Incisional hernias are a very common problem, with an estimated incidence around 15%–20% of all laparotomies. Evisceration is another important problem, with a lower rate (2.5%–3%) but severe consequences for patients. Prevention of both complications is an essential objective of correct patient treatment due to the improved quality of life and cost savings.
This narrative review intends to provide an update on incisional hernia and evisceration prevention. We analyze the current criteria for proper abdominal wall closure and the possibility to add prosthetic reinforcement in certain cases requiring it. Parastomal, trocar-site hernias and hernias developed after stoma closure are included in this review.
La hernia incisional es una patología muy común cuya incidencia se estima en torno al 15-20% de todas las laparotomías. La evisceración es otro problema importante, con una incidencia menor (2,5-3%) pero con graves consecuencias para el paciente. Por todo ello, la prevención de ambas complicaciones surge como un objetivo fundamental para el tratamiento correcto de los pacientes, por la mejora de la calidad de vida y por el ahorro de costes que supondría.
Esta revisión narrativa pretende realizar una puesta al día en la prevención de la hernia incisional y la evisceración. Se analizan los criterios actuales para el cierre correcto de la pared abdominal, seguido de la posibilidad de añadir refuerzos protésicos en aquellos pacientes o casos que así lo requieran. Eventraciones especiales, como las originadas tras la inserción de trócares de laparoscopia o las secundarias a la realización de un estoma, se incluyen también en este trabajo.
Incisional hernias (IH) are one of the most common pathologies treated by the General Surgery Department. The incidence of IH is around 15%–20%, although it can reach 50% in certain contexts.1 This means a large number of patients to be treated, with the corresponding high consumption of resources. In the United States, for example, two million laparotomies are performed each year,2 and more than 100000 incisional hernias are treated surgically. In addition, a significant percentage can present serious complications, such as incarceration, strangulation or bowel obstruction that will require urgent surgery. Evisceration is another serious problem that can arise after laparotomy, with an approximate incidence of 2.5%–3%3 but a mortality rate of 45% and very high morbidity, which is close to 75%.
All the incisions that are used to access the abdominal cavity, whether midline laparotomies or incisions to introduce trocars in laparoscopic approaches, can potentially develop IH. In addition, there are well-known risk factors4 that increase the chances of the patient having an IH, such as obesity, urgent surgery, smoking, abdominal aortic aneurysms or the appearance of postoperative wound infection. For all these reasons, the prevention of IH has emerged as a fundamental objective for correct patient treatment due to the improved quality of life and cost savings that it would entail.
The objective of this study was to provide a narrative review about the prevention of IH and evisceration. First, we describe the aspects related with correct closure of the abdominal wall, followed by the possibility of using prosthetic reinforcement in those patients or cases requiring it. Special cases of incisional hernias, such as those caused after the insertion of laparoscopy trocars or those secondary to the completion of a stoma, are also contemplated in this paper.
Correct Abdominal Wall ClosureThe IH rate in a midline laparotomy is estimated at around 15%–20% (50% in some risk groups) and the evisceration rate is around 3%, which makes it an important problem.1,3 In the last 20 years, clinical, experimental and meta-analysis studies have been published to determine the best materials and methods for abdominal wall closure.1,5
The recommendations made based on these studies established that correct closure should be done with continuous suture, in a single plane and with slow-absorption suture material.1,5 In addition, other clinical and experimental studies6,7 have demonstrated that for greater effectiveness of a continuous suture, this should be done with a suture length/wound length (SL/WL) ratio of 4:1 or higher, as described by Jenkins6 and known in the literature as “the 4:1 rule.”
An SL/WL ratio of less than 4:1 has been shown to be one of the main technical factors associated with the appearance of IH, and some authors even recommend redoing the suture if the suture/incision ratio is not met adequately.7,8 This “rule” is simple to apply: you only need a tape measure and very simple calculations.8
The next evolution in abdominal wall closure was the change in the size of the suture and the distance between stitches. In previous studies, the closure was done with heavy-gauge sutures (0/1), with a separation of 1cm between stitches and a distance to the aponeurotic edge of another 1cm (called the “large stitch” or “large bite” technique), thereby achieving an SL/WL ratio greater than 4:1.7 Israelsson's group described the completion of closure with smaller stitches that were 5mm apart and a distance to the edge of 5–8mm, using a smaller suture gauge (2/0) and small needle.9 This change ensures reaching the 4:1 or higher ratio, and also demonstrates a lower incidence of IH and a lower rate of wound infection than with the large-stitch technique.9 This technique has been called “small stitch” or “small bite” method.
