Tinnitus is defined as the conscious perception of a sensation of sound that occurs in the absence of an external stimulus. This audiological symptom affects 7–19% of the adult population. The aim of this study is to describe the associated comorbidities present in patients with tinnitus using joint and conditional probability analysis.
PatientsPatients of both genders, diagnosed with unilateral or bilateral tinnitus, aged between 20 and 45 years, and who had a full computerised medical record, were selected.
MethodsStudy groups were formed on the basis of the following clinical aspects: (1) audiological findings; (2) vestibular findings; (3) comorbidities such as, temporomandibular dysfunction, tubal dysfunction, otosclerosis and, (4) triggering factors of tinnitus noise exposure, respiratory tract infection, use of ototoxic and/or drugs.
ResultsOf the patients with tinnitus, 27 (65%) reported hearing loss, 11 (26.19%) temporomandibular dysfunction, and 11 (26.19%) with vestibular disorders. When performing the joint probability analysis, it was found that the probability that a patient with tinnitus having hearing loss was 2742 0.65, and 2042 0.47 for bilateral type. The result for P(A ∩ B)=30%. Bayes’ theorem P(AiB) = P(Ai∩B)P(B) was used, and various probabilities were calculated. Therefore, in patients with temporomandibular dysfunction and vestibular disorders, a posterior probability of P(Aі/B)=31.44% was calculated.
ConclusionsConsideration should be given to the joint and conditional probability approach as tools for the study of different pathologies.
El acúfeno se define como la percepción consciente de una sensación sonora en ausencia de un estímulo externo que lo produzca. Este síntoma audiológico, afecta del 7 al 19% de la población adulta. El objetivo del este trabajo es describir las comorbilidades asociadas presentes en los pacientes con acúfeno, mediante el análisis de probabilidad conjunta y condicional.
PacientesSe seleccionaron pacientes diagnosticados con acúfeno unilateral o bilateral, de ambos géneros, con edades comprendidas entre los 20 y 45 años; con expediente electrónico completo.
Material y métodosSe formaron grupos de estudio en base a los siguientes aspectos clínicos: 1) hallazgos audiológicos; 2) hallazgos vestibulares; 3) comorbilidades tales como disfunción temporomadibular, disfunción tubaria, otoesclerosis, y 4) factores desencadenantes del acúfeno como exposición a Noise, infección de vías respiratorias, uso de ototóxicos y/o drogas.
ResultadosDel total de pacientes con acúfeno, 27 (65%) pacientes refirieron hipoacusia; 11 (26.19%) disfunción temporomandibular y 11 (26.19%) alteraciones vestibulares. Al realizar el análisis de probabilidad conjunta, se obtuvo que la probabilidad de que un paciente con acúfeno tenga hipoacusia fue 2742 0.65 y que la hipoacusia sea de tipo bilateral 2042 0.47, el resultado para P(A ∩ B)=30%. Se utilizó el teorema de Bayes P(AiB) = P(Ai∩B)P(B) y se calcularon diversas probabilidades; así, en los pacientes con disfunción temporomandibular y con alteraciones vestibulares, la probabilidad a posteriori calculada fue P(Aі/B)=31.44%.
ConclusionesSe debe considerar la probabilidad conjunta y condicional como herramientas de aproximación probabilística para el estudio de las diversas patologías.
