In pediatrics, complications of the peripherally inserted central venous catheters (PICC) are reported in 30-40% of the cases, being the age of the patient and the location of PICC the most frequent risk factors. This study aimed to describe the characteristics of PICCs and to identify factors related to their withdrawal due to complications.
MethodsA prospective cohort study was conducted. Patients from 2 months to 18 years of age were included and followed-up until PICC was withdrawn. Age, sex, medications administered, PICC location, caliber, the number of lumens, days PICC was on the patient, and the reason for withdrawal was recorded. Univariate and multivariate analyses were performed.
ResultsTwo hundred and twenty-five patients were included. In 33.02%, PICC was withdrawn due to complications. In the bivariate analysis, age and location were factors associated with complications. A Cox proportional hazards model was performed. Only the caliber thickness (hazard ratio (HR): 0.5, confidence interval (CI) 95%: 0.35-0.99) was associated with any complication, and age (HR: 1.02, CI95%: 1-1.04) to the withdrawal of PICC due to suspected phlebitis.
ConclusionsPICC withdrawal due to complications was performed in 33% of the patients. Risk factors related to complications were PICC caliber thickness and age.
En pediatría se reportan complicaciones del catéter central de inserción periférica (PICC) del 30 al 40% de los casos, siendo los factores de riesgo más frecuentes la edad del paciente y la localización del PICC. El objetivo de este trabajo fue describir las características de los PICC e identificar los factores relacionados con su retiro debido a complicaciones.
MétodosEstudio de cohorte prospectiva. Se incluyeron pacientes de 2 meses a 18 años de edad. Se les dio seguimiento hasta el retiro del PICC. Se registró la edad, sexo, medicamentos administrados, localización del PICC, calibre, número de lúmenes, días que estuvo colocado y el motivo del retiro. Se realizó análisis univariado y multivariado.
ResultadosSe incluyeron 225 pacientes. En 33.02%, el PICC fue retirado por alguna complicación. En el análisis bivariado, la edad y la localización fueron los factores asociados con las complicaciones. Se realizó un modelo de riesgos proporcionales de Cox y se identificó que solamente el grosor del calibre se asoció con cualquier complicación (razón de tasas (RT): 0.5, intervalo de confianza (IC)95%: 0.35-0.99), y la edad (RT: 1.02, IC95%: 1-1.04) solamente con el retiro del PICC por sospecha de flebitis.
ConclusionesEl 33% de los pacientes presentaron retiro del CIPP por complicaciones; solamente el calibre del CIPP y la edad de los pacientes estuvieron relacionados con este evento.
The peripherally inserted central catheter (PICC) is the most often invasive procedure applied in hospitalized pediatric patients since up to 90% of patients require parenteral administration of drugs.1 PICC provides access to a central vein (superior vena cava) of large caliber from a peripheral vein.2 The catheters can be made from medium-term, grade III polyurethane and long-lasting silicone. Catheters should be placed by a highly trained nurse under sterile conditions.3
These devices allow the administration of solutions with high pH and osmolarity. However, a pH greater than 7.45 or less than 7.35 and osmolarity ≥ 350 mOsml/l present a high risk of causing phlebitis; therefore, they should not be administered by this route.4
The conditions that must be evaluated before the placement of the device are the characteristics of the patient (quality of the vessels), the treatment to be administered (duration, prescribed infusion therapy) and the device type.5,6
As an advantage, PICC can be placed at the patient's bedside. Furthermore, the potential complication of pneumothorax is eliminated, infections associated with the catheter are significantly reduced, and pain and discomfort associated with frequent punctures for peripheral sites are reduced.7
Despite having multiple advantages, complications of these devices, which can lead to high morbidity in some cases are also described. In general, the PICCs complications may occur in up to 40% of cases, being infiltration (14.4%) and occlusion (13.4%) the most frequent complications. The complication with greater morbidity is thrombosis, which ranges from 0-7% of the cases.8,9
Multiple studies have been conducted to identify factors related to complications. Some risk factors that have been described for the presence of infiltration are a stay longer than five days and predisposing conditions of the patient, such as difficulties with previous venipuncture, history of infiltration, the use of an infusion pump to apply medications,10 young age, and insertion in the curve of the arm or ankle. Occlusion was associated with more days of use and with the insertion in the ankle or foot.11 In older children, an increased risk of infection was identified.12
Given the variability of the risk factors related to the complications of the PICCs, which differ according to each population, the objective of this study was to describe the characteristics of the PICCs installed in pediatric patients attending the Hospital Infantil de México Federico Gómez (HIMFG) and identify factors related to complications.