The results of this first study9 have been confirmed in the STITCH10 trial, which compared closure with a polydioxanone 1/0 loop mounted on a 48-mm needle using the large-stitch technique versus polydioxanone 2/0 caliber mounted on a 31-mm needle using the small-stitch technique, finding a significantly lower IH rate with the latter.
The European Hernia Society (EHS) has developed Clinical Guidelines for the closure of abdominal wall incisions11 based on the GRADE methodology.12 The recommendations that appear in these guidelines are: (1) use non-midline incisions (moderate evidence and strong recommendation); (2) closure of midline laparotomy with continuous suture (low evidence and strong recommendation); (3) avoid rapidly-absorbed material (moderate evidence, strong recommendation); (4) use slow-absorption monofilament material (low evidence, weak recommendation); (5) suture in a single layer, without closing the peritoneum (low evidence and weak recommendation); and (6) use the small-stitch technique (moderate evidence, weak recommendation) with an SL/WL ratio of at least 4:1 (low evidence and weak recommendation).11
Despite all the above, there are still questions on how to proceed with the closure of abdominal wall incisions, such as closing the laparotomy in emergency situations, in contaminated environments, how to close non-midline laparotomies, or closure in difficult situations or in patients at risk.
In short, during abdominal wall closure after midline laparotomy, the amount of thread used should be calculated according to the length of the incision measured, ensuring a ratio of at least 4:1.8,11 It should be mandatory to check these lengths and record this information on the operating room report. The small-stitch technique with small continuous monofilament sutures in a single aponeurotic plane may be recommendable in the closure of elective median laparotomy.11
Use of Prophylactic Reinforcement Mesh in the Prevention of Incisional HerniasDespite correct closure of the abdominal wall following the recommendations outlined above, a systematic review has published an IH rate of 12.8% after a mean follow-up of almost 2 years.13 In patients with risk factors and longer follow-ups, this incidence is even greater.14 These risk factors usually include: obesity, surgical wound infection, aortic aneurysm and oncological surgery. A prospective study conducted in Canada in patients operated on for abdominal aortic aneurysm by means of median laparotomy showed an IH incidence of 69% after a 5-year follow-up.15 In studies performed in patients operated on for colorectal cancer, an incidence of incisional hernias greater than 40% was detected when follow-up radiological scans (CT scans) were analyzed.16,17
To avoid high IH rates in spite of the appropriate closure of the incision, and especially in high-risk patients, prosthetic reinforcement of the abdominal wall closure has been described.11 A very important factor for resistance to the widespread adoption of this preventive measure is the fear of the appearance of infections or other complications related with the use of prostheses.18
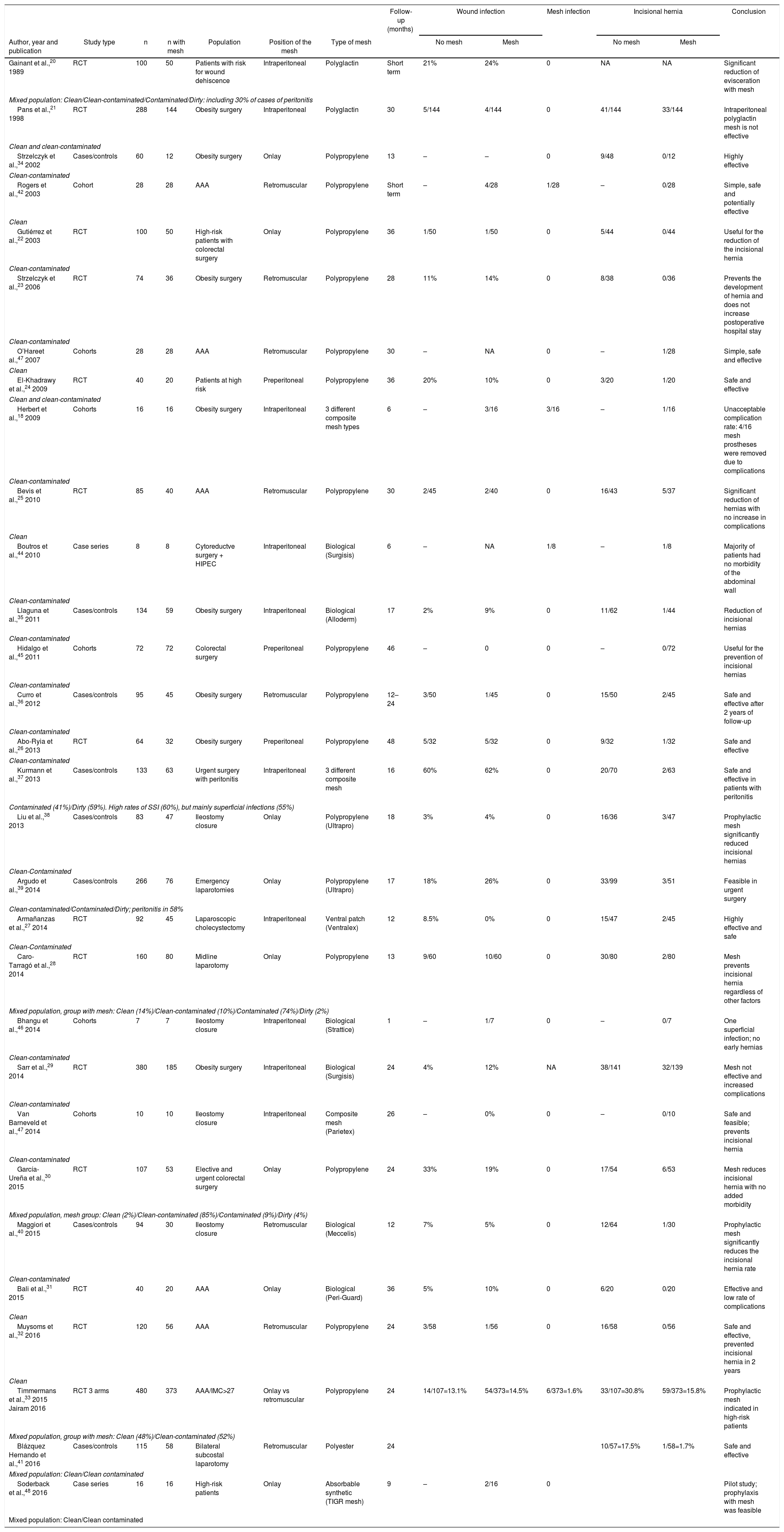
