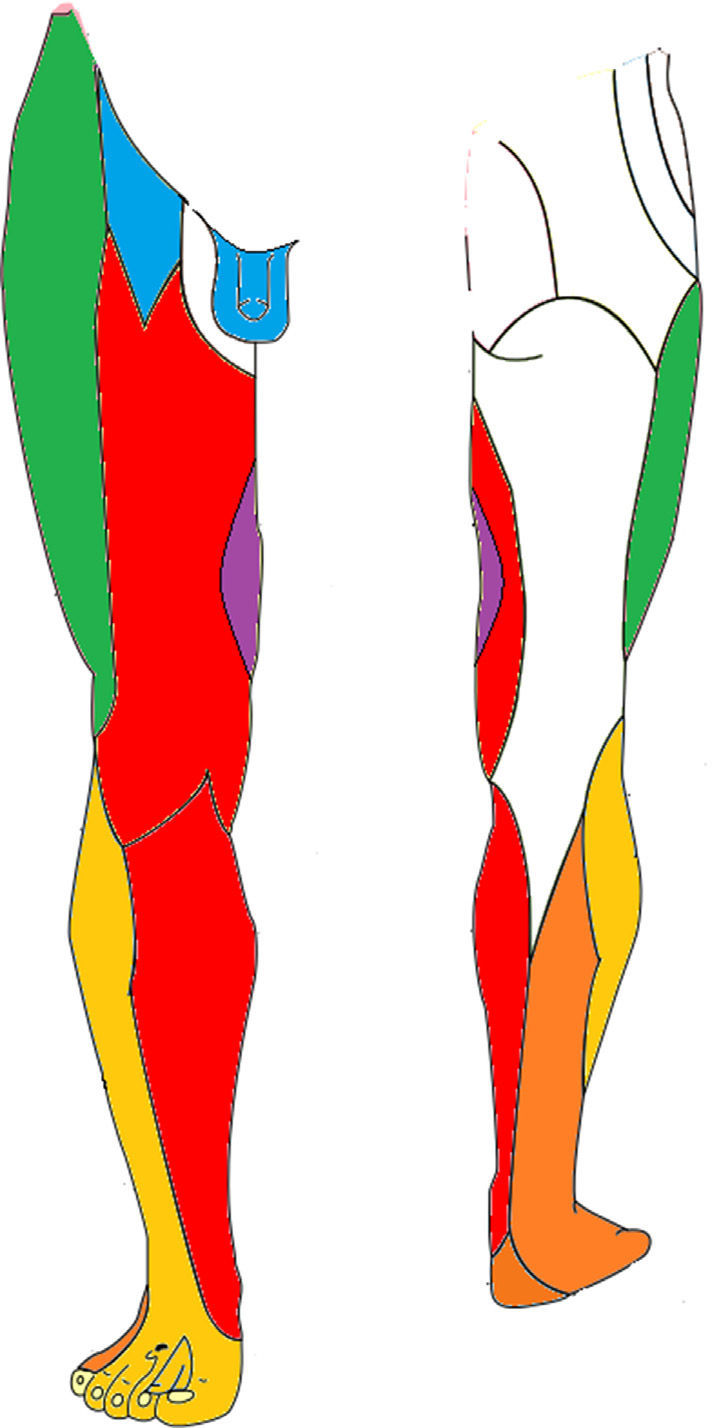
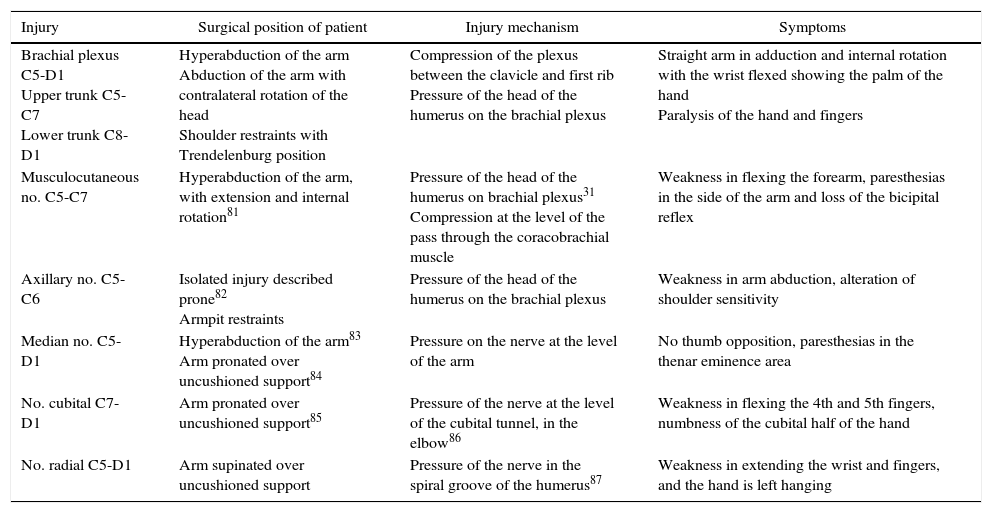
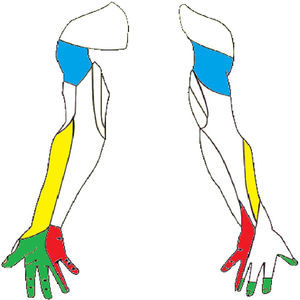
Brachial plexopathy is characterised by alteration of the sensitivity which with varying degrees of intensity may affect the different trunk territories of the upper limb (Fig. 3). Although the complete loss of sensitivity is possible, patients often describe from numbness and paresthesias to intense pain (Table 3). The osteotendinous reflexes of the C5-D1 (bicipital [C5-C6], tricipital [C7], and styloradial [C6]) roots may be lost. The motor sign will take the form of varying degrees of muscular weakness.
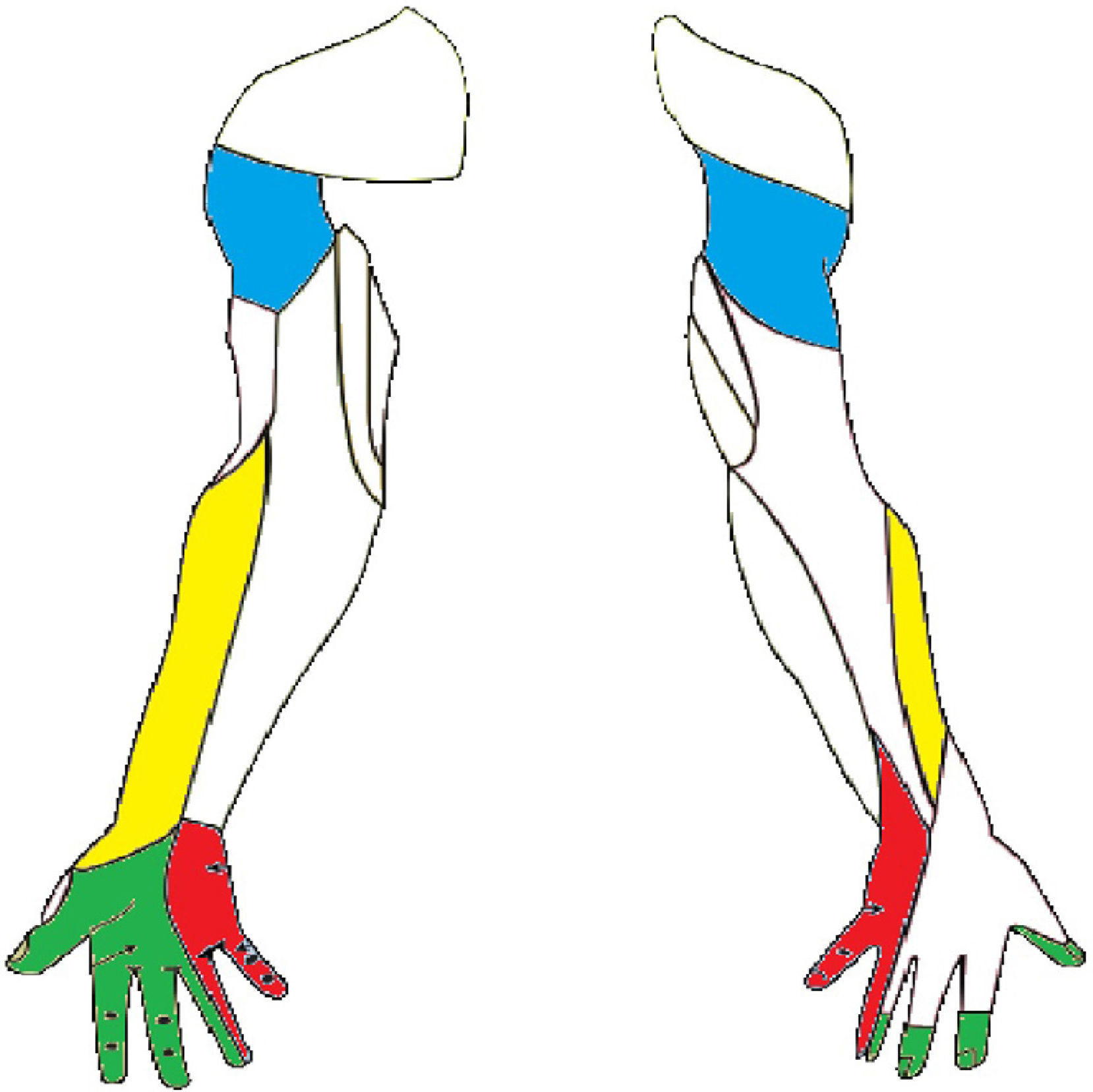
Sensitive innervation of the upper limbs. The nerve trunks have been marked to show the injuries mentioned in the literature. The musculocutaneous nerve (the side of the forearm), the axillary nerve (shoulder area), the median nerve (side of palm) and the cubital nerve (palm and medial dorsal).
Injury Mechanisms and IPNI Symptoms in the Upper Limbs.
| Injury | Surgical position of patient | Injury mechanism | Symptoms |
|---|---|---|---|
| Brachial plexus C5-D1 Upper trunk C5-C7 Lower trunk C8-D1 | Hyperabduction of the arm Abduction of the arm with contralateral rotation of the head Shoulder restraints with Trendelenburg position | Compression of the plexus between the clavicle and first rib Pressure of the head of the humerus on the brachial plexus | Straight arm in adduction and internal rotation with the wrist flexed showing the palm of the hand Paralysis of the hand and fingers |
