Associated liver partition and portal vein ligation for staged hepatectomy (ALPPS) has emerged as an alternative for patients with bilobar colorectal liver metastasis and a small future liver remnant (FLR). In cases of extensive disease, ALPPS can be performed, leaving only one segment of the liver as FLR. We describe a case of monosegmental ALPPS using segment 4 as FLR. In conclusion, ALPPS should be reserved for a selected group of patients. Monosegmental ALPPS is feasible, but should be performed by hepatobiliary surgeons in specialized centers.
Associated liver partition and portal vein ligation for staged hepatectomy (ALPPS) approach has recently emerged as an alternative treatment for patients with bilo-bar colorectal liver metastasis (CRLM) and a small future liver remnant (FLR). ALPPS promotes accelerated growth of the FLR in a short period of time (around seven days), and prevents post-hepatectomy liver failure.1,2 Initially described for patients using the left lateral segments as FLR,1 ALPPS has been adapted to the right posterior segments, two separate segments, or even one segment as FLR.3-6 Herein, we describe a case of segment 4-1 ALPPS on a large bilobar CRLM case associated with liver-first approach.
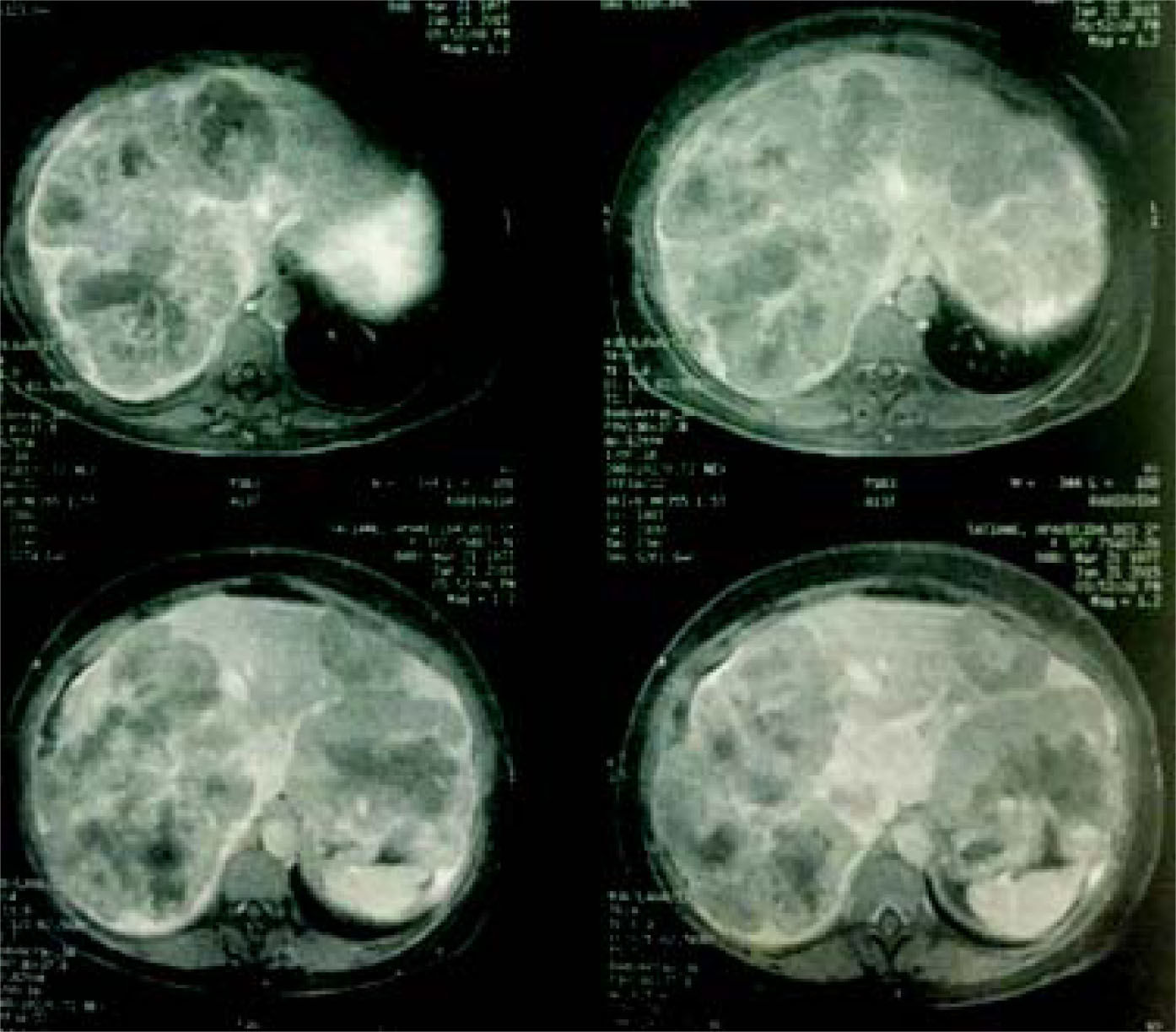