Methods. We reviewed the evidence published about the use of prophylactic mesh in various types of laparotomies (excluding prophylactic mesh in parastomal hernias) and the incidence of complications for the different degrees of contamination of the surgical wound according to the classification by the Centers for Disease Control and Prevention (CDC).19 For this, a bibliographic search was conducted in the Medline (PubMed), Scopus, Web of Science (WOS) and Embase databases of articles published before January 2017, using the key words “prevention of incisional hernia”, “prophylaxis of incisional hernia”, “prophylactic mesh”, “reinforcement mesh”, “laparotomy closure”, “wound infection” and “infected mesh”. A total of 30 studies were identified, including 14 randomized clinical trials,20–33 8 case-control studies34–41 and 8 cohort studies or case series.18,42–48
Table 1 presents a summary of all available data specifically related to the incidence of wound infections and prosthesis-related complications. In addition, the overall IH rate results were extracted. In the studies analyzed, several prophylactic meshes were used (permanent synthetic, absorbable synthetic or biological), and the position in which they are placed also varied (onlay or retromuscular).
Summary of the Evidence From the Review of the Literature for Studies About Prophylactic Reinforcement Mesh.
| Follow-up (months) | Wound infection | Mesh infection | Incisional hernia | Conclusion | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Author, year and publication | Study type | n | n with mesh | Population | Position of the mesh | Type of mesh | No mesh | Mesh | No mesh | Mesh | |||
| Gainant et al.,20 1989 | RCT | 100 | 50 | Patients with risk for wound dehiscence | Intraperitoneal | Polyglactin | Short term | 21% | 24% | 0 | NA | NA | Significant reduction of evisceration with mesh |
| Mixed population: Clean/Clean-contaminated/Contaminated/Dirty: including 30% of cases of peritonitis | |||||||||||||
| Pans et al.,21 1998 | RCT | 288 | 144 | Obesity surgery | Intraperitoneal | Polyglactin | 30 | 5/144 | 4/144 | 0 | 41/144 | 33/144 | Intraperitoneal polyglactin mesh is not effective |
| Clean and clean-contaminated | |||||||||||||
| Strzelczyk et al.,34 2002 | Cases/controls | 60 | 12 | Obesity surgery | Onlay | Polypropylene | 13 | – | – | 0 | 9/48 | 0/12 | Highly effective |
| Clean-contaminated | |||||||||||||
| Rogers et al.,42 2003 | Cohort | 28 | 28 | AAA | Retromuscular | Polypropylene | Short term | – | 4/28 | 1/28 | – | 0/28 | Simple, safe and potentially effective |
| Clean | |||||||||||||
| Gutiérrez et al.,22 2003 | RCT | 100 | 50 | High-risk patients with colorectal surgery | Onlay | Polypropylene | 36 | 1/50 | 1/50 | 0 | 5/44 | 0/44 | Useful for the reduction of the incisional hernia |
| Clean-contaminated | |||||||||||||
| Strzelczyk et al.,23 2006 | RCT | 74 | 36 | Obesity surgery | Retromuscular | Polypropylene | 28 | 11% | 14% | 0 | 8/38 | 0/36 | Prevents the development of hernia and does not increase postoperative hospital stay |
| Clean-contaminated | |||||||||||||
| O’Hareet al.,47 2007 | Cohorts | 28 | 28 | AAA | Retromuscular | Polypropylene | 30 | – | NA | 0 | – | 1/28 | Simple, safe and effective |
| Clean | |||||||||||||
| El-Khadrawy et al.,24 2009 | RCT | 40 | 20 | Patients at high risk | Preperitoneal | Polypropylene | 36 | 20% | 10% | 0 | 3/20 | 1/20 | Safe and effective |
| Clean and clean-contaminated | |||||||||||||