2MethodsA prospective cohort study with pediatric patients from two months to 18 years of age who required the application of a PICC was conducted. When a PICC was required during their hospitalization, patients were included in the cohort. Patients in whom it was feasible to channel peripheral venous access and the expected duration of treatment was < 30 days were included in the study. Patients with venous access exhausted by multiple anterior venipunctures were excluded, as well as those with tortuous or small caliber veins, with thrombocytopenia at the time of performing the procedure, when there was not an appropriate size catheter, and neonatal patients—since resident doctors of the area are responsible for placing catheters in these cases. Patients who were removed from the PICC by a decision of the parents (voluntary discharge) were also excluded.
Under the supervision of a pediatrician, highly trained nurses of the HIMFG Catheter Clinic placed the PICC with the percutaneous Seldinger technique, and low topical anesthetic effect (lidocaine-prilocaine cream applied half hour before the procedure) or moderate sedation (midazolam or ketamine) to minimize the pain caused by the puncture.
Data such as age, sex, medical service to which the patient was admitted, and drugs that were administered (antibiotics, chemotherapy, intravenous solutions, analgesics and parenteral nutrition) were obtained from the medical records.
The location of the PICC, the caliber (Fr), the number of lumens, the duration, and the reason for which it was removed were identified. The latter was classified between programmed withdrawal due to termination of treatment or withdrawal due to complications. Complications were classified as obstruction (resistance to passage of intravenous solution or medication); suspected infection (pain in the area of the PICC, accompanied by erythema in the path of the vein where it was placed); clinical data of phlebitis (pain when passing the solution or medication) or suspicion of infiltration (the presence of edema around the PICC or verification of rupture of the PICC).
The protocol was submitted for evaluation and approval to the Local Committee of Research and Ethics in Health of the HIMFG.
2.1Statistical analysisData are presented as absolute frequencies and percentages. The quantitative variables are presented as median and minimum and maximum values because they did not have a normal distribution. The comparison between patients with the withdrawal of PICC secondary to complication and those with no complications was performed with χ2 or Fisher's exact test for qualitative variables. Mann–Whitney U test was used for the comparison of quantitative variables.
To identify variables related to the withdrawal of the PICC due to complications, a Cox proportional hazards model was performed. Values of p <0.05 were considered statistically significant. Analyses were performed with the statistical package SPSS version 17.0 (IBM).
3ResultsFrom July 2014 to October 2016, patients between two months and 17 years of age hospitalized in the services of Oncology, Infectology, Cardiology, Internal Medicine, Neurology and Nephrology of the HIMFG that required the placement of a PICC were identified. A total of 225 patients were found, of which 10 were excluded because the reason for the withdrawal of the PICC was not registered during the follow-up.
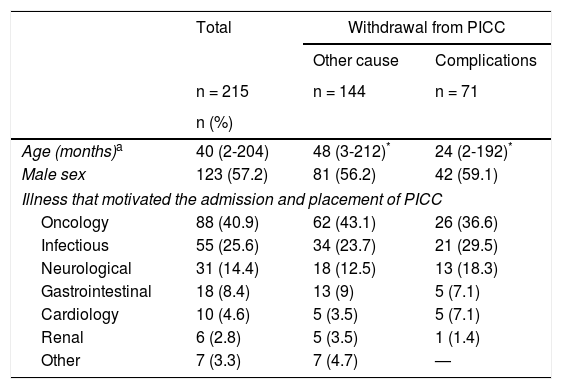
In the 215 patients included, a greater proportion of males (57.21%) was observed, with a median age of three years (minimum of two months and maximum of 17 years). The condition that most frequently required an installation of a PICC was oncological (40.9%), followed by infection (25.6%) and neurological condition (14.4%) (Table 1).