Tinnitus is defined as the conscious perception of a sensation of sound that occurs in the absence of an external stimulus.1 This audiological symptom affects 7–19% of the adult population.2 In England, Baguley reported a prevalence of 10.1%, and classified tinnitus in accordance with the degree of discomfort into: moderate (2.8%), severe (1.6%) and inability to carry out a normal life (0.5%).3
In the United States the reported incidence was 8.2%.4 It has been estimated that in Germany approximately 1.5 million inhabitants present with tinnitus, of whom 800,000 have medical repercussions which require health care.5 Tinnitus is classified as objective or somatosensory, when the sound is generated by the body and is led to the ear either through the blood or muscular contractions1,4; subjective tinnitus is described as referring to auditory hallucinations or phantom sounds present in psychiatric disorders, such as schizophrenia, or through drug use or changes in the temporal lobe.1,3
It has been proven that there is a higher probability of tinnitus in males4 with hearing problems predominantly in the left ear, over 70 years of age5 and those who have been exposed to noise.1,5,6
In women it is related to disorders such as anxiety and/or depression, as well as psychosomatic illnesses.2
Tinnitus is a subjective sound, which may be considered for clinical diagnosis but certain reserve should be made, given its idiosyncratic perception by each individual, and its possible association with a pathology the patient is suffering from at one particular time. For example in Meniére's disease, patients report that there is a low sound but this is made sharper when they suffer from episodes. In chronic acoustic trauma and in otosclerosis, it is referred to as a high pitch; in chronic otitis media, it is referred to as continuous high or low tone; and in sudden onset of hearing loss it is described as low and with an increase in intensity after exposure to external noise.7
Audiological changes commonly associated with tinnitus are: conductive and neurosensorial hearing loss, presbycusis, acute and chronic acoustic trauma, viral or vascular sudden onset of hearing loss, laberynthitis, autoimmune disease of the inner ear, endolymphatic hydrops and barotrauma.8
Tinnitus may have a different origin, but it has been argued that in 90% of cases it is associated with cochlear dysfunctions.4
It is of note that at present tinnitus is not just considered to be a symptom of cochlear origin, but to be extremely closely linked to the function of the central nervous system.9 Any changes in cochlear micromechanics, ionic homeostasis of calcium and in the biochemistry of synaptic transmission may lead to an abnormal activity in the auditory pathway, which is wrongly interpreted by auditory centres and presents as tinnitus.10
In demyelinising pathologies such as Guillain-Barrè syndrome and in neoplasms such as neurinomas, tinnitus usually presents in 80% of cases.8,10,11
In the auditory processing area, degeneration of type I and type II nerve fibres may trigger tinnitus,12 as may Alzheimer and blood perfusion changes, which have been shown in tomography imaging.13
The presence of tinnitus has been studied in some patients and its relationship with several neurotransmitters. The expression of tinnitus in central processing areas provoked by changes in the metabolic cycle and depolarisations of afferent glutamate neurons has been described.14
Certain lesions in the inferior colliculus reduce the activity of the decarboxylase glutamate enzyme leading to the waning of the synthesis of gamma-aminobutyric acid (GABA) in that area, the consequence of which is a reduction of neural inhibitory activity on the inferior colliculus. The presence of tinnitus is the definitive result of these neurochemical changs.8
Cardiovascular pathologies such as high blood pressure, arrhythmias, heart valve diseases, vasculitis, ictus, arteriosclerosis, arterial brain spasms and changes in microcirculation are factors which are closely associated with the generation of tinnitus.8,15,16 Head injuries and cervical injuries associated with the frequency of traffic accidents are also triggers for tinnitus and other concomitant symptoms such as hearing loss.4
Valente analyses the correlation between the degree of disturbance generated by tinnitus and the degree of hearing loss. His study reveals that tinnitus leads to greater discomfort or disturbance in people who have never had a major hearing loss. It also reaffirms the association between tinnitus and medical symptoms such as hearing loss, dizziness, neck pain, headache and excessive intake of caffeine and carbohydrates.6
According to Andersson, headache is associated with the intensity of tinnitus, with a prevalence of 23.6% of cases17; Peñaloza-Cárdenas mentions that metabolic diseases, such as dyslipidemia, thyroid changes and diabetes having a major association with this symptom.18
It is known that tinnitus induces activation of the limbic system, and in particular the amygdala and hypothalamus, which leads to a state of emotional instability altering the quality of life of the person suffering from it.11 It has also been demonstrated that it may be triggered and/or aggravated when associated with psychological aspects such as anxiety, depression, stress, trauma and periods of grief.8,12,19 Andersson reports that these patients may develop cognitive symptoms such as a lack of concentration, problems with memory, states of confusion, difficulty to converse, and inability to carry out everyday and/or working activities.17
Alterations in sleep are a common consequence of this symptom, since patients suffer from insomnia, an increase in the amount of times they wake up, a sensation of sleep being non-restorative and drowsiness during the day, with a prevalence ranging between 16% and 57%.20 Andersson states that patients with tinnitus and sleep disorders present with states of neurosis, anxiety and depression, and may become isolated from society and family in up to 61% of cases.17
Tinnitus is referred to as a symptom which may affect people's quality of life and in serious cases may even lead to disability. Due to the subjectivity of symptoms its phyisopathological mechanisms have not been completely clarified. This type of study in patients of working age who present with tinnitus is essential in order to discover the risk factors and pathologies associated with this condition. Our study is based on joint probability analysis through the intersection of variables and conditional probability analysis which establish the rates of pathologies involved in the presentation of tinnitus through analysis of proportions and the calculation of probability a posteriori. The aim of this study is to describe the associated comorbidities present in patients with tinnitus using joint and conditional probability analysis.
MethodologyStudy typeRetrospective, descriptive.
Sample typeCensus sampling: the study included all patients who presented with tinnitus and who were attended by the National Rehabilitation Institute Luis Guillermo Ibarra Ibarra (INR-LGII), the audiology and otoneurology services, between January and December 2013.