| Musculocutaneous no. C5-C7 | Hyperabduction of the arm, with extension and internal rotation81 | Pressure of the head of the humerus on brachial plexus31 Compression at the level of the pass through the coracobrachial muscle | Weakness in flexing the forearm, paresthesias in the side of the arm and loss of the bicipital reflex |
| Axillary no. C5-C6 | Isolated injury described prone82 Armpit restraints | Pressure of the head of the humerus on the brachial plexus | Weakness in arm abduction, alteration of shoulder sensitivity |
| Median no. C5-D1 | Hyperabduction of the arm83 Arm pronated over uncushioned support84 | Pressure on the nerve at the level of the arm | No thumb opposition, paresthesias in the thenar eminence area |
| No. cubital C7-D1 | Arm pronated over uncushioned support85 | Pressure of the nerve at the level of the cubital tunnel, in the elbow86 | Weakness in flexing the 4th and 5th fingers, numbness of the cubital half of the hand |
| No. radial C5-D1 | Arm supinated over uncushioned support | Pressure of the nerve in the spiral groove of the humerus87 | Weakness in extending the wrist and fingers, and the hand is left hanging |
Upper trunk (Erb's) palsy: this is caused by involvement of roots C5-C6 and sometimes C7. The patient presents with their arm straight in adduction and in internal rotation, with their wrist flexed showing the palm of the hand. The resulting posture is the characteristic “beggars” position.54 The sensory deficit presents along the lateral face of the whole upper limb.55
Injury of the lower trunk (Klumpke's palsy): this is less common than upper trunk injury, and it occurs due to damage to roots C8-D1, causing paralysis of the hand and fingers that is sometimes accompanied by palm hypoesthesia.56 If root D1 is affected close to the cervical sympathetic trunk, this may be associated with Horner's syndrome with ptosis, myosis, and facial anhydrosis.55,57
Given the compressive origin of the injuries to the brachial plexus, several terminals are often injured simultaneously, although clinical signs may predominate for one of them. Differential diagnosis with pure IPNI of the brachial plexus is important as the prognosis for the latter is generally better.58
Navarro-Vicente et al.1 describe 2 cases of IPNI caused by damage to the musculocutaneous and axillary nerves. Involvement of the musculocutaneous nerve usually present as weakness in flexing the elbow, paresthesias in the radial side of the arm and loss of the bicipital reflex. When damage is caused to the axillary nerve atrophy and numbness are usually detected in the region of the deltoids, sometimes accompanied by weakness of the arm in abduction.