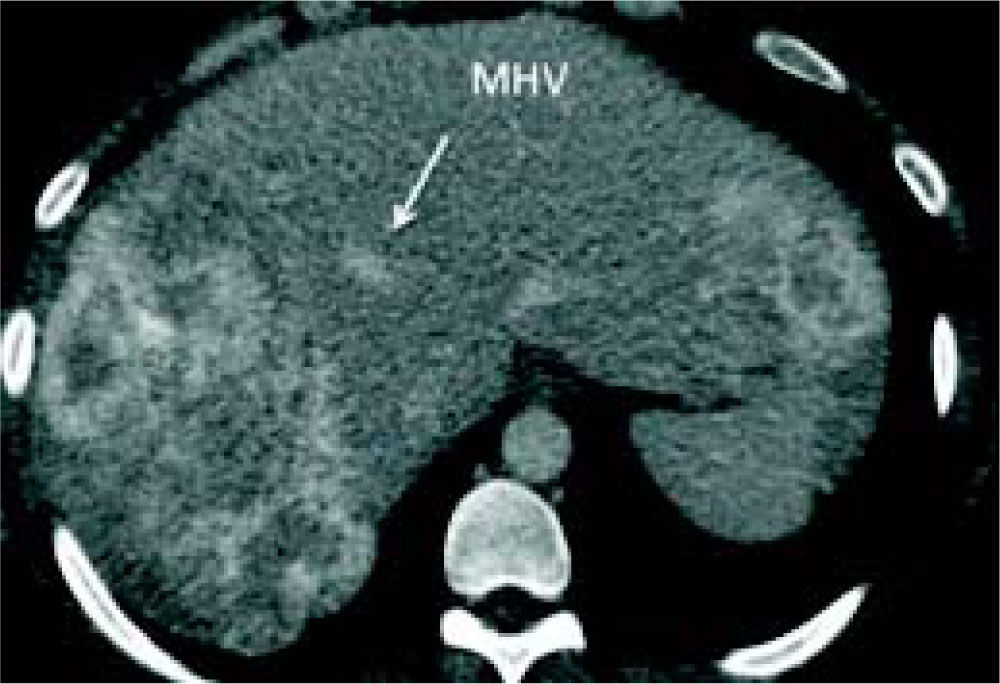
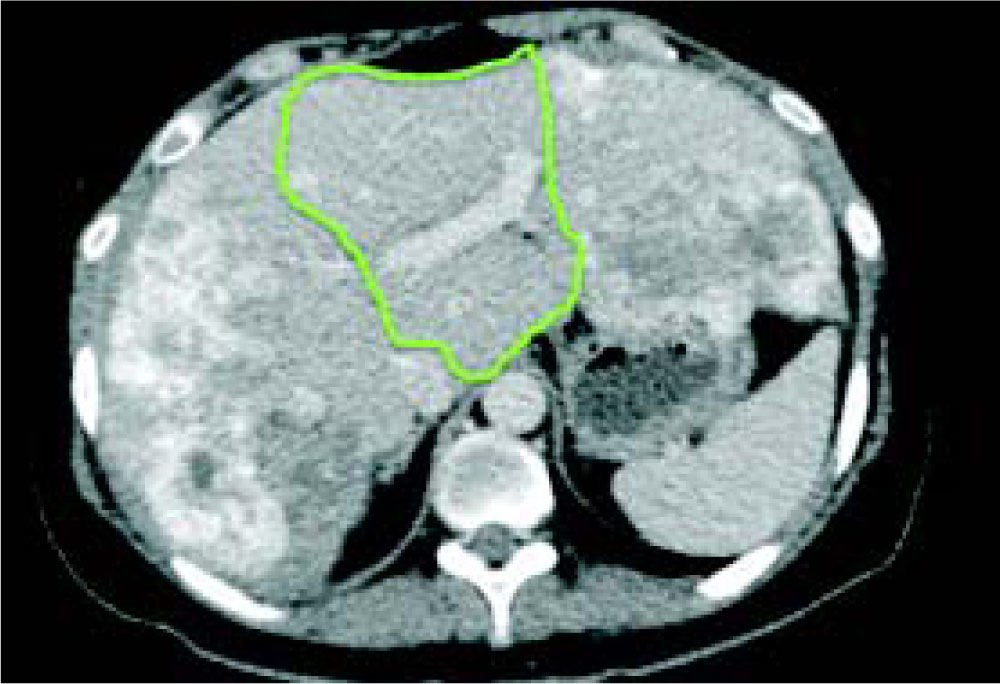
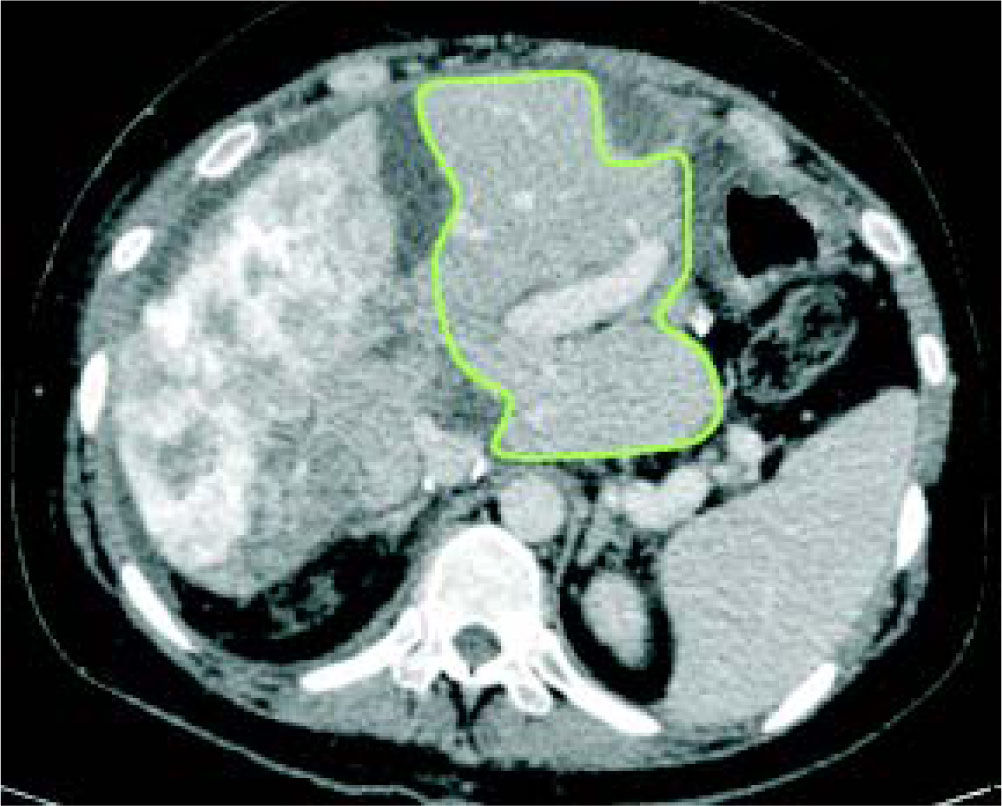
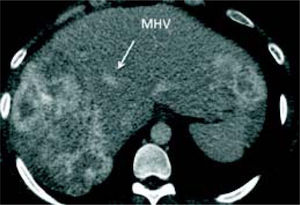
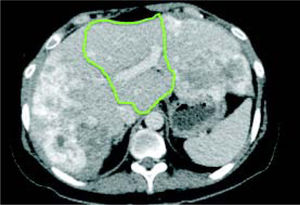
Case reportA 37-year-old female was admitted with a two-month history of progressive epigastric pain, weight loss, fatigue, and dysmenorrhea. Physical examination revealed pale mucosa, tachycardia, and a visible and painful 20 cm mass at the abdominal upper-right-quadrant. There were no stigmas of chronic liver disease. BMI was 27 kg/m2. Patient denied fever, comorbidities, smoking, alcohol, or other substance abuse, and had no previous surgery. Laboratory exams confirmed elevation of serum liver enzymes and INR (AST 54 U/L, ALT 312 U/L, INR 1.39). Serology for hepatitis was negative. CEA value was over 9,000 ng/ mL. An abdominal CT scan showed two bulking liver tumors occupying the right liver and left lateral segments (Figure 1). A colonoscopy revealed a left colon tumor at the spleen flexure, later confirmed as an adenocarcinoma by lesion biopsy. After multidisciplinary team discussion, the patient was referred to chemotherapy and was restaged after six and 13 cycles of Oxaliplatin plus Bevacizumab based therapy. CEA value then dropped to 200 ng/mL. A new