Necrotising enterocolitis is the most lethal gastrointestinal emergency in the neonatal period. Incidence and mortality have remained stable in recent years despite advances in neonatal intensive care.
The aim of this study is to show the general characteristics of patients diagnosed with necrotising enterocolitis at Teresa Herrera's Hospital (La Coruna, Spain) in the last 12 years.
Material and methodsThis study makes a retrospective and prospective descriptive analysis, evaluating the medical records and collecting radiological demographic variables, gestational data, perinatal history, clinical, analytical and perinatal therapeutic management and events in patients diagnosed with and treated for necrotising enterocolitis between 2003 and 2015.
ResultsA total of 124 patients met the criteria for inclusion in the study. The mean gestational age of our patients was 33 weeks and remained stable compared with other studies. The average weight of our patients was 1873g. In our series of cases there was a progressive and significant increase in maternal age and the rate of artificial pregnancies and multiple births. In our series 38.7% of our patients required surgical treatment and the fatality rate was 11.4%.
DiscussionDespite advances in pre- and perinatal care necrotising enterocolitis represents the leading cause of premature mortality. Incidence has remained unchanged in recent decades. More studies are required to identify both, risk and protective factors to reduce the morbidity and mortality of this entity.
La enterocolitis necrosante neonatal es la emergencia gastrointestinal de mayor letalidad. La incidencia y mortalidad permanecen estables en los últimos años a pesar de los avances en los cuidados intensivos neonatales.
El objetivo de este estudio es describir las características generales de los pacientes con enterocolitis necrosante diagnosticados en el Hospital Teresa Herrera de La Coruña, España, en los últimos 12años.
Material y métodosSe realiza un estudio descriptivo, retrospectivo y prospectivo evaluando las historias clínicas y recogiendo variables demográficas, datos gestacionales, antecedentes perinatales, manifestaciones clínicas, analíticas y radiológicas, manejo terapéutico perinatal y eventos en el seguimiento de los pacientes con enterocolitis necrosante diagnosticados y tratados entre los años 2003 y 2015.
ResultadosUn total de 124 pacientes cumplen criterios de inclusión en el estudio. La media de edad gestacional es de 33 semanas y permanece estable en oposición a otros estudios. El peso medio de nuestros pacientes se sitúa en 1,873g. En nuestra serie de casos ha habido un progresivo y significativo aumento de la edad materna, así como de la tasa de gestaciones artificiales y nacimientos múltiples. Han requerido tratamiento quirúrgico el 38.7% de nuestros pacientes y la tasa de letalidad se sitúa en el 11.4%.
DiscusiónLa enterocolitis necrosante neonatal representa, a pesar de los avances en los cuidados pre y perinatales, la principal causa de mortalidad en el paciente prematuro, con una incidencia y letalidad estables en las últimas décadas. Se necesitan más estudios reglados que permitan identificar tanto factores de riesgo como protectores para disminuir la morbimortalidad de esta entidad.
Necrosing enterocolitis is a devastating disease that constitutes a gastrointestinal emergency and most commonly affects newborns, with high associated mortality.
The term “necrosing enterocolitis” was first mentioned in the European literature in the nineteen-fifties, when Schmid and Quaiser described a clinical picture causing the death of children through necrotic lesions in the gastrointestinal tract.1 However, it was not until 1960 when Barlow presented a series of 64 patients with enterocolitis, that it was acknowledged as a defined clinical entity.2
The incidence varies according to the series from 0.5 to 5 per 1000 live births.3 It affects preterm infants almost exclusively, particularly those of very low birth weight. Its incidence increases inversely proportionate to gestational age and birth weight,4 a rate of between 6% and 7% is estimated in neonates of very low birth weight (less than 1500g),5,6 which increases to 14% in newborns weighing less than 1000g.7
Up to 90% of those affected are under 34 weeks’ gestation.8
Epidemiologically this disorder represents 7.7 per 100 admissions to neonatal care units.9 The mean gestational age is around 31 weeks, with a birth weight of 1400g.
In recent decades, various studies have estimated an incidence that has remained stable due to a drop in the incidence in children born at term or only slightly preterm, and patients affected in the presurfactant era, but has increased in extremely preterm infants who before did not survive.10
Thus, the increased incidence in extremely preterm babies in recent decades is associated with different factors: increased maternal age, a greater rate of pregnancies through assisted reproduction techniques, and improved pre and perinatal care. This results in the increased survival of preterm newborns, who are currently the population at greatest risk of developing this condition.