Characteristics of patients who required peripherally inserted central venous catheter.
| Total | Withdrawal from PICC | ||
|---|---|---|---|
| Other cause | Complications | ||
| n = 215 | n = 144 | n = 71 | |
| n (%) | |||
| Age (months)a | 40 (2-204) | 48 (3-212)* | 24 (2-192)* |
| Male sex | 123 (57.2) | 81 (56.2) | 42 (59.1) |
| Illness that motivated the admission and placement of PICC | |||
| Oncology | 88 (40.9) | 62 (43.1) | 26 (36.6) |
| Infectious | 55 (25.6) | 34 (23.7) | 21 (29.5) |
| Neurological | 31 (14.4) | 18 (12.5) | 13 (18.3) |
| Gastrointestinal | 18 (8.4) | 13 (9) | 5 (7.1) |
| Cardiology | 10 (4.6) | 5 (3.5) | 5 (7.1) |
| Renal | 6 (2.8) | 5 (3.5) | 1 (1.4) |
| Other | 7 (3.3) | 7 (4.7) | — |
PICC, peripherally inserted central venous catheter.
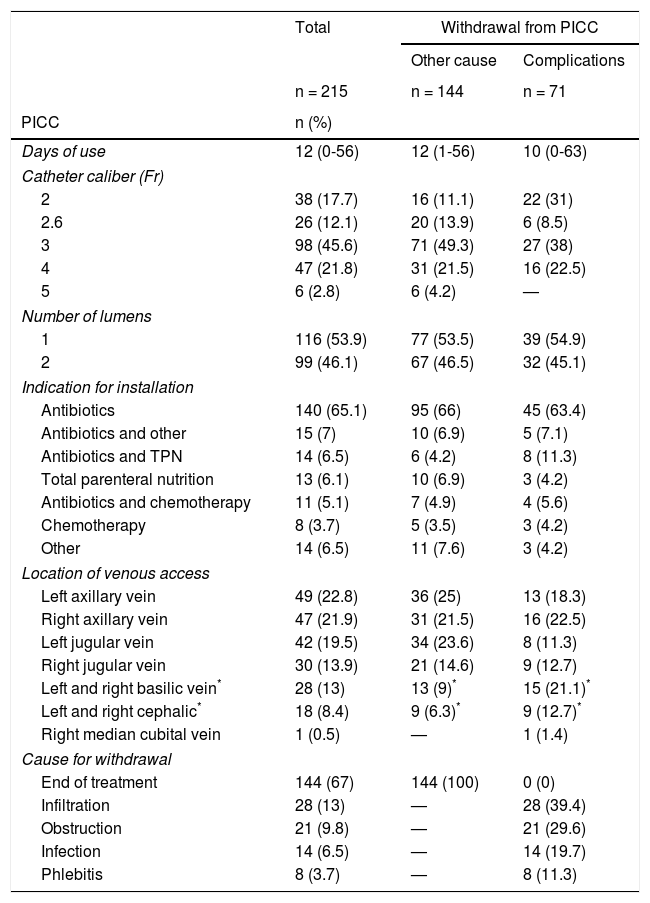
The primary indication for the catheter was the need for antimicrobial treatment (65.1%). The median duration of the catheter was of 12 days (minimum 0 days and maximum 56 days). The catheter caliber that was most frequently placed was 4-Fr (n= 47, 21.9%) and 53.9% with a single lumen. The most frequent location of venous access was in the axillary vein (44.7%), followed by the jugular vein (33.5%). In 33% of cases (n = 71), the PICC was withdrawn due to complications. The most frequent complications were dysfunction, in 28 cases (13%), and occlusion, in 21 cases (9.78%) (Table 2).