CT scan showed that the tumors had reduced in volume, leaving segments 1 and 4 free of disease, but with margins close to the middle hepatic vein (MHV) (Figure 2). Liver-first approach was proposed, but FLR was estimated at 389 g, corresponding to 22% of standard liver volume (SLV) and 0.51 FLR/BW ratio (Figure 3). Based on recent literature, we decided to perform ALPPS seeking to achieve a rapid growth of the FLR.
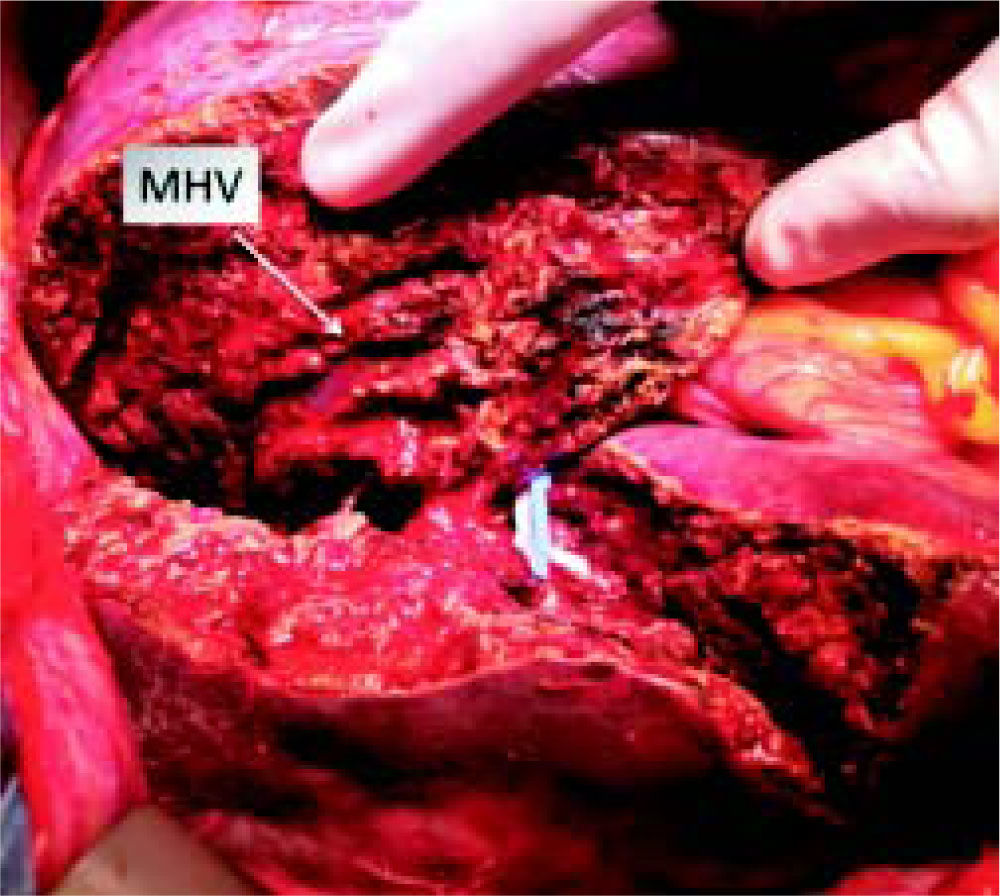
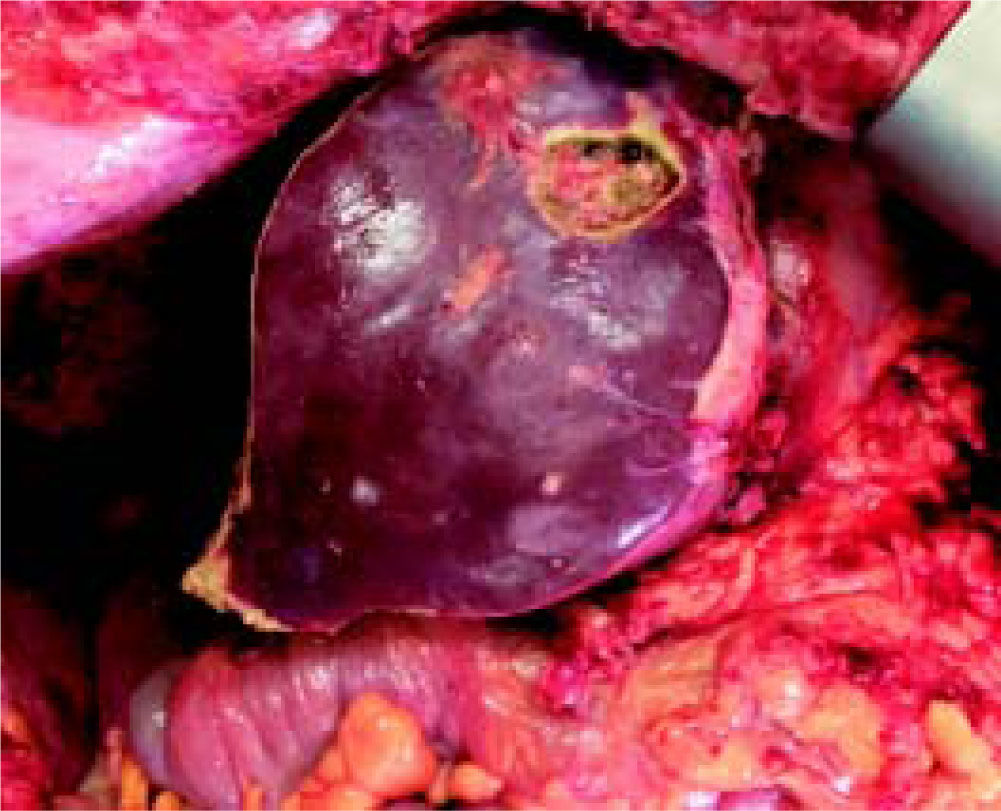
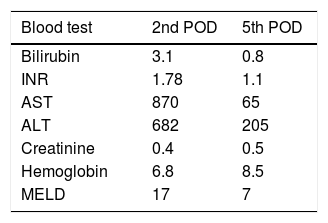
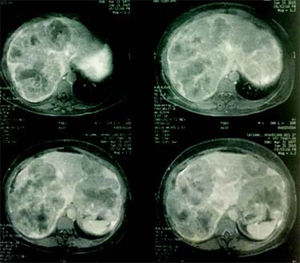
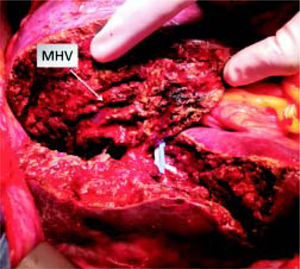
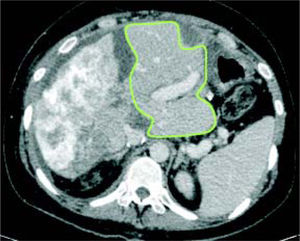
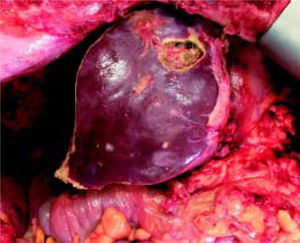
Stage 1 surgery was carried out under open technique with a bilateral subcostal incision with midline extension. Due to the large volume of tumor load preventing liver mobilization, we opted first to do the left lateral segment resection associated with the clean up of hepatic lesions in segment 4, then proceeded with parenchyma transection using the anterior approach and hanging maneuver. Transection of the liver was done using an ultrasonic dissector/aspirator and bipolar diathermy. MHV was dissected with special care not to compromise the outflow of segment 4. The