Similarly, despite the medical advances in the management of these patients in intensive care units, the associated mortality remains practically unchanged,11 and is estimated at between 20% and 40%,12 and up to 50% when the patient requires surgical intervention.13
Most cases of necrosing enterocolitis present sporadically. However, it also occurs in temporary and geographic epidemic outbreaks, where different characteristics are found, such as higher birth weight, fewer complications and higher survival rates, which also endorses the infectious origin of this disorder.
Despite the numerous studies undertaken, the physiopathology of this disease is still not well defined. Various theories and injurious mechanisms have been proposed to explain the aetiology of enterocolitis.
Despite the research of recent decades, the aetiology is still not well defined and one single mechanism does not explain the pathogenesis of the disease. In most cases, multiple factors have been identified as responsible for triggering a sequence of events that end in intestinal necrosis. These “trigger events” can act in isolation or together, not all of them need to be present simultaneously. Therefore, it is considered that there are 3 determining factors for this entity: prematurity, bacterial colonisation and early enteral feeding.14
Most authors agree, therefore, that the disease is the end result of a multifactorial process in a predisposed patient, and causes an inflammatory response with necrosis of the intestinal wall.
The clinical picture of these patients is very varied, from abdominal distension or feeding intolerance to sudden, fulminating symptoms with peritonitis, sepsis, intravascular coagulation, multi-organ failure and death.
There is an inverse relationship with gestational age with regard to the onset of the disease: the lower the gestational age, the later the onset of necrosing enterocolitis. Thus, in patients born at term, the disease manifests in the first days of life, while in preterm births the mean time of onset is from 2 to 4 weeks.
Radiology is essential in the diagnosis and follow-up of this condition, distension of the bowel loops is the most common radiological finding. Others, such as pneumatosis (pathognomonic of enterocolitis) or portal gas, can orientate us as to the severity of the disorder, since a pneumoperitoneum defines the surgical approach for these patients.
In 1978, Bell introduced a classification for NEC in 3 stages, according to gastrointestinal, systemic and radiological manifestations. Subsequently, Kliegman and Walsh modified this classification in 1986, and the latter is currently the most used to establish therapeutic and prognostic behaviours.
The treatment of these children is based on 2 strategies: medical and surgical, the latter being controversial, since the precise time for action has not been established, although this would ideally be described as when there is “intestinal necrosis without perforation”.
Approximately 70% of patients with necrosing enterocolitis can be managed conservatively. The remainder progress to more advanced stages of the disease and require surgical treatment.
Material and methodsA total of 124 patients diagnosed with necrosing enterocolitis were included in the study. These patients presented a mean follow-up of 2.5 months and total follow-up of 304.9 months.
We performed a retrospective and prospective descriptive study analysing clinical histories and gathering demographic variables, gestational data, perinatal background, clinical, analytical and radiological manifestations, perinatal therapeutic management and events in the follow-up of patients with necrosing enterocolitis diagnosed and treated in the Hospital Materno Infantil Teresa Herrera of La Coruña, Spain, between 2003 and 2015.
Diagnosis was based on clinical, radiological and histopathological criteria according to the modified Bell classification, and therefore we included patients in stage II or III of this classification.
We excluded the patients who did not meet the inclusion criteria for the study: those referred from other centres with no initial data of a diagnosis of necrosing enterocolitis, those with intraoperative findings of focal bowel perforation and those without signed informed consent.
The study was approved by the Ethics Committee of the Complejo Hospitalario Universitario of A Coruña as well as the Clinical Research Ethics Committee (CREC) of Galicia. This is a retrospective study where some of the patients died and others had the disorder 10 years previously. This is why obtaining informed consent was not viable for the majority of these patients. For this reason, we asked permission to review these patients’ clinical histories, taking the following into consideration:
- •
Law 41/2002 regulating the autonomy of patients and rights and obligations with regard to clinical information and documentation.
- •
Law 3/2005, amending Law 3/2001, regulating informed consent and patients’ clinical records.
- •
Decree 29/2009, of 5 February, regulating the use and access to electronic clinical records.
- •
Compliance with Law 15/1999, on personal data protection, and its implementing regulation.
We guarantee not to identify these patients via a codification process.
We shall ask the parents or legal guardians of the patients diagnosed during the course of 2015 and currently undergoing follow-up for their informed consent.