| Herbert et al.,18 2009 | Cohorts | 16 | 16 | Obesity surgery | Intraperitoneal | 3 different composite mesh types | 6 | – | 3/16 | 3/16 | – | 1/16 | Unacceptable complication rate: 4/16 mesh prostheses were removed due to complications |
| Clean-contaminated | |||||||||||||
| Bevis et al.,25 2010 | RCT | 85 | 40 | AAA | Retromuscular | Polypropylene | 30 | 2/45 | 2/40 | 0 | 16/43 | 5/37 | Significant reduction of hernias with no increase in complications |
| Clean | |||||||||||||
| Boutros et al.,44 2010 | Case series | 8 | 8 | Cytoreductve surgery + HIPEC | Intraperitoneal | Biological (Surgisis) | 6 | – | NA | 1/8 | – | 1/8 | Majority of patients had no morbidity of the abdominal wall |
| Clean-contaminated | |||||||||||||
| Llaguna et al.,35 2011 | Cases/controls | 134 | 59 | Obesity surgery | Intraperitoneal | Biological (Alloderm) | 17 | 2% | 9% | 0 | 11/62 | 1/44 | Reduction of incisional hernias |
| Clean-contaminated | |||||||||||||
| Hidalgo et al.,45 2011 | Cohorts | 72 | 72 | Colorectal surgery | Preperitoneal | Polypropylene | 46 | – | 0 | 0 | – | 0/72 | Useful for the prevention of incisional hernias |
| Clean-contaminated | |||||||||||||
| Curro et al.,36 2012 | Cases/controls | 95 | 45 | Obesity surgery | Retromuscular | Polypropylene | 12–24 | 3/50 | 1/45 | 0 | 15/50 | 2/45 | Safe and effective after 2 years of follow-up |
| Clean-contaminated | |||||||||||||
| Abo-Ryia et al.,26 2013 | RCT | 64 | 32 | Obesity surgery | Preperitoneal | Polypropylene | 48 | 5/32 | 5/32 | 0 | 9/32 | 1/32 | Safe and effective |
| Clean-contaminated | |||||||||||||
| Kurmann et al.,37 2013 | Cases/controls | 133 | 63 | Urgent surgery with peritonitis | Intraperitoneal | 3 different composite mesh | 16 | 60% | 62% | 0 | 20/70 | 2/63 | Safe and effective in patients with peritonitis |
| Contaminated (41%)/Dirty (59%). High rates of SSI (60%), but mainly superficial infections (55%) | |||||||||||||
| Liu et al.,38 2013 | Cases/controls | 83 | 47 | Ileostomy closure | Onlay | Polypropylene (Ultrapro) | 18 | 3% | 4% | 0 | 16/36 | 3/47 | Prophylactic mesh significantly reduced incisional hernias |
| Clean-Contaminated | |||||||||||||
| Argudo et al.,39 2014 | Cases/controls | 266 | 76 | Emergency laparotomies | Onlay | Polypropylene (Ultrapro) | 17 | 18% | 26% | 0 | 33/99 | 3/51 | Feasible in urgent surgery |
| Clean-contaminated/Contaminated/Dirty; peritonitis in 58% | |||||||||||||
| Armañanzas et al.,27 2014 | RCT | 92 | 45 | Laparoscopic cholecystectomy | Intraperitoneal | Ventral patch (Ventralex) | 12 | 8.5% | 0% | 0 | 15/47 | 2/45 | Highly effective and safe |
| Clean-Contaminated | |||||||||||||
| Caro-Tarragó et al.,28 2014 | RCT | 160 | 80 | Midline laparotomy | Onlay | Polypropylene | 13 | 9/60 | 10/60 | 0 | 30/80 | 2/80 | Mesh prevents incisional hernia regardless of other factors |
| Mixed population, group with mesh: Clean (14%)/Clean-contaminated (10%)/Contaminated (74%)/Dirty (2%) | |||||||||||||
| Bhangu et al.,46 2014 | Cohorts | 7 | 7 | Ileostomy closure | Intraperitoneal | Biological (Strattice) | 1 | – | 1/7 | 0 | – | 0/7 | One superficial infection; no early hernias |
| Clean-contaminated | |||||||||||||
| Sarr et al.,29 2014 | RCT | 380 | 185 | Obesity surgery | Intraperitoneal | Biological (Surgisis) | 24 | 4% | 12% | NA | 38/141 | 32/139 | Mesh not effective and increased complications |
| Clean-contaminated | |||||||||||||