Study participantsPatients diagnosed with unilateral or bilateral tinnitus were selected. Patients were of both genders, aged between 20 and 45, who had a full computerised medical record, clinical history, anamnesis, otoscopy, audiometry and impedancometry. Patients who were not within the established age range were excluded, as were those who suffered with chronic degenerative diseases, an incomplete medical record, or high blood pressure and diabetes mellitus.
ProcedureElectronic files of 200 patients were reviewed using the Hospital information computerised system. 42 patients were elected in accordance with the inclusion and exclusion criteria.
Variables which were analysed in the selected patients were as follows: tone, laterality and medical history.
Study groups were formed based on the following clinical traits:
- 1)
Audiological clinical findings such as degree, laterality and type of hearing loss.
- 2)
Vesticular symptomatology: dizziness and vertigo.
- 3)
Other comorbidities including temporomandibular dysfunction (TMD), tubal dysfunction otosclerosis, depression, and middle ear pathology.
- 4)
Triggering factors of tinnitus such as exposure to noise, use of ototoxics, respiratory tract infections, drug and medication usage.
Contingency tables were established and intersections were made to determine the conditional probabilities between variables.
The protocol was approved by the research committee of the National rehabilitation Institute LGII. The authors have no conflict of interest to declare.
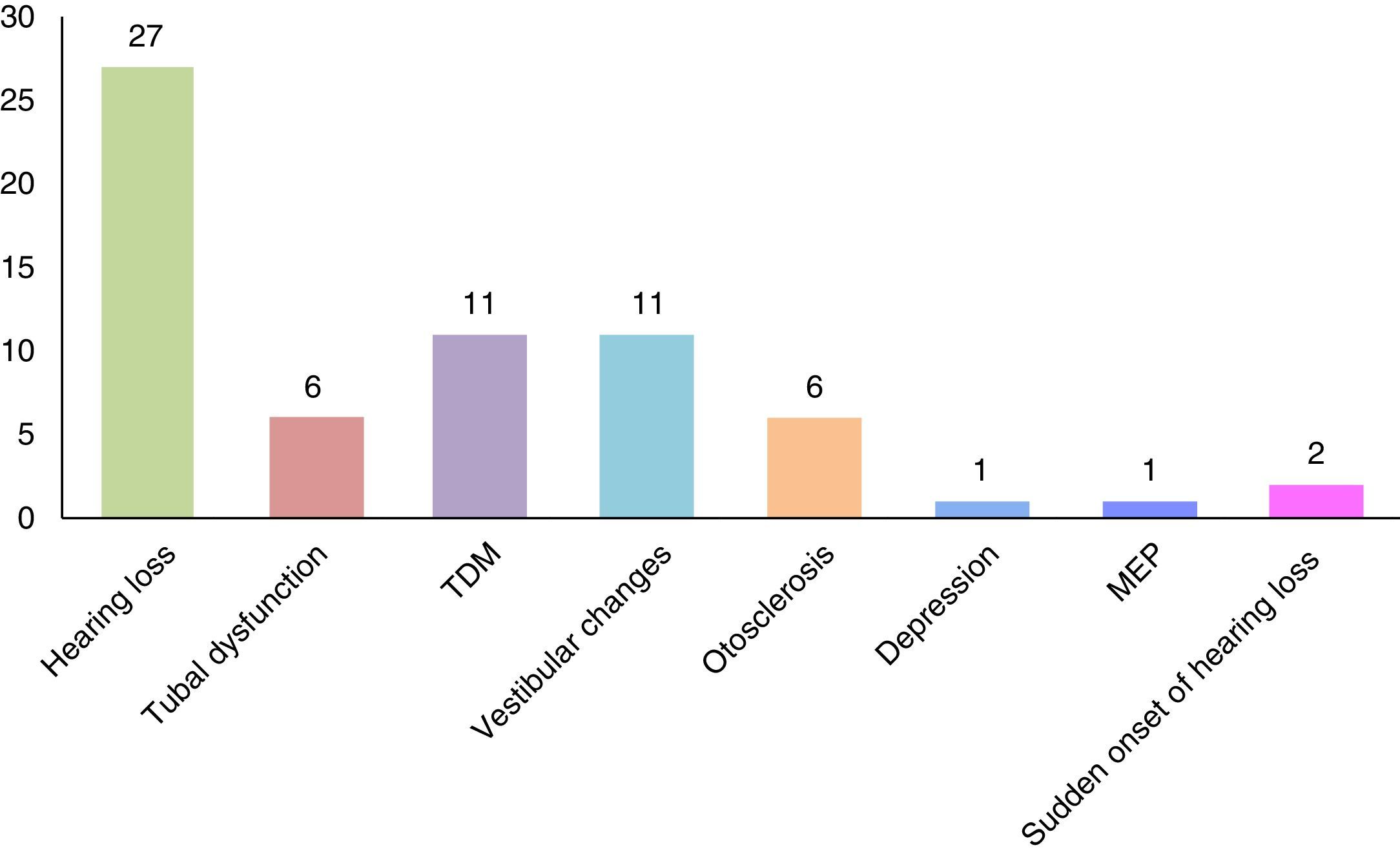
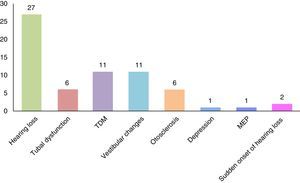
ResultsThe sample consisted of 42 patients of both genders, 18 of whom (42.86%) were male, with an average age of 33.78±5.48 and 24 of whom were female (57.14%), with an average age of 31.29±6.84; The age range in females was 20–40 years, and in males 22–42. The comorbidity percentages present in the patients with tinnitus were obtained; findings indicated that 27 (65%) patients presented with hearing loss, and this was the primary pathology associated with tinnitus; 11 (26.19%) TMD; 11 (26.19%) vestibular changes; 6 (14.28%) tubal dysfunction; 6 (14.28%) otosclerosis; 2 (4.76%) sudden onset of hearing loss; 1 (2.38%) depression, and 1 (2.38%) middle ear pathology (Fig. 1).