This sample size (n=124) enabled us to estimate parameters of interest with a 95% confidence interval (α=0.005 and 8.9% precision). The quantitative variables were expressed as mean and standard deviation (SD), and the qualitative variables were expressed in absolute values (n) and percentage, with 95% estimate of the confidence interval (CI). The Student's t-test or Mann–Whitney tests were used to compare the quantitative variables depending on the normality, for which the Kolmogorov–Smirnov test was used. The association between variables was analysed using Pearson's chi-squared test.
ResultsA total of 124 patients were included in the study.
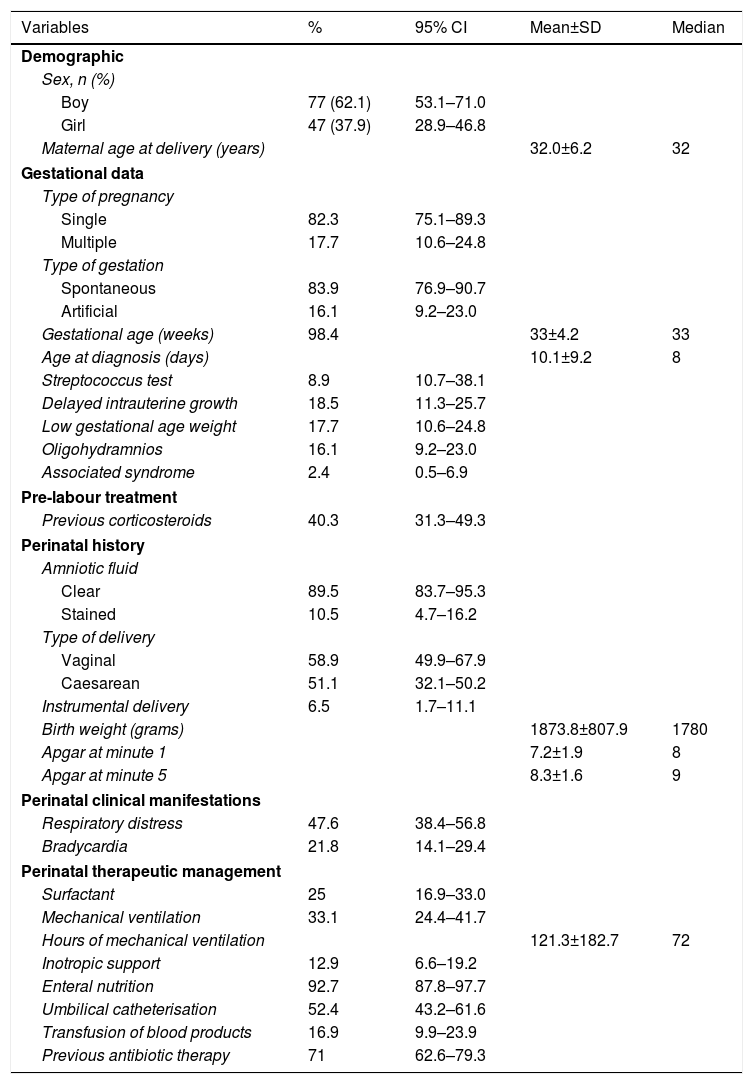
The general characteristics of the sample studied are shown in Table 1.
Description of the patients studies according to perinatal history and comorbidity.