right portal vein was divided and vessel loops were placed around the right hilar plate and the right hepatic vein (Figure 4). The procedure lasted 390 min and the patient received two blood units. Laboratory exams on the 2nd and 5th post-operative days (POD) are described in table 1. Eight days after the stage 1 surgery, a CT scan revealed growth of the FRL to 633 g (an increase of 62%), corresponding to 36% of SLV and 0.83 FLR/BW ratio (Figure 5). Stage 2 surgery was carried out on the 10th POD without complications (Figure 6). The patient developed ascites and mild encephalopathy after stage 2, probably due to venous congestion on liver remnant, but recovered promptly with no infections or surgical complications. A control CT scan was done on the 19th POD and the patient was discharged on the 20th POD. She was subjected to a left colectomy after six months and is disease-free twenty one months after surgery.
Since the initial series published by Schnitzbauer, et al.,1 later named by Santibañes and Clavien,2 ALPPS has been regularly performed in many centers around the globe. Initially reported for patients with primary or metastatic liver tumors,1 ALLPS is now commonly recommended for patients with CRLM, especially in the presence of bilobar disease.7 In cases of HCC, intrahepatic cholangiocarcino-ma, and perihilar cholangiocarcinoma, ALPPS should be restricted due to higher associated morbidity and mortali-ty.7 Originally performed for a FLR consisting of the left lateral segment with or without segment 4,1 ALPPS has been adapted, and reports using the right posterior segments, two separate segments, or even one segment as FLR have been released.3-6
We present here a case of a young woman with large bilobar CRLM, initially considered unresectable. However, after preliminary response to chemotherapy, the patient was submitted to ALPPS leaving segments 1 and 4 as liver remnant. According to a “consensus” proposed by specialists, this kind of surgery should be called segment 4-1 ALPPS.8 This technique was first described by Santi-bañes, who performed it on two patients with CRLM.4 In a recent retrospective analysis of the ALPPS registry (www.alpps.net), Schadde, et al. considered this kind of surgery as monosegment ALPPS (segment 1 was considered an accessory segment). Twelve patients were evaluated in this analysis, all of them with CRLM.9 The median time between stage 1 and 2 was 13 days (range 6-21) and the median degree of hypertrophy was 160% (range 93-250). Four out of the 12 patients experienced liver failure after stage 2. Eight out of 10 patients reaching 1-year follow-up were alive at 1 year (80%) and five out of 10 patients reaching 1-year follow-up were disease free at 1 year (50%). Our patient had similar results, with the exception of lower liver hypertrophy, probably due to congestion of the FLR. She is now in good condition and disease-free 21 months after surgery. Her last CEA value was 1.8 ng/mL. Naturally, ongoing followups will be necessary to determine the real disease-free and overall survivals.
We understand that ALPPS is the decisive treatment for a select group of patients with advanced CRLM. Advanced liver resections for small liver remnants composed by one or two segments are feasible and safe, but should be performed by a multidisciplinary hepatobiliary team composed by surgeons, anesthesiologists, intensive care specialists, and hepatologists with great expertise.
Supportive foundationsThere was no funding source.
Conflicts of InterestThere are no conflicts.
Abbreviations- •
ALPPS: associated liver partition and portal vein liga-tion for staged hepatectomy.
- •
BW: body weight.
- •
CRLM: colorectal liver metastasis.
- •
FLR: future liver remnant.
- •
MHV: middle hepatic vein.
- •
POD: post operative day
- •
SLV: standard liver volume.