Mizuno et al.39 refer to a median nerve injury during laparoscopic sigmoidectomy. The injury to this branch of the brachial plexus started with paresthesias in the region of the thenar eminence (Fig. 3) and weakness in making the pincer movement between the first and second fingers of the hand.
Although no references have been described in colorectal surgery, injury to the cubital nerve is, nevertheless, one of the most frequent in a laparoscopic approach.59 It usually presents clinically as numbness of the cubital half of the hand, accompanied by weakness in flexion between the 4th and 5th fingers. On the other hand, radial neuropathy manifests as a “hanging hand”, with weakness in extending the wrist and fingers (Table 3).
Brachial plexopathy generally gives rise to a neuropraxy with symptoms that remit in 2–7 months,3,31,32,51 with gradual recovery, first sensory and then motor.3 Ben-David and Stahl have suggested that diabetic patients require a longer recovery time.32
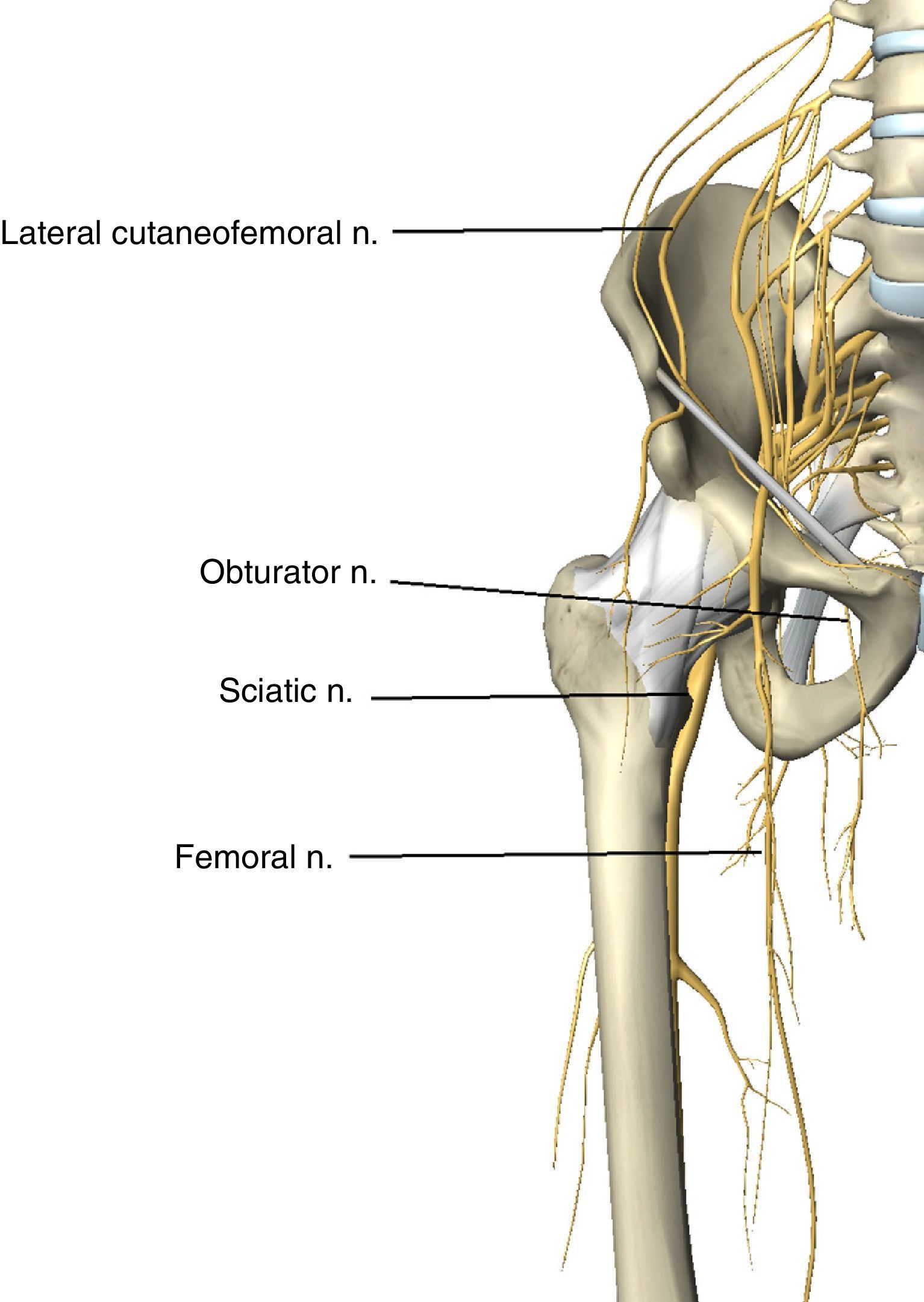
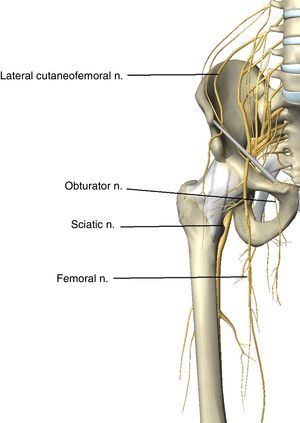