| Van Barneveld et al.,47 2014 | Cohorts | 10 | 10 | Ileostomy closure | Intraperitoneal | Composite mesh (Parietex) | 26 | – | 0% | 0 | – | 0/10 | Safe and feasible; prevents incisional hernia |
| Clean-contaminated | |||||||||||||
| García-Ureña et al.,30 2015 | RCT | 107 | 53 | Elective and urgent colorectal surgery | Onlay | Polypropylene | 24 | 33% | 19% | 0 | 17/54 | 6/53 | Mesh reduces incisional hernia with no added morbidity |
| Mixed population, mesh group: Clean (2%)/Clean-contaminated (85%)/Contaminated (9%)/Dirty (4%) | |||||||||||||
| Maggiori et al.,40 2015 | Cases/controls | 94 | 30 | Ileostomy closure | Retromuscular | Biological (Meccelis) | 12 | 7% | 5% | 0 | 12/64 | 1/30 | Prophylactic mesh significantly reduces the incisional hernia rate |
| Clean-contaminated | |||||||||||||
| Bali et al.,31 2015 | RCT | 40 | 20 | AAA | Onlay | Biological (Peri-Guard) | 36 | 5% | 10% | 0 | 6/20 | 0/20 | Effective and low rate of complications |
| Clean | |||||||||||||
| Muysoms et al.,32 2016 | RCT | 120 | 56 | AAA | Retromuscular | Polypropylene | 24 | 3/58 | 1/56 | 0 | 16/58 | 0/56 | Safe and effective, prevented incisional hernia in 2 years |
| Clean | |||||||||||||
| Timmermans et al.,33 2015 Jairam 2016 | RCT 3 arms | 480 | 373 | AAA/IMC>27 | Onlay vs retromuscular | Polypropylene | 24 | 14/107=13.1% | 54/373=14.5% | 6/373=1.6% | 33/107=30.8% | 59/373=15.8% | Prophylactic mesh indicated in high-risk patients |
| Mixed population, group with mesh: Clean (48%)/Clean-contaminated (52%) | |||||||||||||
| Blázquez Hernando et al.,41 2016 | Cases/controls | 115 | 58 | Bilateral subcostal laparotomy | Retromuscular | Polyester | 24 | 10/57=17.5% | 1/58=1.7% | Safe and effective | |||
| Mixed population: Clean/Clean contaminated | |||||||||||||
| Soderback et al.,48 2016 | Case series | 16 | 16 | High-risk patients | Onlay | Absorbable synthetic (TIGR mesh) | 9 | – | 2/16 | 0 | Pilot study; prophylaxis with mesh was feasible | ||
| Mixed population: Clean/Clean contaminated | |||||||||||||
AAA: abdominal aortic aneurysm; HIPEC: hyperthermic intraperitoneal chemotherapy; NA: not available; RCT: randomized controlled trial.
In the global analysis, the use of prophylactic reinforcement mesh to prevent IH was reported in 1759 patients, with an overall wound infection rate of 12% and a mesh infection rate of 0.6%. The evidence analyzed indicates that the procedure can be safe and effective both in clean and in clean-contaminated surgeries. There is not enough evidence available about safety in contaminated or dirty surgeries.
The fundamental limitation of this analysis is the quality of the included studies, which varies greatly as the studies range from large multicenter randomized studies to small case series, as well as the diversity of mesh used and their placement positions. Despite the limitations of this review, it appears that prophylactic mesh patches could potentially be used to prevent IH in clean or clean-contaminated surgery. Most studies have used permanent synthetic mesh, while some studies have used absorbable synthetic or biological mesh.20,29,31,35,40,44,46,48 We should also consider that a permanent synthetic mesh is cheaper than absorbable synthetic or biological ones.49 As for the position of the prophylactic mesh, both the onlay and retromuscular positions seem equally safe and effective. The position of the mesh will probably depend on the preference of each surgeon.