Joint probability using the intersection of variables was calculated, to determine the proportion of comorbidity presentation associated with tinnitus, the formula P(A ∩ B)=P(A) · P(B) was used, where A and B, were calculated. On conducting joint probability analysis P(A ∩ B) where the probability of a patient with tinnitus (A) had hearing loss 2742 0.65 and the hearing loss was bilateral (B) 2042 0.47, the result for P(A ∩ B)=0.30 or 30%; in the intersection P(A ∩ B) formed by patients with hearing loss a 2742 0.65 and where hearing loss was unilateral 742 0.16, the result is 10%. The ratio of patients with tinnitus who presented with hearing loss (A) 2742 0.65 was calculated, which was bilateral (B) 2042 0.47 and of sensory type (C) 2142 0.5, joint probability calculated was P(A ∩ B ∩ C)=0.15,or 15%; finally, on analysing the intersection for hearing loss patients with (A) tinnitus 2742 0.65, (B) unilateral hearing loss 742 0.16 and (C) left sensory type with normal hearing in the right ear 442, the result of its joint probability P(A ∩ B ∩ C)=0.0097.
35% of patients with tinnitus did not present with hearing loss (Table 1).
Joint probability of patients with tinnitus and hearing loss.
| A/B proportion | Proportion outcome | Percentage | |
|---|---|---|---|
| Normal hearing patients did not present signs of hearing loss | 1542 | (0.35) | 35% |
| Patients with hearing loss | 2742 | (0.65) | 65% |
| Intersection (A∩B) | Intersection outcome P(A∩B) | % | |
| Bilateral hearing loss | 2742∩2042 | (0.65) (0.47)=0.30 | 30% |
| Unilateral hearing loss | 2742∩742 | (0.65) (0.16)=0.10 | 10% |
| Sensory bilateral hearing loss | 2742∩2042∩2142 | (0.65) (0.47) (0.5)=0.15 | 15% |
| Left sensory unilateral hearing loss with normal hearing in right ear | 2742∩742∩442 | (0.65) (0.16) (0.095)=0.009 | 0.97% |
Calculations of the intersections which are shown in this work were made taking as reference all study participants: N=42.
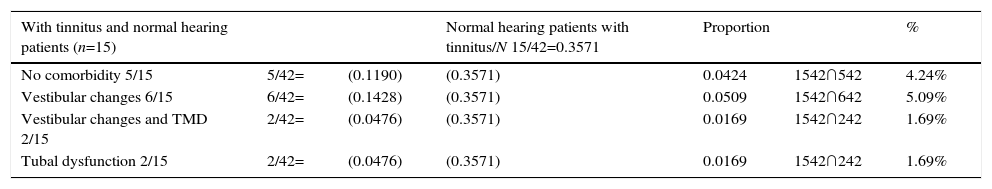
Joint probability P(A ∩ B) for patients with tinnitus and with normal hearing was also calculated. The intersection between normal hearing patients with (A) tinnitus 1542 and (B) with no comorbidities 542 corresponded to the value P(A ∩ B)=0.04. In the intersection of normal hearing patients (A) with tinnitus 1542 and (B) patients with vestibular changes 642 the intersection value was P(A ∩ B)=0.05; in other words, the probability that a patient with tinnitus and normal hearing had vestibular changes was 5.09%. Finally, in the intersection of patients with (A) tinnitus and no hearing loss 1542, with patients who presented with (B) vestibular changes and TMD 242, and those patients with (B) tubal dysfunction 242, the conditional probability value in both intersections P(A ∩ B)=0.01 (Table 2).