| Variables | % | 95% CI | Mean±SD | Median |
|---|---|---|---|---|
| Demographic | ||||
| Sex, n (%) | ||||
| Boy | 77 (62.1) | 53.1–71.0 | ||
| Girl | 47 (37.9) | 28.9–46.8 | ||
| Maternal age at delivery (years) | 32.0±6.2 | 32 | ||
| Gestational data | ||||
| Type of pregnancy | ||||
| Single | 82.3 | 75.1–89.3 | ||
| Multiple | 17.7 | 10.6–24.8 | ||
| Type of gestation | ||||
| Spontaneous | 83.9 | 76.9–90.7 | ||
| Artificial | 16.1 | 9.2–23.0 | ||
| Gestational age (weeks) | 98.4 | 33±4.2 | 33 | |
| Age at diagnosis (days) | 10.1±9.2 | 8 | ||
| Streptococcus test | 8.9 | 10.7–38.1 | ||
| Delayed intrauterine growth | 18.5 | 11.3–25.7 | ||
| Low gestational age weight | 17.7 | 10.6–24.8 | ||
| Oligohydramnios | 16.1 | 9.2–23.0 | ||
| Associated syndrome | 2.4 | 0.5–6.9 | ||
| Pre-labour treatment | ||||
| Previous corticosteroids | 40.3 | 31.3–49.3 | ||
| Perinatal history | ||||
| Amniotic fluid | ||||
| Clear | 89.5 | 83.7–95.3 | ||
| Stained | 10.5 | 4.7–16.2 | ||
| Type of delivery | ||||
| Vaginal | 58.9 | 49.9–67.9 | ||
| Caesarean | 51.1 | 32.1–50.2 | ||
| Instrumental delivery | 6.5 | 1.7–11.1 | ||
| Birth weight (grams) | 1873.8±807.9 | 1780 | ||
| Apgar at minute 1 | 7.2±1.9 | 8 | ||
| Apgar at minute 5 | 8.3±1.6 | 9 | ||
| Perinatal clinical manifestations | ||||
| Respiratory distress | 47.6 | 38.4–56.8 | ||
| Bradycardia | 21.8 | 14.1–29.4 | ||
| Perinatal therapeutic management | ||||
| Surfactant | 25 | 16.9–33.0 | ||
| Mechanical ventilation | 33.1 | 24.4–41.7 | ||
| Hours of mechanical ventilation | 121.3±182.7 | 72 | ||
| Inotropic support | 12.9 | 6.6–19.2 | ||
| Enteral nutrition | 92.7 | 87.8–97.7 | ||
| Umbilical catheterisation | 52.4 | 43.2–61.6 | ||
| Transfusion of blood products | 16.9 | 9.9–23.9 | ||
| Previous antibiotic therapy | 71 | 62.6–79.3 | ||
The mean gestational age in our case series was 33 weeks.
In our study, a total of 45 neonates had a gestational age at birth of over 34 weeks, which comprises 36% of the total number of cases studied, which is high compared to other series that report a rate of around 10–15% of patients with necrosing enterocolitis at term.15 This mean age is due in large part to the cases recorded at the start of our series between 2003 and 2009 – the period when the great revolution in the care of extremely preterm infants started.
There is a slight predominance of males (62.1% vs 37.9%), in line with the literature.
Sixteen point one percent had a history of artificial gestation, and the mean maternal age was 32 years.
Throughout the period studied, the maternal age increased progressively and significantly, from the period 2003–2006, with a mean age of 30.69±6.3 years, compared to 34.09±5.5 years in the period 2011–2015.
There was a progressive increase in artificial gestation, from 16.1% in the period 2003–2006 to 25.7% in the period 2011–2015.
In turn, 17.7% of patients had multiple gestations. This prevalence of multiple gestations increased over the study period, but not significantly, from 16.1% in the period 2003–2006 to 18.2% in the period 2007–2010, and to 20% in the period 2011–2015.
With regard to umbilical catheterisation, which is described in the literature as a risk factor,16 it is worth mentioning that 52% of our series had an umbilical catheter.
Forty point three percent of our patients underwent prenatal corticosteroid treatment, which is a protective factor according to various published studies.
The mean age of onset of the disease in our patients was 10 days, and was later the more preterm the infant, in line with previous studies.
In our series, 93% of patients had received enteral nutrition before the onset of the disease, in line with the published literature.
With regard to factors suggestive of intestinal ischaemia, 10.5% of our patients presented persistent ductus arteriosus at birth, and up to 18.5% associated heart anomalies. These patients were of greater gestational age and higher birth weight.
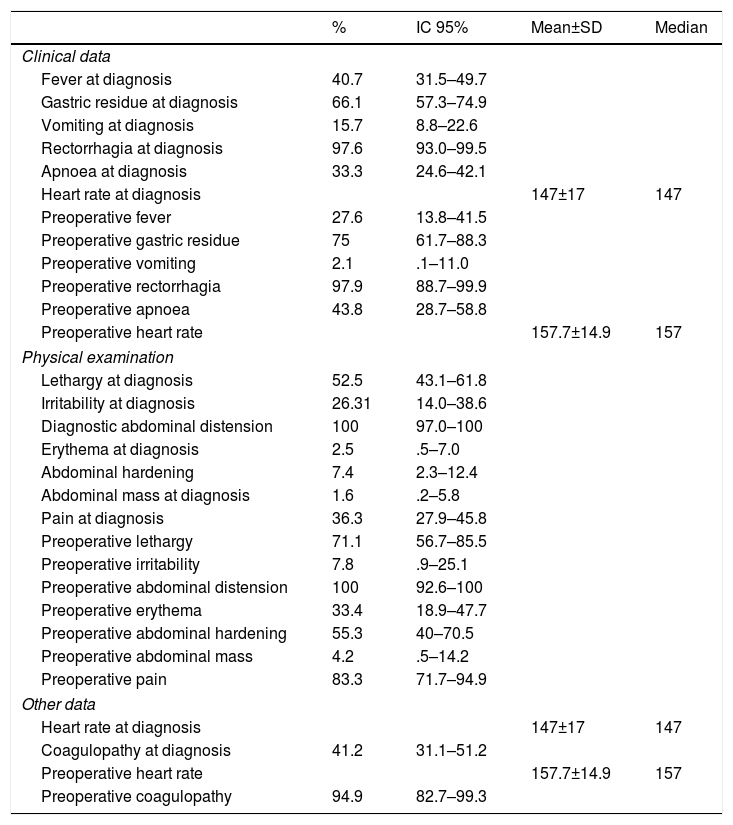
The most frequent clinical manifestation (Table 2) was rectorrhagia (97.6%), followed by gastric residue (66.1%).
Description of the patients studied according to their clinical manifestations.
| % | IC 95% | Mean±SD | Median | |
|---|---|---|---|---|
| Clinical data | ||||
| Fever at diagnosis | 40.7 | 31.5–49.7 | ||
| Gastric residue at diagnosis | 66.1 | 57.3–74.9 | ||
| Vomiting at diagnosis | 15.7 | 8.8–22.6 | ||
| Rectorrhagia at diagnosis | 97.6 | 93.0–99.5 | ||
| Apnoea at diagnosis | 33.3 | 24.6–42.1 | ||
| Heart rate at diagnosis | 147±17 | 147 | ||
| Preoperative fever | 27.6 | 13.8–41.5 | ||
| Preoperative gastric residue | 75 | 61.7–88.3 | ||
| Preoperative vomiting | 2.1 | .1–11.0 | ||
| Preoperative rectorrhagia | 97.9 | 88.7–99.9 | ||
| Preoperative apnoea | 43.8 | 28.7–58.8 | ||
| Preoperative heart rate | 157.7±14.9 | 157 | ||
| Physical examination | ||||
| Lethargy at diagnosis | 52.5 | 43.1–61.8 | ||
| Irritability at diagnosis | 26.31 | 14.0–38.6 | ||
| Diagnostic abdominal distension | 100 | 97.0–100 | ||
| Erythema at diagnosis | 2.5 | .5–7.0 | ||
| Abdominal hardening | 7.4 | 2.3–12.4 | ||
| Abdominal mass at diagnosis | 1.6 | .2–5.8 | ||
| Pain at diagnosis | 36.3 | 27.9–45.8 | ||
| Preoperative lethargy | 71.1 | 56.7–85.5 | ||
| Preoperative irritability | 7.8 | .9–25.1 | ||
| Preoperative abdominal distension | 100 | 92.6–100 | ||
| Preoperative erythema | 33.4 | 18.9–47.7 | ||
| Preoperative abdominal hardening | 55.3 | 40–70.5 | ||
| Preoperative abdominal mass | 4.2 | .5–14.2 | ||
| Preoperative pain | 83.3 | 71.7–94.9 | ||
| Other data | ||||
| Heart rate at diagnosis | 147±17 | 147 | ||
| Coagulopathy at diagnosis | 41.2 | 31.1–51.2 | ||
| Preoperative heart rate | 157.7±14.9 | 157 | ||
| Preoperative coagulopathy | 94.9 | 82.7–99.3 | ||
All the patients had abdominal distension, often followed by lethargy, on physical examination (52.5%).
It is also worth mentioning that 47.6% of the children presented perinatal respiratory distress, described in the recent literature as one of the most relevant risk factors for the development of this disease.17
In our case series, Escherichia coli, Klebsiella and Staphylococcus were the germs most frequently isolated in our patients’ blood cultures, in line with the published references.18 It is considered that, secondary to bacterial translocation of the intestinal wall, 20–30% of these patients might have associated bacteraemia, as shown in our series (15.3%).