To identify in which group of patients at risk a prophylactic mesh should be recommended, further studies are necessary. Fischer et al.50 described a preoperative risk model that can be used to target prophylaxis, based on a large database of patients, from which a risk score was calculated to stratify patients from low risk to extreme risk. These risk models could allow for the identification of patients who could benefit more from the use of prophylactic reinforcement mesh.
Prevention of Parastomal Hernia With ProsthesesThe incidence of parastomal hernias (PH) in patients with intestinal stomata may vary depending on the type of stoma in question. PH associated with a terminal colostomy can fluctuate between 4% and 93%, depending on whether the diagnosis is clinical51 or radiological.52 The PH figures associated with terminal ileostomy have been reported between 2% and 28%,53 and the percentages associated with ureteroileostomy (Bricker) can reach 29%.54 Some authors argue that PH are the “natural” consequence of creating a stoma.53 PH cause significant problems in patient quality of life,55 and in some cases urgent surgery is needed due to complications. In addition, PH repair is associated with a high recurrence rate.56
After analyzing the existing literature on the use of prophylactic mesh in the construction of stomata to prevent PH57 and with regards to terminal colostomy, most of the published studies are observational and the techniques used for the prevention of PH had a series of common characteristics: (a) the most common technique was an open surgical approach with retromuscular mesh positioning58–62; (b) the majority of studies made an orifice in the center of the mesh (keyhole) through which the intestine is exteriorized59–61,63; and (c) the majority used non-absorbable flat synthetic mesh.58–63 In all of these observational studies, the results were positive in favor of using a mesh to prevent PH.
Furthermore, in published randomized clinical trials (RCT), the open surgical approach with a retromuscular mesh positioning technique is most extensively used.64,65 Almost all studies used a non-absorbable flat synthetic mesh,64,65 and most made a hole in the center (keyhole).63–65 Also in the RCT, the results were favorable toward using mesh with preventive indication.
Several systematic reviews or meta-analyses have also been published, some quite recently, which uniformly conclude that a non-absorbable synthetic mesh in the retromuscular position reduces the incidence of PH, with no increase in morbidity related to the presence of the prosthesis.66–68 The last published meta-analysis69 includes a new methodology (trial sequential analysis) that controls error type 1 and calculates the size of the information required to reach valid conclusions. The results show that prophylactic mesh reduces the incidence of PH without increasing complications, and since the sample size required to demonstrate the effect of the procedure has been reached, more RCT may not be needed in this context.
Regarding terminal ileostomy, until now there are no primary data in the literature from studies specifically designed to evaluate the effectiveness of mesh in the prevention of PH. There are only indirect data from studies including fundamentally patients with colostomies and a few with terminal ileostomy.70 Therefore, no practical conclusions can be drawn about the value of a preventive strategy with mesh in these cases.
As for ureteroileostomy (Bricker), there is very little information available in the literature, and only two observational studies have been found with a small number of patients where it seems that the placement of a mesh for the prevention of PH may be possible and safe.71,72 Therefore, it is not possible to make practical recommendations for prevention in this type of definitive stoma.
In short, there is evidence that the efficacy of non-absorbable synthetic mesh in the retromuscular position using an open approach is useful for the prevention of PH during the construction of a terminal colostomy. However, more data are needed in the literature to definitively endorse the usefulness of preventive mesh with a laparoscopic approach, the use of mesh other than non-absorbable synthetic types, and to define the best patient profile or whether it is useful in terminal ileostomies or in ureteroileostomies (Bricker).
Prevention of Hernias in the Trocar OrificeCurrently, in the national registry of incisional hernias (EVEREG), 16% of registered procedures are trocar site hernias (TSH).73 The published incidence of TSH is between 0.14% and 22%, depending on the series.74,75 The range of this incidence may be due to the fact that many TSH can remain asymptomatic, develop long-term or simply not be reported.
With high TSH figures, prevention is important. Therefore, a series of factors must be considered:
- (a)
Is it necessary to close all trocar sites? According to published reviews,75 the recommendation is that all trocar orifices that are 10mm or greater should be closed, and in children even those measuring 5mm should be closed. Another recommendation would be to reduce the diameter of the trocars that are used as much as possible, avoiding the use of trocars larger than 5mm, which is not always possible, using 3-mm trocars and mini-instruments.