Joint probability among normal hearing patients and tinnitus.
| With tinnitus and normal hearing patients (n=15) | Normal hearing patients with tinnitus/N 15/42=0.3571 | Proportion | % | |||
|---|---|---|---|---|---|---|
| No comorbidity 5/15 | 5/42= | (0.1190) | (0.3571) | 0.0424 | 1542∩542 | 4.24% |
| Vestibular changes 6/15 | 6/42= | (0.1428) | (0.3571) | 0.0509 | 1542∩642 | 5.09% |
| Vestibular changes and TMD 2/15 | 2/42= | (0.0476) | (0.3571) | 0.0169 | 1542∩242 | 1.69% |
| Tubal dysfunction 2/15 | 2/42= | (0.0476) | (0.3571) | 0.0169 | 1542∩242 | 1.69% |
TMD: temporomandibular dysfunction.
The greatest probability calculated in patients with tinnitus and normal hearing was in those who presented with vestibular changes.
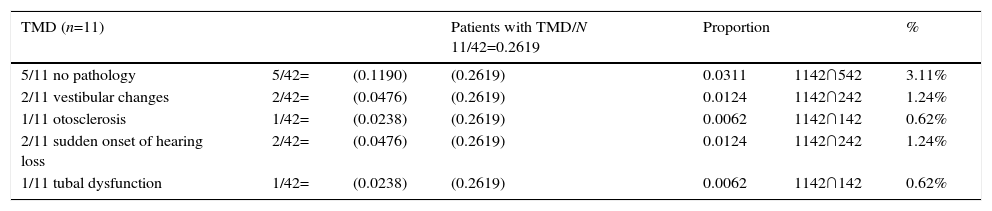
TMD is an entity associated with the presence of tinnitus; through joint probability analysis the proportion was obtained with P(A ∩ B)=P(A) · P(B). The probability P(A ∩ B) that a patient with (A) tinnitus has TMD 1142 and does not present with (B) any associated pathology 542 was P(A ∩ B)=0.03; in other words, 3.11%; on calculating the intersections between the patients with (A) tinnitus who have TMD 1142 with (B) vestibular changes 242, the result of P(A ∩ B)=0.0124, the same value was calculated for the patients with sudden onset of hearing loss. On calculating P(A ∩ B) that a patient with (A) tinnitus has TMD 1142 and presents with otosclerosis 142, the value of the intersection P(A ∩ B)=0.0062: or 0.6% (Table 3).
Joint probability between tinnitus and temporomandibular dysfunction.
| TMD (n=11) | Patients with TMD/N 11/42=0.2619 | Proportion | % | |||
|---|---|---|---|---|---|---|
| 5/11 no pathology | 5/42= | (0.1190) | (0.2619) | 0.0311 | 1142∩542 | 3.11% |
| 2/11 vestibular changes | 2/42= | (0.0476) | (0.2619) | 0.0124 | 1142∩242 | 1.24% |
| 1/11 otosclerosis | 1/42= | (0.0238) | (0.2619) | 0.0062 | 1142∩142 | 0.62% |
| 2/11 sudden onset of hearing loss | 2/42= | (0.0476) | (0.2619) | 0.0124 | 1142∩242 | 1.24% |
| 1/11 tubal dysfunction | 1/42= | (0.0238) | (0.2619) | 0.0062 | 1142∩142 | 0.62% |
TMD: temporomandibular dysfunction.
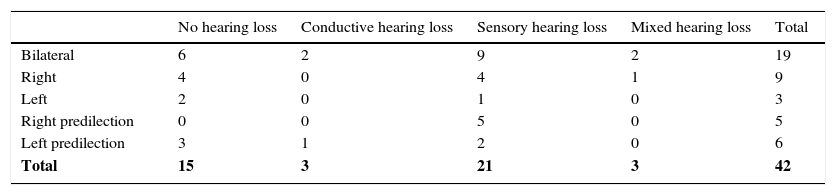
Tinnitus is a subjective symptom which is perceived in different ways. When describing tinnitus in participants who did not present with auditory perception disorders 15 (35.71%), laterality was identified in the following way: 6 patients (40%) presented with bilateral tinnitus, 4 (26.66%) in the right ear, 2 (13.33%) in the left ear and 3 (20%) bilateral with left predominance.
In patients with tinnitus and conductive hearing loss, laterality observed was described as follows: one patient (2.38%) with bilateral conductive hearing loss had bilateral type tinnitus; another patient (2.38%) with left sensory hearing loss and conductive hearing loss in the right ear also had bilateral tinnitus and one (2.38%) patient with conductive hearing loss in the right ear and mixed left hearing loss had predominant left bilateral tinnitus.