Lower Limb NeuropathyThe crural or femoral nerve (L2–L4) originates in the lumbar plexus. It emerges from the lower third of the larger psoas muscle before descending in the pelvis between the iliac and psoas muscles. It enters the thigh behind the inguinal ligament, covered by the psoas fascia. At this level the nerve, as it is superficial with precarious vascularisation,60 is vulnerable to compression (Fig. 4).
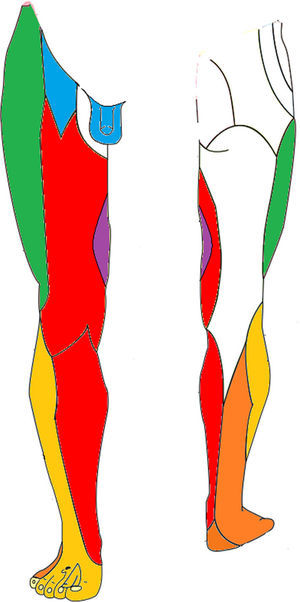
It innervates the quadriceps muscle and receives sensitivity from the anteromedial face of the thigh and leg. Injury to the femoral nerve has a described incidence of 0.12%.35 When it is injured, the first sign is usually that the patient falls when commencing to walk61 (Table 4). If the injury was caused by compression at a proximal level in the lumbar plexus or pelvis (flexion of the hip or self-retainers), the symptoms will be weakness in extending the knee, flexing the thigh, hypoesthesia, and weak patella reflex.62 If the injury is caused where the nerve emerges from the pelvis under the inguinal ligament,43 this will only be differentiated from a pelvic injury by conservation of hip flexion.63 Injuries in the thigh may present as a motor deficit or isolated sensory deficit.43
Injury Mechanisms and Symptoms of IPNI in the Lower Limbs.