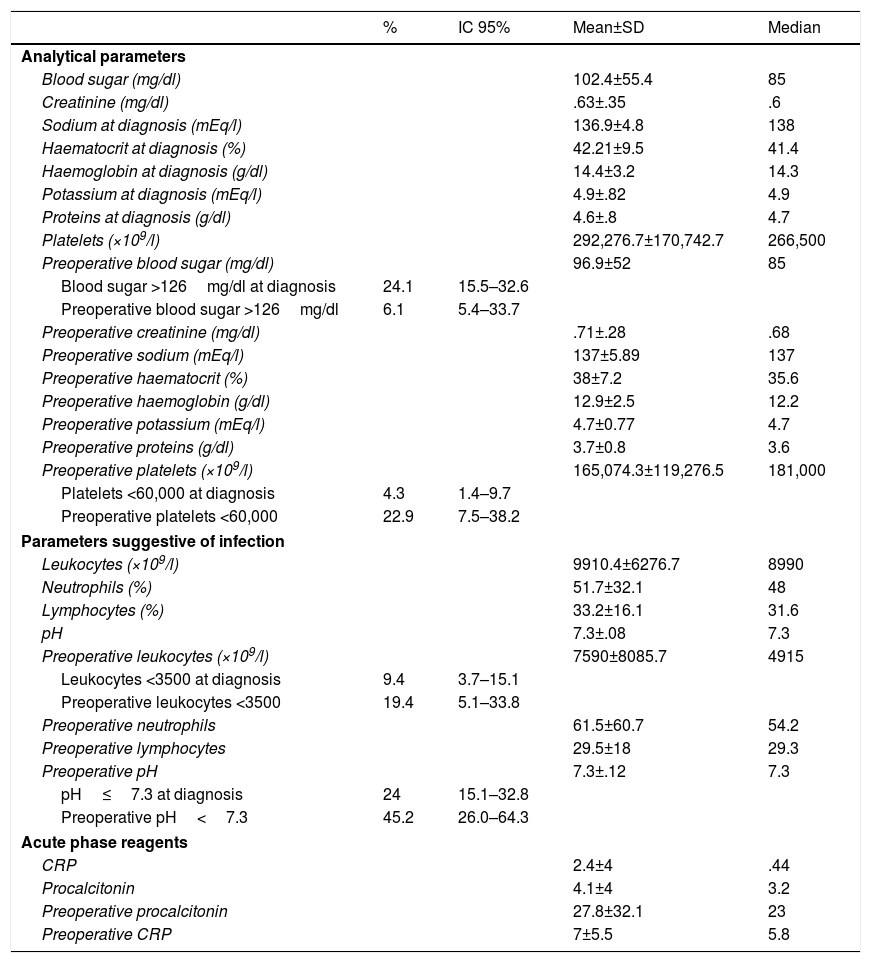
The remaining analytical parameters are shown in Table 3, where it can be observed that 24% of the patients had blood sugar levels above 126mg/dl on diagnosis, 4.3% had severe thrombocytopenia (fewer than 60,000 platelets), 9.4% had severe leukopenia (less than 3500) and 24%, acidosis (pH≤7.30). Forty-one percent had coagulopathy.
Description of the patients studied according to analytical parameters.