On calculating the proportion of patients with tinnitus who stated they had sensory type hearing loss P(A ∩ B) = 2742∩2142 the result of the intersection was P(A ∩ B)=0.3214; this means that 32.14% of patients with tinnitus had sensory hearing loss. On carrying out the calculation of the intersection for P(A ∩ B) = 2742∩2142∩942, the result was P(A ∩ B)=0.0688; this means that 6.88% of patients of this study present with sensory hearing loss and bilateral type tinnitus. The majority of patients presented with bilateral sensory hearing loss, with 9 cases (Table 4).
Type of hearing loss and tinnitus laterality.
| No hearing loss | Conductive hearing loss | Sensory hearing loss | Mixed hearing loss | Total | |
|---|---|---|---|---|---|
| Bilateral | 6 | 2 | 9 | 2 | 19 |
| Right | 4 | 0 | 4 | 1 | 9 |
| Left | 2 | 0 | 1 | 0 | 3 |
| Right predilection | 0 | 0 | 5 | 0 | 5 |
| Left predilection | 3 | 1 | 2 | 0 | 6 |
| Total | 15 | 3 | 21 | 3 | 42 |
It is noted that tinnitus predominates in sensory hearing loss patients and most commonly bilaterally.
With regard to the audiological backgrounds which may have an effect on the presentation of tinnitus, 12 patients (28.57%) were found not to have any relevant clinical histories, whilst 30 (71.42%) patients did have major histories. Out of these, 18 (60%) reported only one outstanding factor and 12 (40%) reported more than one major factor. The main histories referred to by patients were: exposure to noise from heavy machinery, being factory workers or chauffeurs, including: medical treatment with amino glycosides (amikacin, gentamicin), drug intake (tobacco, alcohol and banned psychotropic drugs), recurrent airway infections and head injury trauma. Of the 18 patients who attributed the presence of tinnitus to a single condition, 3 (16.66%) said they had been exposed to noise, 6 (33.33%) to having been medicated with ototoxics, 6 (33.33%) to the ingestion of some type of drug, 1 (5.55%) to infection of upper respiratory tract infection (URTI) and 2 (11.11%) to head injury (HI). The 12 patients who had more than one conditoin associated with tinnitus, were divided as follows: 4 (33.33%) presented with exposure to noise and ototoxics, 3 (25%) to exposure to noise and drugs, 2 (16.66%) to exposure to noise and recurrent airway infections, 2 (16.66%) to exposure to ototoxics and upper respiratory tract infections, 1 (8.33%) to use of ototoxics and drugs. To determine whether there was a link between variables (otologic background), the Chi-square test was conducted with a confidence level of p<0.05. The value obtained was 9.18<26.29, which means that there was no association between the otologic history and presentation of tinnitus (Table 5).
Otologic history and tinnitus.
| Pathologies associated with tinnitus | Total | |||||
|---|---|---|---|---|---|---|
| 1 history | Noise 3 | Ototoxics 6 | Drugs 6 | URTI 1 | HI 2 | 18 |
| 2 History | Noise/ototoxics 4 | Noise/drugs 3 | Noise/URTI 2 | Ototoxics/drugs 1 | Ototoxics/URTI 2 | 12 |
URTI: upper respiratory tract infection; HI: head injuries.
It may be observed in the table that ototoxics and drug use are the factors of greatest incidence leading to the presentation of tinnitus.
With regards to the tinnitus tone and time of evolution, the high tone presented in 23 (54.76%) of patients and the low tone in 17 (40.47%); in 2 (4.76%) the tone was variable. The patients who had tinnitus with a high tone went for audiologic consultation on average at 22.57±37.38 months of evolution, whilst those who describe their tone as low went on average at 18.88±30.78 months. In the case of patients with variable tone they went at 6.50±3.53 months. This indicates that the low tone is more disturbing and patients seek consultation earlier. To determine whether an association between variables exists (associated pathologies and tinnitus tone), the chi-test was conducted with a confidence value of p<0.05. The value obtained was 6.30<31.41, which means that there is no link between associated pathologies and the presentation of tinnitus.
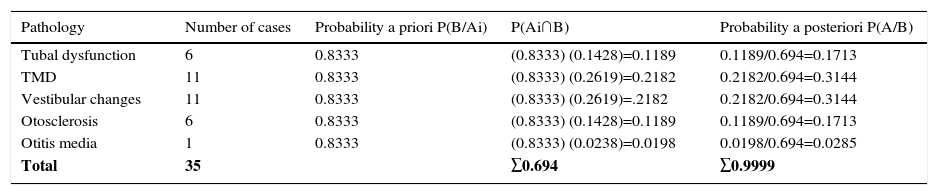
Bayesian analysis. Posteriori probabilityConditional probability of tinnitus was calculated with the following associated comorbidities: tubal dysfunction, TMD, vestibular changes, otosclerosis and otitis media, used in Bayes’ theorem P(AiB) = P(Ai∩B)P(B) and the probability a priori was calculated as a result of the total ratio of patients with no associated pathology (35/42) which was P(B/Aі)=0.8333. On calculating probability a posteriori of the patients with tinnitus and tubal dysfunction, the probability calculated was P(Aі/B)=0.1713 or 17.13%; in patients with TMD, probability a posteriori calculated was P(Aі/B)=0.3144 or 31.44%, similar to patients with vestibular changes. In patients with otosclerosis probability a posteriori calculated was P(Aі/B)=0.1713 or 17.13% and in patients with tinnitus associated with otitis media, the value of the probability a posteriori calculated was established at P(Aі/B)=0.0285 or 2.85% (Table 6). It is worth noting that 12 patients who showed no changes to audition were not included in this table.
Bayesian Analysis. List of comorbidities associated with tinnitus.
| Pathology | Number of cases | Probability a priori P(B/Aі) | P(Aі∩B) | Probability a posteriori P(A/B) |
|---|---|---|---|---|
| Tubal dysfunction | 6 | 0.8333 | (0.8333) (0.1428)=0.1189 | 0.1189/0.694=0.1713 |
| TMD | 11 | 0.8333 | (0.8333) (0.2619)=0.2182 | 0.2182/0.694=0.3144 |
| Vestibular changes | 11 | 0.8333 | (0.8333) (0.2619)=.2182 | 0.2182/0.694=0.3144 |
| Otosclerosis | 6 | 0.8333 | (0.8333) (0.1428)=0.1189 | 0.1189/0.694=0.1713 |
| Otitis media | 1 | 0.8333 | (0.8333) (0.0238)=0.0198 | 0.0198/0.694=0.0285 |
| Total | 35 | ∑0.694 | ∑0.9999 |
TMD: temporomandibular dysfunction.
This work describes associated comorbidities, the main symptoms and aspects such as laterality and the tinnitus tone in patients who attended the INR-LGII (tertiary hospital of the City of Mexico) in the period between January and December 2013.
It has been estimated that between 10% and 15% of the worldwide population have suffered from tinnitus at least once.1,6 Martines Clos et al. refer to the fact that tinnitus may present at any age with no predominance of gender, although incidence does tend to be higher5 in males, probably due to greater exposure to noise in a working environment. However, in the population studied in INR-LGII, tinnitus was more prevalent in females (57.14%), rather than males (42.86%).
A major aspect of this work is the homogeneity of the population studied, since the average age of the patients ranged between 33.78 and 31.29 years for men and women, respectively. The lower age of the range was 20 in both cases. As a result, the clinical finding that patients with tinnitus are at a potentially productive age gathers momentum, and should lead to the proposal for strategies designed to resolve those cases where tinnitus is a disabling factor both emotionally and for a productive working life. Reports have thus described heterogeneous populations with age ranges between 22 and 90.13
With regards to the main factors associated with the presence of tinnitus, our findings reveal that 65% of patients have hearing loss, similar data to those reported by Vernon and Meikle, who pointed out that between 70% and 80% of patients with tinnitus suffer from alterations to their hearing.21 Apart from hearing loss, other percentages referred to in this work are for TMD and vestibular changes, in 26.19% of cases. Tubal dysfunction and otosclerosis also make up 14.28%.
Lockwood states that exposure to noise, presbycusis, otosclerosis, otitis, cerumen impaction, Menière's disease and sensory neural hearing loss are the most important comorbidities associated with tinnitus.22 For his part, Henry states that hearing trauma, head and neck trauma, airway infection, otitis, allergies, stress and TMD, are associated factors.7 Both Lockwood and Henry highlight associated pathologies but do not provide any indication or reference percentage.
Several studies report that sensory hearing loss is one of the main findings in patients with tinnitus, but they do not determine any probalistic causation.5,7,16,21 Our work therefore grows in importance, since it allows us to calculate the intersection of variables. For example what would be the probability that a patient with tinnitus presents with hearing loss and that it be sensory?, P(A ∩ B)=0.32; or what probability is there that a patient with tinnitus would have sensory hearing loss and that it would be bilateral? P(A ∩ B)=0.068. This type of analysis increases the relevance of the study since joint probability analysis is another tool for the analysis of association between pathologies.