| Injury | Surgical position of the patient | Injury mechanism | Symptoms |
|---|---|---|---|
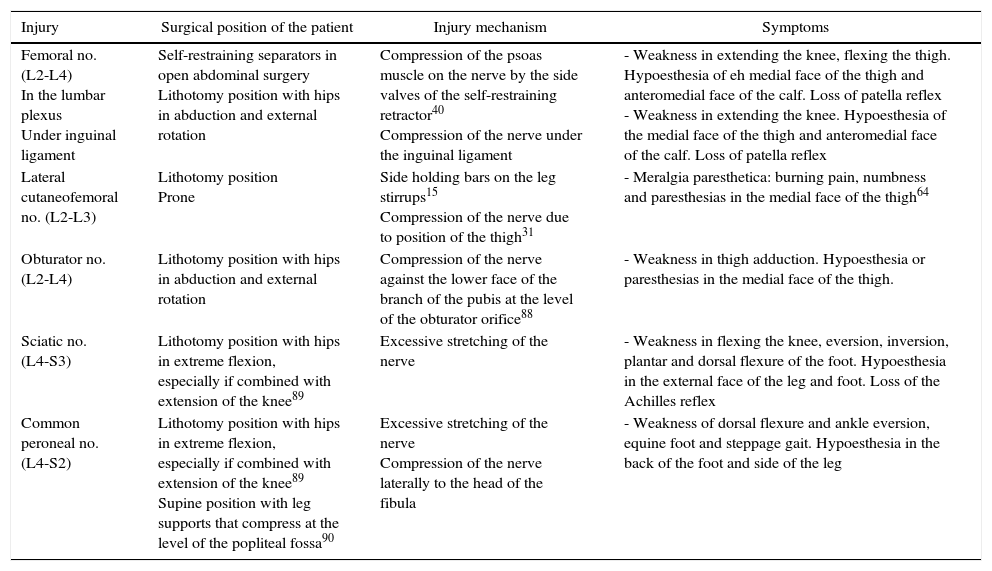
| Femoral no. (L2-L4) In the lumbar plexus Under inguinal ligament | Self-restraining separators in open abdominal surgery Lithotomy position with hips in abduction and external rotation | Compression of the psoas muscle on the nerve by the side valves of the self-restraining retractor40 Compression of the nerve under the inguinal ligament | - Weakness in extending the knee, flexing the thigh. Hypoesthesia of eh medial face of the thigh and anteromedial face of the calf. Loss of patella reflex - Weakness in extending the knee. Hypoesthesia of the medial face of the thigh and anteromedial face of the calf. Loss of patella reflex |
| Lateral cutaneofemoral no. (L2-L3) | Lithotomy position Prone | Side holding bars on the leg stirrups15 Compression of the nerve due to position of the thigh31 | - Meralgia paresthetica: burning pain, numbness and paresthesias in the medial face of the thigh64 |
| Obturator no. (L2-L4) | Lithotomy position with hips in abduction and external rotation | Compression of the nerve against the lower face of the branch of the pubis at the level of the obturator orifice88 | - Weakness in thigh adduction. Hypoesthesia or paresthesias in the medial face of the thigh. |
| Sciatic no. (L4-S3) | Lithotomy position with hips in extreme flexion, especially if combined with extension of the knee89 | Excessive stretching of the nerve | - Weakness in flexing the knee, eversion, inversion, plantar and dorsal flexure of the foot. Hypoesthesia in the external face of the leg and foot. Loss of the Achilles reflex |
| Common peroneal no. (L4-S2) | Lithotomy position with hips in extreme flexion, especially if combined with extension of the knee89 Supine position with leg supports that compress at the level of the popliteal fossa90 | Excessive stretching of the nerve Compression of the nerve laterally to the head of the fibula | - Weakness of dorsal flexure and ankle eversion, equine foot and steppage gait. Hypoesthesia in the back of the foot and side of the leg |
The femoral nerve is especially vulnerable to injury in its path through the pelvis64; in this it is vascularised from the iliolumbar artery and the deep circumflex iliac artery. However, there is a notable difference in vascularisation between the left and right nerves. Thus the right femoral nerve receives more vascularisation from the deep circumflex iliac artery, as well as branches of the fourth and fifth lumbar arteries, making the left nerve more vulnerable to injury.64
The lateral cutaneofemoral or femorocutaneous nerve (L2–L3) runs along the side of the psoas muscle before entering the thigh. This nerve is entirely sensitive, so that if it is compressed in the abdomen or inguinal region this causes burning pain, numbness, and paresthesias in the region of the thigh which it innervates (Fig. 5). These symptoms, which are termed meralgia paresthetica, worsen when walking, and usually disappear spontaneously.65
Sensitive innervation of the lower limbs. The nerve trunks with injuries mentioned in the literature have been marked. The genitofemoral nerve (genital and upper medial area of the thigh), the obturator nerve (medial thigh), the femoral nerve (anteromedial thigh and calf) and the sciatic nerve (side of calf and foot).