- (b)
Umbilical access: One author76 reported that up to 18% of patients who undergo laparoscopic surgery have an associated umbilical hernia. The recommendation is to introduce the trocar through the hernia defect and repair it with mesh at the end of the operation.
- (c)
Bariatric surgery: In these patients, two risk factors are involved: obesity itself and the use of trocars larger than 10mm, so their closure should be mandatory. One study77 shows that the incidence of HT is very low with long-term follow-up, so if the trocars are placed obliquely, it would not be necessary to close them later. In any event, a prudent recommendation would be to close all the trocar orifices in bariatric surgery, and always close those orifices that are extended for the extraction of the surgical piece from the sleeve gastrectomy.
- (d)
How should we close? Many devices have been used to facilitate the closing of trocar orifices. All studies highlight the need to include the aponeurosis in the closure,78 but the importance of including or not including the peritoneum has not been established. To date, the most appropriate suture type (i.e. long-lasting absorbable suture or non-absorbable suture) has not been established.
- (e)
Use of prophylactic mesh. Short series79 have published the initial experience in laparoscopic cholecystectomy, with a prophylactic mesh of titanized polypropylene introduced through the trocar and affixed with cyanoacrylate. After a minimum follow-up of 6 months, TSH were not observed. Another author80 described a series of patients in whom a bioabsorbable device was used in the closure of the umbilical trocar, with no complications and no appearance of TSH at the one-year follow-up. Finally, an RCT with almost 50 patients in each arm compared the closure of the umbilical trocar with non-absorbable suture with the placement of a mesh composed of polypropylene and omega-3 acid, in an intraperitoneal position; the TSH rate was 31.9% in the control group and 4.4% with the use of mesh after one year of follow-up.27 Previous studies seemed to indicate that the use of prophylactic mesh can decrease the incidence of TSH, although more research is needed in this field.
Single-port surgery seems to have a greater incidence of TSH,81 but no specific studies have been published about how these incisions should be closed or whether preventive prostheses should be used in these cases.
Prevention of Incisional Hernia After the Closure of a Temporary StomaThere are several circumstances that require the closure of a temporary stoma: reconstruction of the intestinal tract after a protective stoma (ileostomy or colostomy) performed in patients with low colorectal anastomosis, restoration of intestinal continuity after a terminal colostomy performed in an emergency situation (for example, after acute perforated diverticulitis) or reconstruction after terminal ileostomy in patients with inflammatory bowel disease. Stoma closure surgery entails high morbidity, mainly derived from anastomotic problems, especially dehiscence, bowel obstruction, wound infection or evisceration. Another often underestimated complication is the development of a IH in the stoma closure scar. In a recent meta-analysis82 including 34 studies with 2729 stoma closures, the overall incidence of hernia was 7.4%, although the range was wide among the studies (0%–48%). The heterogeneity of the studies, their duration and type of follow-up (clinical or radiological) make it difficult to interpret and apply the results. Thus, if only the highest quality studies focused on IH incidence are selected, whether detected clinically or with radiological methods, this incidence reaches 30%. This review demonstrates that the clinical incidence of IH after the closure of a stoma is greater than expected and may be so high as to affect one in three patients. Another recent systematic review83 including 16 studies and 1613 patients has confirmed these conclusions. If diagnostic imaging methods are also used, the incidence is even higher,52 reaching almost 50%.
Correct Closure of the Abdominal Wall After a Temporary StomaJust as there is scientific evidence on how to close a midline laparotomy, there is no consensus84 on which is the best method of closing the wall after the reversal of a temporary stoma: in two fascial planes or in a single plane, continuous suture or interrupted stitched, non-absorbable or long-lasting absorbable suture material. There is no “gold standard” in this regard, and each surgeon performs a “personal” closure based on his/her surgical experience. There is only consensus regarding cutaneous closure, and tobacco pouch closure is considered to provide a lower incidence of wound infection.85 With this lack of evidence, no recommendations can be established about which closure method is best in this type of incision. Common sense makes us think that if the closure with continuous suture with long-lasting absorbable or non-absorbable material following the 4:1 rule is the current gold standard in the closure of midline laparotomy, then it could also be the standard in the closure of this type of incisions.
Prophylactic Mesh in the Closure of a Temporary StomaIn 2013, Liu et al.38 published a retrospective review of 83 consecutive non-randomized patients with temporary ileostomy closure using primary fascial closure with interrupted stitches of long-lasting absorbable material in all cases, and in 47 of them polypropylene mesh for reinforcement in the onlay position. After a median follow-up of 18 months, with physical examination and abdominal CT scan, the incidence of IH in the mesh group was 6.4% and 36.1% in the non-mesh group, with no differences in the incidence of wound infection and no need for mesh explantation in any case.