| % | IC 95% | Mean±SD | Median | |
|---|---|---|---|---|
| Analytical parameters | ||||
| Blood sugar (mg/dl) | 102.4±55.4 | 85 | ||
| Creatinine (mg/dl) | .63±.35 | .6 | ||
| Sodium at diagnosis (mEq/l) | 136.9±4.8 | 138 | ||
| Haematocrit at diagnosis (%) | 42.21±9.5 | 41.4 | ||
| Haemoglobin at diagnosis (g/dl) | 14.4±3.2 | 14.3 | ||
| Potassium at diagnosis (mEq/l) | 4.9±.82 | 4.9 | ||
| Proteins at diagnosis (g/dl) | 4.6±.8 | 4.7 | ||
| Platelets (×109/l) | 292,276.7±170,742.7 | 266,500 | ||
| Preoperative blood sugar (mg/dl) | 96.9±52 | 85 | ||
| Blood sugar >126mg/dl at diagnosis | 24.1 | 15.5–32.6 | ||
| Preoperative blood sugar >126mg/dl | 6.1 | 5.4–33.7 | ||
| Preoperative creatinine (mg/dl) | .71±.28 | .68 | ||
| Preoperative sodium (mEq/l) | 137±5.89 | 137 | ||
| Preoperative haematocrit (%) | 38±7.2 | 35.6 | ||
| Preoperative haemoglobin (g/dl) | 12.9±2.5 | 12.2 | ||
| Preoperative potassium (mEq/l) | 4.7±0.77 | 4.7 | ||
| Preoperative proteins (g/dl) | 3.7±0.8 | 3.6 | ||
| Preoperative platelets (×109/l) | 165,074.3±119,276.5 | 181,000 | ||
| Platelets <60,000 at diagnosis | 4.3 | 1.4–9.7 | ||
| Preoperative platelets <60,000 | 22.9 | 7.5–38.2 | ||
| Parameters suggestive of infection | ||||
| Leukocytes (×109/l) | 9910.4±6276.7 | 8990 | ||
| Neutrophils (%) | 51.7±32.1 | 48 | ||
| Lymphocytes (%) | 33.2±16.1 | 31.6 | ||
| pH | 7.3±.08 | 7.3 | ||
| Preoperative leukocytes (×109/l) | 7590±8085.7 | 4915 | ||
| Leukocytes <3500 at diagnosis | 9.4 | 3.7–15.1 | ||
| Preoperative leukocytes <3500 | 19.4 | 5.1–33.8 | ||
| Preoperative neutrophils | 61.5±60.7 | 54.2 | ||
| Preoperative lymphocytes | 29.5±18 | 29.3 | ||
| Preoperative pH | 7.3±.12 | 7.3 | ||
| pH≤7.3 at diagnosis | 24 | 15.1–32.8 | ||
| Preoperative pH<7.3 | 45.2 | 26.0–64.3 | ||
| Acute phase reagents | ||||
| CRP | 2.4±4 | .44 | ||
| Procalcitonin | 4.1±4 | 3.2 | ||
| Preoperative procalcitonin | 27.8±32.1 | 23 | ||
| Preoperative CRP | 7±5.5 | 5.8 | ||
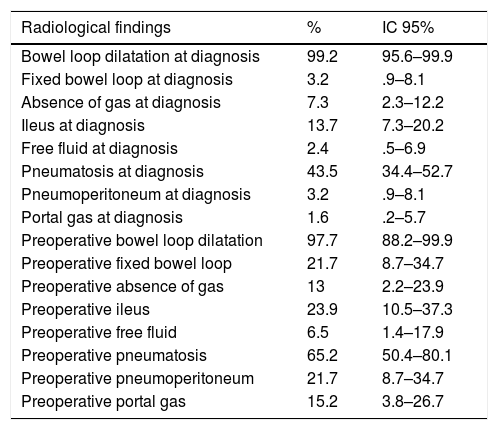
The most frequent radiological findings were bowel loop dilatation (99.2%) and intestinal pneumatosis (43.5%) (Table 4).
Description of the patients studied according to radiological findings.
| Radiological findings | % | IC 95% |
|---|---|---|
| Bowel loop dilatation at diagnosis | 99.2 | 95.6–99.9 |
| Fixed bowel loop at diagnosis | 3.2 | .9–8.1 |
| Absence of gas at diagnosis | 7.3 | 2.3–12.2 |
| Ileus at diagnosis | 13.7 | 7.3–20.2 |
| Free fluid at diagnosis | 2.4 | .5–6.9 |
| Pneumatosis at diagnosis | 43.5 | 34.4–52.7 |
| Pneumoperitoneum at diagnosis | 3.2 | .9–8.1 |
| Portal gas at diagnosis | 1.6 | .2–5.7 |
| Preoperative bowel loop dilatation | 97.7 | 88.2–99.9 |
| Preoperative fixed bowel loop | 21.7 | 8.7–34.7 |
| Preoperative absence of gas | 13 | 2.2–23.9 |
| Preoperative ileus | 23.9 | 10.5–37.3 |
| Preoperative free fluid | 6.5 | 1.4–17.9 |
| Preoperative pneumatosis | 65.2 | 50.4–80.1 |
| Preoperative pneumoperitoneum | 21.7 | 8.7–34.7 |
| Preoperative portal gas | 15.2 | 3.8–26.7 |
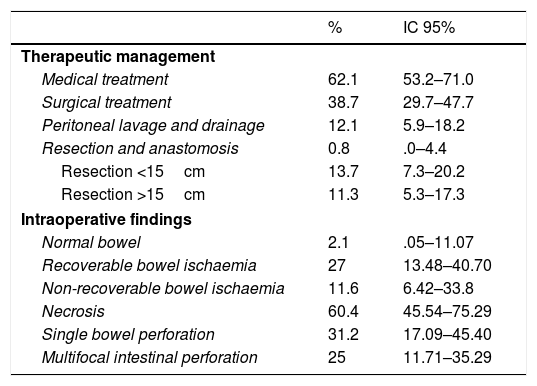
Thirty-eight point seven percent of the patients required surgical treatment, 11.3% required resections greater than 15cm. Single bowel perforation was found during surgery with signs of intestinal ischaemia in 31.2% of cases. It is relevant to highlight that patients with focal intestinal perforation were removed from the study (Table 5).