The obturator nerve (L2–L4) collects the sensitivity of the inner part of the thigh and innervates the leg adductors. Obturator nerve neuropathy is characterised by weakness and atrophy of the thigh adductor muscles accompanied by hypoesthesia and paresthesias in the medial face of the thigh (Fig. 5). It may be injured bilaterally when the nerve is stretched due to abduction with prolonged external rotation of the hip.66 To reduce the risk of this it is recommended to simultaneously flex the hip when positioning the patient.20 Other possible mechanisms which cause injuries are pelvic tumour surgery67,68 or the presence of an obturator hernia (Table 4).
The sciatic nerve (L4–S3) can be compressed by a pelvic haemorrhage as a complication of laparotomy68 or direct crushing of the nerve in the gluteal region during anaesthesia or when lying down for a long time on a hard surface,19,69 or due to severe flexion of the hip which stretches the nerve when the patient is positioned in lithotomy (Fig. 5).
As a result of sciatic neuropathy hypoesthesia occurs on the external face of the leg and foot except for the malleolus. This also causes paresthesia and atrophy of the knee flexor (posterior thigh) muscles and all of the muscles below the knee (eversion and inversion, plantar, and dorsal flexion of the foot) together with reduction of the Achilles reflex.
The common peroneal or external sciatic popliteal nerve (L4-S2). This originates laterally at the head of the fibula, as a branch of the sciatic nerve. It may be injured at this level as a result of compression by leg supports.19 This injury causes hypoesthesia in the back of the foot and the distal side of the leg. It also causes paresis in dorsal flexion and eversion of the ankle, giving rise to an equine foot and steppage gait.
DiagnosisThe postoperative diagnosis of IPNI requires a high degree of suspicion from the surgeon. Shoulder pain caused by brachial plexopathy may often be mistakenly attributed to residual pneumoperitoneum or musculotendinous injuries; the first sign of injury to the femoral nerve may be that the patient falls when starting to walk.70 Due to all of these reasons special attention must be paid during the postoperative period to symptoms of numbness and weakness in the limbs, especially if they are asymmetrical.
The neurological examination must include a detailed history and an evaluation of the motor and sensory functions of the nerve roots.17,71 To confirm the injury, locate its level and evaluate its intensity. This examination may be complemented by an electroneurogram and an electromyogram.72 Nerve conduction characteristics (latency, amplitude, and velocity) are used to diagnose IPNI and to determine its prognosis and severity. It is also useful to differentiate IPNI due to stretching of the nerve from acute plexitis (the Parsonage-Turner syndrome).31 The typical changes of de-innervation do not appear until 2–3 weeks after the injury, so that determination before this time will detect the pre-existing physiological state of the nerve.54
Complementary imaging studies may be necessary to exclude organic disease such as perineural haematomas (cranial CT or spinal magnetic resonance).
