Background. Bile acid sequestration (BAS) with resins has shown antidiabetic effects in both humans and animals. Since hepatic steatosis is commonly associated with type 2 diabetes mellitus and the effects of BAS on steatosis have not been explored in detail, we evaluated the effects of cholestyramine (CTM) administration on fatty liver development in the leptin-deficient obese mice.
Aim. To study the effects of BAS on fatty liver development in obese (ob/ob) mice.
Material and methods. 4 week-old ob/ob mice (B6.V-Le-pob/J, n = 4-6 per group) were fed with or without CTM (control group) during 8 weeks. Serum and biliary parameters, glucose tolerance test (GTT), hepatic triglyceride content, liver histology and hepatic gene expression of relevant genes related to bile secretion, lipid and glucose metabolism were assessed.
Results. Control 12-week-old mice exhibited marked obesity and hepatic steatosis. CTM administration expectedly determined a marked de-repression of 7-α-hydroxylase and decreased biliary bile acid secretion as well as improved GTT. CTM feeding showed no effects on hepatic triglyceride content or in the degree of steatosis on liver histology. CTM was associated with increased levels of serum alanine-aminotransferase.
Conclusion. Although CTM administration positively affects glucose tolerance it does not prevent hepatic steatosis development in obese mice. Moreover, CTM feeding was associated to liver enzyme elevation in this model of NAFLD. