Acne is one of the dermatological pathologies with the highest incidence around the world. It is a multifactorial disease and its treatment can be complex. Propionibacterium acnes play a key role in the inflammation of this dermatosis. Topical antibiotics, including mainly erythromycin and clindamycin, have been used, but there is controversy over their use due to the widely documented bacterial resistance. For this reason a meta-analysis of the publications over the past 10 years is presented in order to confirm this hypothesis.
Material and methodsA search was made of the publications over the past 10 years that included the results of antibiogams of patients with acne. MeSH type searches were performed with the terms “acne vulgaris”, “Propionibacterium acnes”, “topical administration”, “treatment”, “erythromycin”, “clindamycin”, “nadifloxacin”, “antibacterial agent”, “bacterial drug resistance” in PubMed, Ovid, EBSCO, Cochrane, ScienceDirect and ClinicalKey meta-searches.
ResultsA total of 13 articles were found that met the inclusion criteria. The mean odds ratio (OR 1.24, 95% CI) of the articles showed a slight tendency towards resistance of P. acnes.
ConclusionsAn increase in bacterial resistance to topical erythromycin and clindamycin can be confirmed, thus the use of these antibiotics is recommended in selective cases for short periods, and in combination with benzoyl peroxide for the best clinical outcome in patients with acne vulgaris.
El acné es una de las afecciones dermatológicas con mayor incidencia a nivel mundial; su origen es multifactorial y, por lo tanto, su tratamiento puede ser complejo. Propionibacterium acnes tiene un papel primordial en la inflamación de esta dermatosis y para su tratamiento se usan antibióticos tópicos; entre los principales se encuentran eritromicina y clindamicina, en los que se ha documentado una amplia resistencia bacteriana, lo que genera controversia respecto a su uso. Por este motivo se presenta un metaanálisis de las publicaciones de los últimos 10 años para confirmar esta hipótesis.
Material y métodosEn la literatura de los últimos 10 años se buscaron artículos sobre resultados de cultivos con antibiograma de pacientes con acné. Se realizaron búsquedas tipo MeSH con los términos «acne vulgaris», «Propionibacterium acnes», «topical administration», «treatment», «erythromycin», «clindamycin», «nadifloxacin», «antibacterial agent», «bacterial drug resistance», en los metabuscadores PubMed, Ovid, EBSCO, Cochrane, ScienceDirect y ClinicalKey.
ResultadosSe encontraron 13 artículos que cumplieron con los criterios de inclusión. La razón de momios promedio (OR 1.24, IC 95%) de los artículos demostró una ligera tendencia hacia la resistencia de Propionibacterium acnes.
ConclusionesSe confirma el aumento en la resistencia bacteriana de Propionibacterium acnés a eritromicina y clindamicina tópica, por lo que recomendamos el uso de estos antibióticos combinados en casos selectos por periodos cortos y en combinación con peróxido de benzoilo, para obtener el mejor resultado clínico en los pacientes con Acne vulgaris.
Acne is the second major cause for dermatology consultation, therefore it is important to know how to treat it appropriately.1 The treatment guidelines for acne vulgaris establish as their criteria: (a) the clinical variety of acne, classified as comedonal, papulo-postular, nodulocystic and (b) severity.2 The most frequently used treatment for acne, and especially for papulo-postular acne, are topical antibiotics.2,3 Because they have been used indiscriminately, an increased resistance to topical antibiotics4 has been confirmed in Propionibacterium acnes (P. acnes), by means of cultures and antibiograms.5
In order to evaluate whether there is sufficient evidence to consider P. acnes resistant to topical antibiotics, we performed a meta-analysis of the articles published on the subject, and our research covered the last 10 years (2004–2013).
Material and methodsSearch strategyThe literature published from 1 January to 31 December 2013 on the topical treatment was reviewed. The metasearch engines we used were PubMed, Ovid, EBSCO, Cochrane, ScienceDirect and ClinicalKey.
Searches were made using the following MeSH terms: “acne vulgaris”, “Propionibacterium acnes”, “topical administration”, “treatment”, “erythromycin”, “clindamycin”, “nadifloxacin”, “antibacterial agent”, “bacterial drug resistance”.
Selection of studiesStudies published from 1 January 2004 until 31 December 2013 (10 years) and written in English and/or Spanish were chosen. Two authors (Alvarez-Sánchez and Arellano-Mendoza) independently reviewed, assessed and chose the studies for analysis.
Microbiological analysisData relating to the method of handling samples, transport, cultures, isolation and identification of P. acnes were gathered and the antibiogram results of the antibiotics that are available for topical administration (clindamycin, erythromycin and nadifloxacin). They were assessed with results for: sensitivity and resistance, cut-off points of minimum inhibitory concentration or E-strip based on the standards of the European Committee on Antimicrobial Susceptibility Testing and the Clinical and Laboratory Standards Institute.6
Data extractionThe data gathered from published studies were evaluated. The following information was extracted from each study: total number of patients, patients with identification of P. acnes and the abovementioned microbiological data. Any discrepancy between the observers was resolved by consensus or by including a third observer.
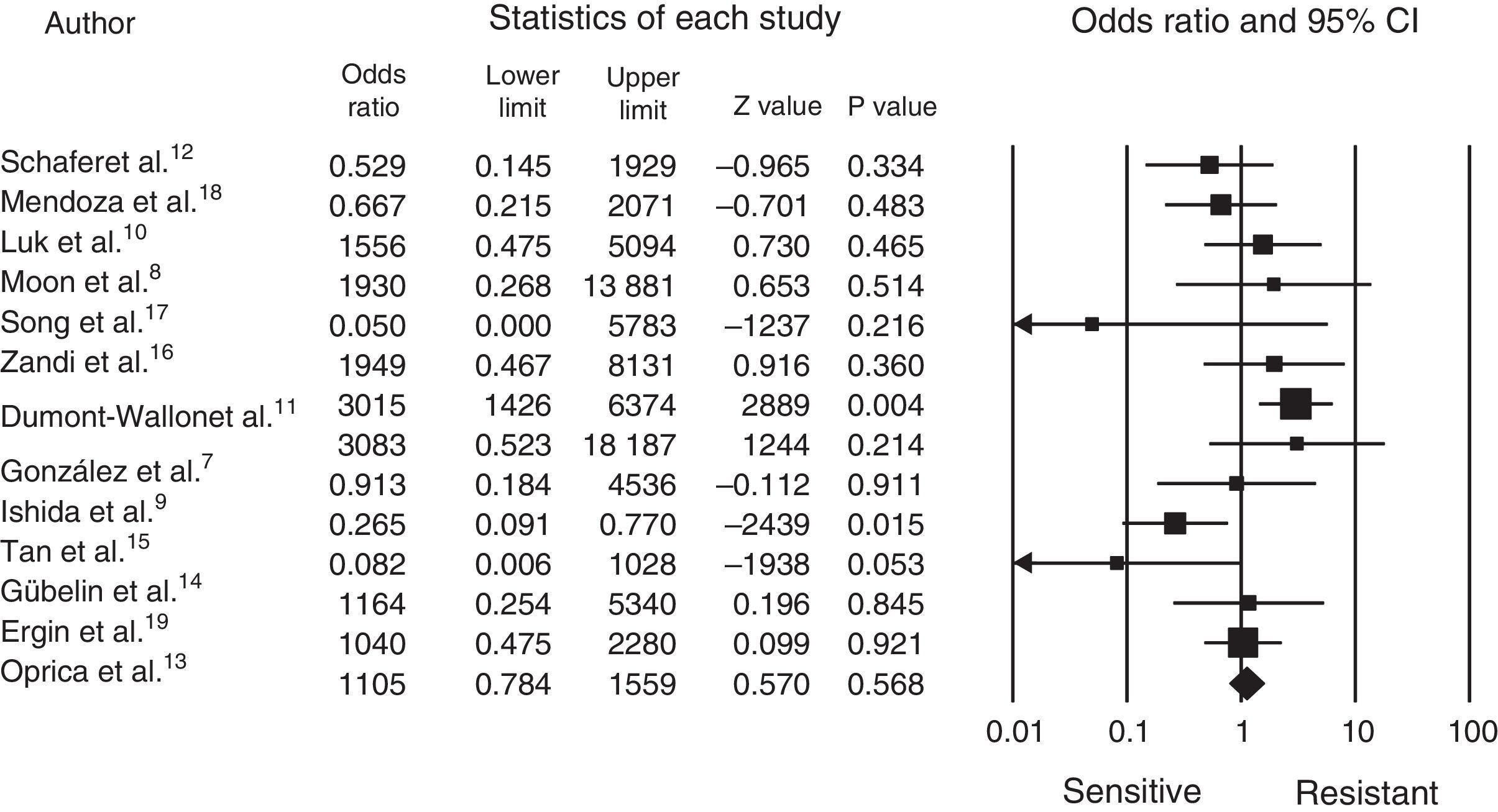
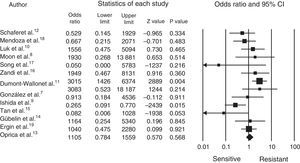
Statistical analysisThe comprehensive meta-analysis (CMA version 2.2.064) software package was used for processing the data, the data was entered and the statistical analysis made, which can be seen in the forest plot (Fig. 1).
The cut-off point for determining resistance or sensitivity is an odds ratio (OR) of 1, where less than 1 indicates sensitivity and greater than 1 indicates resistance; p-values<0.05 were considered statistically significant.
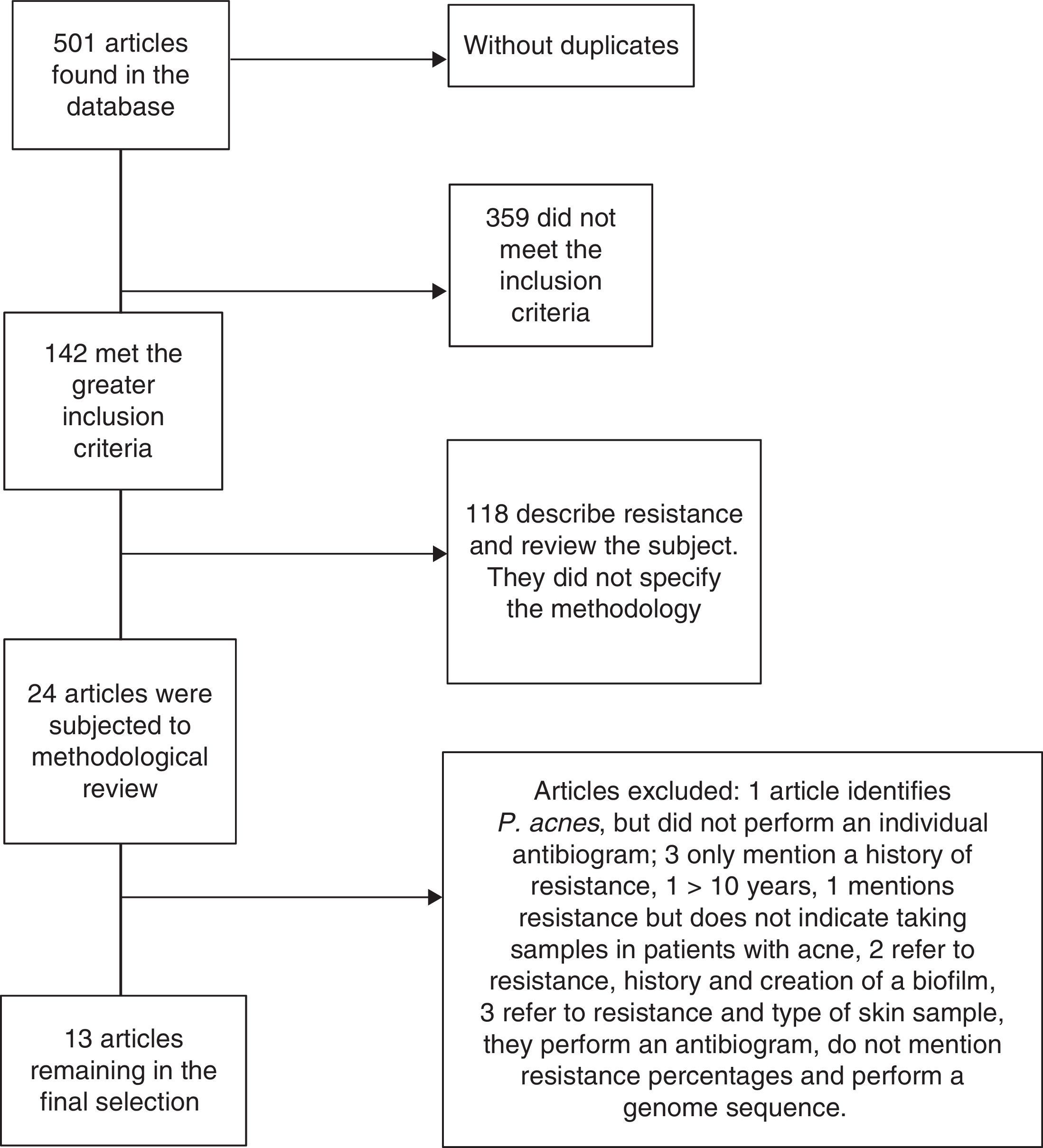
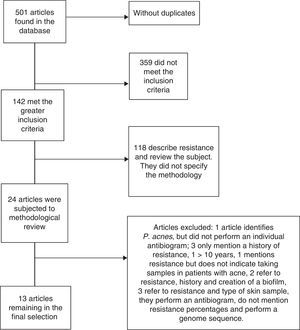
ResultsAround 501 articles were found in the above mentioned search in the period selected (January 2004 to December 2013). We found no duplicated articles, and only 142 of the total met the criteria of identifying P. acnes, and the analysis of resistance and sensitivity with an antibiogram. However, only 24 studies were selected for methodological review and analysis, leaving only 13 which met all the microbiological and clinical data to be analysed; the remaining studies were excluded due to a lack of data or because their methodology was incomplete (Fig. 2).
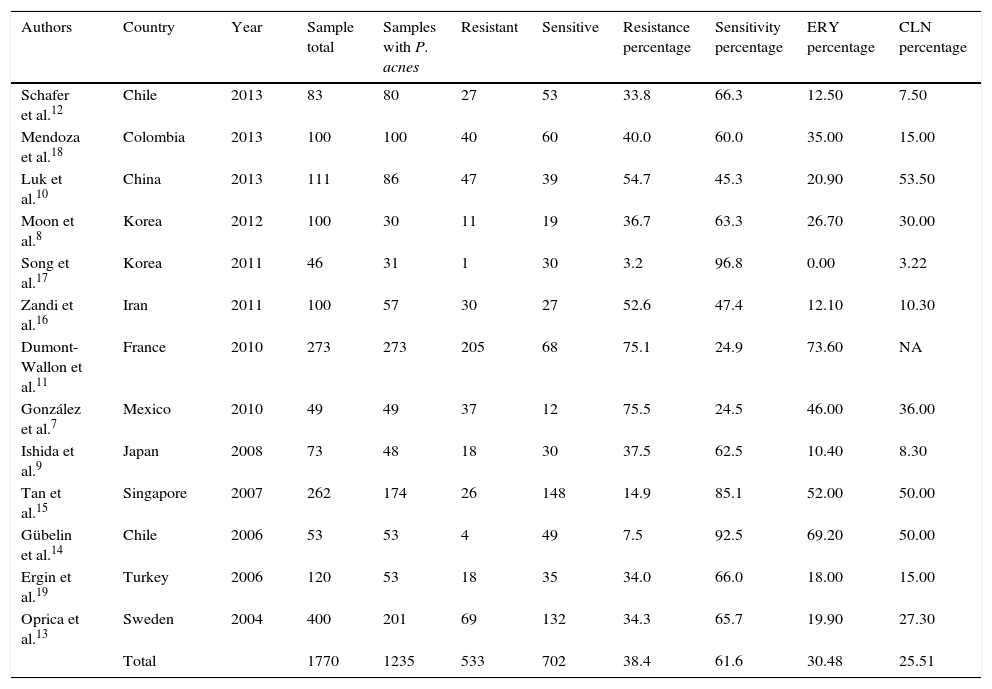
It was established that 13 articles had a classification of cases and controls with a grade IIIB level of evidence and recommendation, without obtaining the clinical trial level, which is preferable for this statistical design (Table 1).7–19
Selected studies.
| Authors | Country | Year | Sample total | Samples with P. acnes | Resistant | Sensitive | Resistance percentage | Sensitivity percentage | ERY percentage | CLN percentage |
|---|---|---|---|---|---|---|---|---|---|---|
| Schafer et al.12 | Chile | 2013 | 83 | 80 | 27 | 53 | 33.8 | 66.3 | 12.50 | 7.50 |
| Mendoza et al.18 | Colombia | 2013 | 100 | 100 | 40 | 60 | 40.0 | 60.0 | 35.00 | 15.00 |
| Luk et al.10 | China | 2013 | 111 | 86 | 47 | 39 | 54.7 | 45.3 | 20.90 | 53.50 |
| Moon et al.8 | Korea | 2012 | 100 | 30 | 11 | 19 | 36.7 | 63.3 | 26.70 | 30.00 |
| Song et al.17 | Korea | 2011 | 46 | 31 | 1 | 30 | 3.2 | 96.8 | 0.00 | 3.22 |
| Zandi et al.16 | Iran | 2011 | 100 | 57 | 30 | 27 | 52.6 | 47.4 | 12.10 | 10.30 |
| Dumont-Wallon et al.11 | France | 2010 | 273 | 273 | 205 | 68 | 75.1 | 24.9 | 73.60 | NA |
| González et al.7 | Mexico | 2010 | 49 | 49 | 37 | 12 | 75.5 | 24.5 | 46.00 | 36.00 |
| Ishida et al.9 | Japan | 2008 | 73 | 48 | 18 | 30 | 37.5 | 62.5 | 10.40 | 8.30 |
| Tan et al.15 | Singapore | 2007 | 262 | 174 | 26 | 148 | 14.9 | 85.1 | 52.00 | 50.00 |
| Gübelin et al.14 | Chile | 2006 | 53 | 53 | 4 | 49 | 7.5 | 92.5 | 69.20 | 50.00 |
| Ergin et al.19 | Turkey | 2006 | 120 | 53 | 18 | 35 | 34.0 | 66.0 | 18.00 | 15.00 |
| Oprica et al.13 | Sweden | 2004 | 400 | 201 | 69 | 132 | 34.3 | 65.7 | 19.90 | 27.30 |
| Total | 1770 | 1235 | 533 | 702 | 38.4 | 61.6 | 30.48 | 25.51 |
CLN: clindamycin; ERY: erythromycin; NA: not assigned; P. acnes: Propionibacterium acnes.
The following data were extracted from the studies chosen: sample with P. acnes, sensitivity and resistance to clindamycin and erythromycin. Nadifloxacin was not found in any of the publications, only levofloxacin was tested, but its administration route meant it was ruled out for the study.7
Once these data had been selected, the odd ratio OR was calculated for each, and the upper limit and the statistical p value in each article.
Fig. 1 shows us how the mean OR is 1.24, with a 95% CI of 0.7048–2.3090. A total of 1770 patients was obtained from the 13 articles, with a range from 40 to 400 and a mean of 136.15; P. acnes was identified in 70% of the patients and resistance to treatment demonstrated in 43%, with sensitivity in 57%. Most of the articles were from different countries, except 2 from Chile (Schafer et al.12 and Gübelin et al.14) and 2 from Korea (Moon et al.8 and Song et al.17). The other countries were: México7 (González et al.), Japan9 (Ishida et al.), China10 (Luk et al.), France11 (Dumont-Wallon et al.), Sweden13 (Oprica et al.), Singapore15 (Tan et al.), Iran16 (Zandi et al.), Colombia18 (Mendoza et al.) and Turkey19 (Ergin et al.). The greatest resistance was observed between 2010 and 2012 (Table 1).
DiscussionAcne is a frequent reason for consultation in our clinical practice.1 In recent years resistance to topical antibiotics has become evident in various publications, principally to topical erythromycin and clindamycin.4 However, in practice its clinical significance has not been fully clarified and they are still used in routine practice to manage these patients, principally for patients with papulo-postular acne. In our review of the literature, we observed that there are few publications containing strict criteria for demonstrating the resistance of P. acnes to topical antibiotics; 7 articles presented an OR greater than 1, which shows a tendency to bacterial resistance. We also found 2 studies which presented excellent sensitivity to antibiotics.14,17 In Song et al. study17 of 31 samples diagnosed with P. acnes, only one isolate was resistant to clindamycin, which moves the general mean of the studies towards sensitivity, and this is also demonstrated with the above mentioned analysis in all of the patients.
Erythromycin and clindamycin are the 2 topical antibiotics which all the articles tested. In the case of erythromycin, the mean resistance percentage in all the publications is 30%, with a maximum of 73.6% in Dumont-Wallon et al. study11 and nil resistance Song et al. study.17 The case of clindamycin is similar, with a resistance mean of 25.5% and a maximum of 53% in Luk et al. study10 with a minimum of 3% in the aforementioned study by Song et al.17
We should highlight that despite the fact that there is a statistical tendency towards bacterial resistance, it is very weak and clinically the topical treatment might fail; however it should be remembered that the combination of topical antibiotics with benzoyl peroxide, as indicated in the treatment guidelines, reduces the incidence of resistance. Recent publications confirm that patients with resistant P. acnes have a clinical benefit if the area is washed with benzoyl peroxide at 6% for 20s before treatment with the topical antibiotic, this reduces bacterial resistance considerably (1 log after one week's treatment and at least 2 log after 3 weeks of treatment).20 In our country, the commercial presentation of benzoyl peroxide at 5% can be found in combination with topical antibiotics, which helps dermatologists to achieve a good therapeutic response in their patients.
We observed that there are few studies that would have made cultures of P. acnes, and those available had an evidence grade IIIB, therefore it is imperative to perform more rigorous clinical–microbiological studies with scientific evidence for a highly prevalent disease like acne vulgaris.
The resistance indices observed in the studies might be lower, when short courses of antibiotics were used and in combination with benzoyl peroxide.
In conclusion, a tendency towards P. acnes bacterial resistance has been found in patients with acne vulgaris. Monotherapy using clindamycin and erythromycin should not be the basis for treatment of this condition; these topical antibiotics can be used in very select cases, for short periods, and should always be used in combination with benzoyl peroxide to diminish, reduce inflammation and avoid P. acnes bacterial resistance in patients with acne vulgaris.
We recommend that clinicians should use the evidence found in this meta-analysis in order to select the best treatment and obtain the best results for their patients with acne vulgaris.
Conflict of interestsThe authors have no conflict of interest to declare.
Please cite this article as: Alvarez-Sánchez M, Rodríguez-Ayala E, Ponce-Olivera RM, Tirado-Sánchez A, Arellano-Mendoza MI. ¿Resistencia en el acné? Un metaanálisis a propósito de la controversia. Cirugía y Cirujanos. 2016;84:190–195.