Characteristics of the placed peripherally inserted central venous catheter.
| Total | Withdrawal from PICC | ||
|---|---|---|---|
| Other cause | Complications | ||
| n = 215 | n = 144 | n = 71 | |
| PICC | n (%) | ||
| Days of use | 12 (0-56) | 12 (1-56) | 10 (0-63) |
| Catheter caliber (Fr) | |||
| 2 | 38 (17.7) | 16 (11.1) | 22 (31) |
| 2.6 | 26 (12.1) | 20 (13.9) | 6 (8.5) |
| 3 | 98 (45.6) | 71 (49.3) | 27 (38) |
| 4 | 47 (21.8) | 31 (21.5) | 16 (22.5) |
| 5 | 6 (2.8) | 6 (4.2) | — |
| Number of lumens | |||
| 1 | 116 (53.9) | 77 (53.5) | 39 (54.9) |
| 2 | 99 (46.1) | 67 (46.5) | 32 (45.1) |
| Indication for installation | |||
| Antibiotics | 140 (65.1) | 95 (66) | 45 (63.4) |
| Antibiotics and other | 15 (7) | 10 (6.9) | 5 (7.1) |
| Antibiotics and TPN | 14 (6.5) | 6 (4.2) | 8 (11.3) |
| Total parenteral nutrition | 13 (6.1) | 10 (6.9) | 3 (4.2) |
| Antibiotics and chemotherapy | 11 (5.1) | 7 (4.9) | 4 (5.6) |
| Chemotherapy | 8 (3.7) | 5 (3.5) | 3 (4.2) |
| Other | 14 (6.5) | 11 (7.6) | 3 (4.2) |
| Location of venous access | |||
| Left axillary vein | 49 (22.8) | 36 (25) | 13 (18.3) |
| Right axillary vein | 47 (21.9) | 31 (21.5) | 16 (22.5) |
| Left jugular vein | 42 (19.5) | 34 (23.6) | 8 (11.3) |
| Right jugular vein | 30 (13.9) | 21 (14.6) | 9 (12.7) |
| Left and right basilic vein* | 28 (13) | 13 (9)* | 15 (21.1)* |
| Left and right cephalic* | 18 (8.4) | 9 (6.3)* | 9 (12.7)* |
| Right median cubital vein | 1 (0.5) | — | 1 (1.4) |
| Cause for withdrawal | |||
| End of treatment | 144 (67) | 144 (100) | 0 (0) |
| Infiltration | 28 (13) | — | 28 (39.4) |
| Obstruction | 21 (9.8) | — | 21 (29.6) |
| Infection | 14 (6.5) | — | 14 (19.7) |
| Phlebitis | 8 (3.7) | — | 8 (11.3) |
PICC, peripherally inserted central venous catheter; TPN, total parenteral nutrition.
+Median (min-max).
The most severe complication was thrombophlebitis, which occurred in a 12-year-old male with an oncological pathology (0.4%). The catheter, 4-Fr caliber of two lumens, was located in the right cephalic vein. The patient was being treated with antibiotics and analgesics, and the catheter duration was of seven days.
When comparing the possible factors (age, condition that caused the placement of the PICC, catheter caliber, number of lumens, indication for its installation and access location), it was identified that young children (median age: 48-month-old (no complications) vs 24-month-old (complications), p = 0.03) and basilic and cephalic catheter placement (p = 0.024) were factors associated with withdrawal of the PICC due to any complication (Tables 1 and 2).
Subsequently, the same factors related to each of the complications were analyzed (obstruction, suspicion of infection, clinical data of phlebitis, and suspicion of infiltration). It was detected that the eight patients from whom the PICC was withdrawn due to clinical data of phlebitis presented a higher tendency of phlebitis compared to those who were not withdrawn from the PICC due to a complication, without a statistically significant difference (median age: 126-month-old vs 36-month-old, p = 0.09). No other related factor was identified for the rest of the complications.