Description of the patients studied according to therapeutic management and intraoperative findings.
| % | IC 95% | |
|---|---|---|
| Therapeutic management | ||
| Medical treatment | 62.1 | 53.2–71.0 |
| Surgical treatment | 38.7 | 29.7–47.7 |
| Peritoneal lavage and drainage | 12.1 | 5.9–18.2 |
| Resection and anastomosis | 0.8 | .0–4.4 |
| Resection <15cm | 13.7 | 7.3–20.2 |
| Resection >15cm | 11.3 | 5.3–17.3 |
| Intraoperative findings | ||
| Normal bowel | 2.1 | .05–11.07 |
| Recoverable bowel ischaemia | 27 | 13.48–40.70 |
| Non-recoverable bowel ischaemia | 11.6 | 6.42–33.8 |
| Necrosis | 60.4 | 45.54–75.29 |
| Single bowel perforation | 31.2 | 17.09–45.40 |
| Multifocal intestinal perforation | 25 | 11.71–35.29 |
This cohort of patients (n=124) generated a total 305 months of follow-up, with a mean of 0.82 months and a range varying between 0 and 36 months.
During this time, the events that presented resulted in a fatality rate of 11.3%. The mortality rate was higher in the males in our series (64%), in line with other published series.19 Among the events that presented during follow-up, it is important to highlight bowel perforation in 8.9% of the cases (Table 6).
DiscussionPreterm infants are undoubtedly vulnerable hosts, and therefore prematurity is the most significant isolated risk factor for developing this disorder. Only around 7–15% of cases of necrosing enterocolitis occur in infants born at term,15 and these usually have another associated or underlying risk factor in the context of a epidemic, neonatal outbreak.
Although in the presurfactant era, the typical patient with this disease was an infant born at term, of a healthy weight and with no associated risk factors, the changes in maternal demographic characteristics and in the progress of extremely preterm infants, with the advances in pre- and perinatal care, have resulted in a new type of patient with radically different demographic and gestational characteristics.20
In our case study, we were able to confirm a major increase in maternal age, artificial gestations and multiple pregnancies in patients affected by necrosing enterocolitis. The change in the epidemiological characteristics of these patients is clear given the evolution and development of the assisted reproduction techniques in recent years.
However, it is worth noting in our case series that the gestational age of our patients remained stable, from the cases recorded in the first years of the series (2003–2009).
Factors such as a low Apgar score,21 umbilical catheterisation, episodes of apnoea and bradycardia, respiratory distress, anaemia, hypothermia, ischaemic events,22–24 hypotension, the use of vasoactive agents,25 the use of surfactant, among others, have been described as contributing factors for developing necrosing enterocolitis, although more reviews are required than those presented in this article.
As our case series demonstrates, necrosing enterocolitis is a disorder that, despite the advances in pre-and perinatal care, remains stable in terms of incidence26 and mortality, and is the most common cause of death in preterm infants that survive the first week of life. Moreover, those that do survive the disease have high morbidity, with the consequent impact on family and social health.
Considering the impact on our society of necrosing enterocolitis, it is important to undertake studies and develop strategies that bring about a reduction in its incidence, and its morbidity and mortality.
To conclude, it is worth stressing that, despite the advances, this disease remains to be fully addressed and its aetiopathogenesis is still unclear. High diagnostic suspicion, together with multidisciplinary and dynamic management, is essential for the correct management of this disorder.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflict of interestThe authors have no conflict of interest to declare.
Please cite this article as: García-González M, Pita-Fernández S, Caramés-Bouzán J. Análisis de las características poblacionales de neonatos afectos de enterocolitis necrosante en un centro terciario en los últimos 12 años. Cir Cir. 2017;85:411–418.