PreventionTo prevent IPNI during laparoscopic surgical procedures, the duration of surgery should be reduced and the position of the patient should be optimised. Arm abduction greater than 80° should be avoided, keeping the arm on the trunk whenever possible. The elbows should be slightly flexed to discharge the tension in the brachial plexus and median nerve, and supination of the forearm must also be avoided.18,73 The head should be kept in a neutral position so that the neck is not turned or hyperextended,50 and restraints should not be used at the shoulders, as they may compress the head of the humerus and the acromioclavicular joint against the brachial plexus.17,33,51–53,74
New restraining devices have been coming into use recently. “Allen” type leg supports fulfil a dual purpose. On the one hand they contribute to the caudal support of the foot so that the weight of the legs rests on the heel and sole of the foot, not on the popliteal fossa. This prevents compression of the peroneal nerve and compression of the venous return. On the other hand, at any time during surgery these leg supports allow the surgeon to freely modify the state of hip flexoabduction.35 Vacuum “bean bags” are restraining devices that adapt to patient anatomy, making it possible to hold the shoulders while keeping the arm held to the body and supporting the trunk and abdomen in lateral movements.
Some authors described the measures which are necessary to prevent lumbar and sacral nerve root injuries during open abdominal operations. The self-retaining retractor valves must be as short as possible to ensure correct retraction of the abdominal wall45 without also pulling on the psoas muscle.46 Pads can be placed under the valves to cushion the pressure they exert,75 checking the femoral pulse after putting them into place43,44 and regularly checking the positions of the valves during the operation. When using the lithotomy position the hip should not be flexed, abducted or rotated externally, to prevent compressing the nerves under the inguinal ligament.61 When the surgeon and assistant are both working on the same side of the patient, they must avoid resting on the abducted upper limb as this could increase the abduction.
TreatmentTreatment of IPNI must start as soon as possible to ensure optimum recovery.43,76,77 These patients have to be treated with physiotherapy, including passive muscular exercises, stretching and galvanic stimulation to prevent muscular atrophy. Hypoesthetic zones must be protected to prevent injuries which the patient does not notice. Neuropathic pain requires specific drugs such as gabapentin, pregabalin, amitriptyline and topiramate.74,76
During the postoperative period regular neurological checks must take place; if there is no recovery 3 or 4 months after the injury, a surgical exploration will be performed9,42,44,49,62,78 to evaluate the possibility of repair, graft or neurolysis.9 Intraoperative evaluation of nerve action potentials in the injured limbs may help to decide whether neurolysis or resection with a graft should be used.9,78
The prognosis for IPNI depends on the injured nerve, the intensity of pressure and its duration.43 In the majority of cases recovery is complete, especially in muscular deficit,41–43,79,80 although symptoms of pain or residual paresthesia may persist, especially in diabetic subjects.32 Goldman et al.41 reported a total recovery rate of 94% in a large series of 282 patients, as opposed to 6% of patients in whom symptoms persisted 116 days after surgery.
ConclusionsWhile surgery is being carried out, the absence of pain and muscular relaxation facilitate the creation of nerve injuries due to stretching and distension.
A revision of the literature on IPNI showed that in colorectal surgery neuropathy occurs the most often when a laparoscopic approach is used, fundamentally of the brachial plexus. In open colorectal surgery the cases described refer to sciatic and femoral neuropathy which is generally attributed to the use of self-retracting separators and the lithotomy position.
A high level of diagnostic suspicion is required for the early detection of this postoperative complication so that the appropriate rehabilitation treatment can be started.
FinancingNo support for this study was received in the form of grants.
Conflict of InterestsThe authors have no conflict of interests to declare.
Please cite this article as: Colsa Gutiérrez P, Viadero Cervera R, Morales-García D, Ingelmo Setién A. Lesión intraoperatoria de nervio periférico en cirugía colorrectal. Revisión de conjunto. Cir Esp. 2016;94:125–136.