When a Cox proportional-hazards model was performed to identify the factors that were associated with the withdrawal of the PICC due to complications, only the thickness of the caliber showed a significant association (hazard ratio (HR): 0.5; 95% confidence interval (CI): 0.35-0.99). A Cox proportional hazards model was performed for each reported complication (obstruction, suspected infection, clinical data of phlebitis, and suspected infiltration) and it was identified that age was an associated factor for the presence of clinical data of phlebitis (HR: 1.02; 95% CI: 1-1.04) (Table 3).
Cox proportional hazards model of the factors associated with the withdrawal of the PICC by complication*
| Complication | Associated factor | HR | 95% CI |
|---|---|---|---|
| Any complication | Caliber | 0.5 | 0.35-0.99 |
| Clinical data of phlebitis | Age (months) | 1.02 | 1-1.04 |
In this study, it was shown that the caliber of the PICC was related to its withdrawal by any complication and that the age of the patient was a factor associated with the withdrawal of the PICC by clinical data of phlebitis.
HIMFG is a tertiary hospital where a large number of patients with hematologic and neurological diseases can present infectious complications. In particular, this group of patients undergoes multiple medical treatments, such as chemotherapy, antimicrobials, analgesia, total parenteral nutrition and, in many cases, the combination of these. Consequently, they represent a population with a higher risk of presenting complications of the PICC.13 Despite this, the frequency of complications was similar to that reported on other studies in the pediatric population. For example, Malyon et al. reported 25% of complications in 458 patients from 0 to 15 years old, being the infiltration the most frequent complication (14.3%). The risk factor that was identified was the location of the PICC in the antecubital fossa (OR: 1.6; 95% CI: 1.1-2.3).14 In Lima, Peru, Jacinto et al. conducted a study in 338 patients aged 13 days to 16 years. In this study, they reported 26.9% of complications, with an infiltration frequency of 16%. Also, they identified infiltration history as a risk factor (OR: 31.28; 95% CI: 8.16-72.87).10 Levy et al. reported 36.5% of complications in 221 patients from 7 days to 21 years of age, being infection and mechanical problems the most common in 13.6%, respectively, with no risk factors reported.12 Unbeck et al. studied 2032 patients aged 0 to 18 years, where 35.4% had complications with the PICC. The authors identified that neonates showed a greater infiltration risk compared to the rest of the patients.11
A risk factor identified in these studies was the young age. These authors included the neonatal population, which was not included in the present study. This could explain why the same association was not detected in our patients.
Although all the studies aforementioned were carried out in the pediatric population, the criteria used to report complications by the PICC and the risk factors were very heterogeneous, which explains the varied results.
The greatest complication identified in the analyzed patients was thrombosis (0.4%), which concurs with that reported by Jumani et al., who studied 2574 patients with PICC and observed 20.8% of complications and 0.5% of thrombosis.15 In a meta-analysis of 11 studies, it was concluded that the risk factors for the development of thrombosis were the presence of a malignant disease and the critical condition of the patient.16
One of the limitations of the present study was the failure to include patients younger than two months, in whom more complications of the PICC such as infiltration or obstruction have been described. The complications identified in this study are similar to those described worldwide, which indicate the commitment and proper work that is performed in the Catheter Clinic at the HIMFG. Therefore, it is suggested to continue with this line of research and conduct future studies where a greater technology could be included for the placement of the PICC, such as ultrasound, which has been shown to substantially improve this procedure.17,18
In conclusion, it was necessary to withdraw the PICC due to complications in up to 33% of the patients. This percentage is similar to that reported on a global scale. The withdrawal of the PICC due to any complication was related only to the caliber of the PICC and, in particular, the withdrawal of the PICC due to the presence of phlebitis was associated to a greater age of the patient.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflict of interestsThe authors declare no conflicts of interest of any nature.
Please cite this article as: Flores MM, Pueblas BKS, Ojeda SA, Zurita-Cruz N. Factores de riesgo asociados con complicaciones que obligaron al retiro catéteres venosos centrales de inserción periférica en un hospital pediátrico de tercer nivel. Bol Med Hosp Infant Mex. 2017;74:289–294.