Van Barneveld et al.47 published a study about the safety of the technique in 10 patients with protective ileostomy in rectal cancer surgery, in whom synthetic mesh was implanted (polyester plus collagen, which can be used in contact with the viscera) in the intra-abdominal position at the moment of the creation of the stoma, which was exteriorized through it. In the reversion of the stoma, the defect of the fascia and the mesh was closed with continuous polypropylene suture or PDS.1 After a median of 26 months of follow-up, IH was not detected either on physical examination or by ultrasound.
One of the most important studies (currently recruiting) is the Reinforcement of Closure of Stoma Site (ROCSS)86 (ClinicalTrials.gov NCT02238964). It is a randomized, multicenter, prospective, phase iii study that includes 790 patients in two groups. In the experimental group, a biological mesh (porcine collagen without cross-linking) is used in the intraperitoneal position affixed with transfascial sutures, after which the fascia is closed over the mesh with long-term absorbable suture material.46 In the control group, the closure of the incision is done in the usual manner of each surgeon, with the requirement of not using short-lasting absorbable suture in the closure of the fascia. The follow-up has an expected duration of 2 years, with clinical evaluation and CT scan after one year and only clinical evaluation after 2 years. The results are expected by the end of 2017.
In 2015, Maggiori et al.40 published a case-control study aimed at evaluating the feasibility and potential benefits of placing a porcine collagen prophylactic mesh in the closure of a temporary ileostomy after rectal cancer surgery. The case group included 30 consecutive patients scheduled for temporary ileostomy closure, and 64 control subjects were selected from the hospital's prospective database, matched according to age, sex, associated diseases, radiotherapy and delay between the initial surgery and stoma closure. In all patients, closure was carried out in two planes: posterior and anterior fascia of the muscle with continuous polyglactin suture. In the group with mesh, a porcine collagen mesh measuring 10×10cm was placed in the retromuscular position without cross-linking, affixed with polypropylene suture. The skin was closed in all cases with a tobacco pouch suture. In the short-term follow-up, there were no differences in the incidence of wound infection (7% vs 5%). At the one-year follow-up study with control CT, the incidence of IH in the mesh group was 3% and 16% in the control group.
This study has been used as a basis for the design of a national randomized study in France (ClinicalTrials.gov NCT02576184)87 in which the objective was to evaluate the long-term incidence of IH after the closure of a temporary ileostomy after rectal cancer surgery. Patients were randomized into three groups: without mesh, with synthetic mesh and with biological mesh. The mesh prostheses were placed in retromuscular position, as in the case-control study indicated above.40 The study began in December 2015 and the results are expected for December 2018.
Another study underway is the ILEOCLOSE (ClinicalTrials.gov (NCT02226887), whose objective is to evaluate the efficacy of the placement of an absorbable mesh (Gore-Bio A) using the sandwich method during the ileostomy closure to prevent the development of an IH.88 The study is still in recruitment phase and results are therefore not available.
In conclusion, and based on the few cases published, a preventive prosthesis in this context can reduce the incidence of IH. The recommendations for mesh type and placement position will have to wait until results are published after the completion of the active clinical trials.
ConclusionThe most important step in the prevention of IH in laparotomies is the correct closure of the abdominal wall, following the 4:1 rule and using a 2/0 suture of long-lasting absorbable material with the small stitch or small bite technique. Prophylactic mesh used in patients at risk with laparotomies can drastically reduce the incidence of IH, although the type of mesh to be used and its placement position still have not been specifically defined. In parastomal hernias, the placement of a prophylactic mesh during the creation of the stoma can reduce the incidence of PH and the majority of studies coincide in the use of permanent synthetic mesh placed in the retromuscular position. To prevent trocar site hernias, it is necessary to close all the trocar orifices measuring 10mm or more, and it seems that the use of prophylactic mesh in patients at risk can reduce the incidence of TSH. As for the prevention of hernias that develop after the reversal of temporary stomata, it seems that the use of prophylactic mesh could reduce their incidence, although the studies that are currently underway may provide more answers in the near future.
Conflict of InterestsThe authors have no conflict of interests to declare.
Please cite this article as: Hernández-Granados P, López-Cano M, Morales-Conde S, Muysoms F, García-Alamino J, Pereira-Rodríguez JA. Profilaxis de la hernia incisional y utilización de mallas. Revisión narrativa. Cir Esp. 2018;96:76–87.