Hearing loss is the main pathology associated with tinnitus, only 35% of patients show no signs of it. This behaviour is understood thanks to the different triggers of hearing loss which may vary from an obstructive mechanical process at middle ear level to a loss of hearing from a head injury or central neurological hearing pathology.4,7,13,21,23
With regard to tinnitus tone, it was found that the high tone predominates in 54.76%, the low tone in 40.47% of cases and a variable tone in 4.76% of cases. The literature mentions that the tone may be associated with the pathology, for example patients with sudden onset of deafness had a low tone tinnitus. Those who suffered from otosclerosis and vestibular changes suffered from a high tone. The high tone was also primarily present in Menière's disease. In cases where there was more than one comobidity there was no tendency towards any particular tone.7
TMD is another comorbidity related to tinnitus, frequently associated with ontological symptoms; it has been estimated that in the United States approximately 10,000,000 people present with it and its prevalence on a worldwide scale is between 19% and 69%, being most frequent in women rather than in men at a 2:1 ratio. Delgado et al. found that 36% of patients with TMD had tinnitus24; in the study carried out in the INR-LGII, it was reported that out of the total patients with tinnitus 26.19% presented with TMD. On calculating the probability a posteriori in patients with TMD plus the presence of tinnitus, the value obtained was P(A/B)=0.3144, i.e. 31.44%. Park et al. studied patients with tubal dysfunction and determined that 28.3% of these patients presented with tinnitus.25 In the sample we analysed the percentage of patients with tinnitus who presented with tubal dysfunction was 14.28%. Otosclerosis is associated with a high incidence of vestibular changes, since it involves impairment of the otic capsule, and as a result sensory or mixed hearing loss often coexists with tinnitus and vertigo.7,15 Gristwood refers to the rate of tinnitus in patients with otosclerosis as being 65%.26
Deggouj suggests that dysfunction of the stapes is one of the primary aethiopathogenic factors for the development of tinnitus in patients with otosclerosis.27 In our study, otosclerosis was prevalent in 14.28% of patients and 17.13% in Bayesian analysis was expressed through probability a posteriori.
With regard to the factors which the patient says have had an impact on the presentation of tinnitus, 30 (71.42%) consider that if there were major background factors 18 (60%) stated there was only one outstanding factor and 12 (40%) stated there were more than one.
According to the literature, exposure to noise and the use of ototoxics are the principal risk factors to triggering tinnitus.28,29 In our study, the main triggers of tinnitus stated by the patients when referring to one only were medication with ototoxics 6/18 (33.33%), intake of drugs (alcohol, tobacco) 6/18 (33.33%), exposure to noise 4/18 (22.22%) and TCE in two cases 2/18 (11.11%).
Furthermore, the 12 patients who referred to more than one background factor associated with the presentation of tinnitus distributed them accordingly: 4 (33.33%) to exposure to noise and ototoxics, 3 (25%) to exposure to noise and drugs, 2 (16.66%) to exposure to noise and recurrent airway infections, 2 (16.66%) to exposure to ototoxics and upper respiratory tractinfecitons, 1 (8.33%) to use of otortoxics and drugs.
The combination is thus that noise is identified as the main associated factor with a frequency of 9 occasions, followed by ototoxics with 7 and less frequently drug usage and respiratory tract infections.
Eggermont reported that 23.6% of patients subjected to prolonged or loud noises had tinnitus, whilst 17.7% was triggered by TCE and neck injury.12 Seidman et al. stated that in patients with schwannoma, 57% developed tinnitus, and finally those patients with a background of depression presented with tinnitus in 34% of cases.15
ConclusionTinnitus is clinically relevant in audiology, since it substantially affects the patient's quality of life. Due to the heterogeneity of tinnitus aetiology is difficult to determine, as is the site of the lesion and therefore the treatment. In this study comobidities were found which are frequently related to the appearance and permanence of tinnitus, such as hearing loss and medication with aminoglycosides. Joint probability analysis and conditional probability are a supportive tool to be considered when clinical protocols are designed for the prevention, control and treatment of this pathology. The aim is to improve the patient's quality of life and enable them to reuptake their daily and workplace activities.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that the procedures followed were in accordance with the regulations of the relevant clinical research ethics committee and with those of the Code of Ethics of the World Medical Association (Declaration of Helsinki).
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors have obtained the written informed consent of the patients or subjects mentioned in the article. The corresponding author is in possession of this document.
Conflict of interestsThe authors have no conflict of interests to declare.
Please cite this article as: Toledo VG, Farfán IG, Verduzco-Mendoza A, Arch-Tirado E. Análisis de probabilidad condicional entre el acúfeno y comorbilidades asociadas en pacientes que acudieron al Instituto Nacional de Rehabilitación-LGII en el periodo 2012-2013. Cir Cir. 2017;85:225–233.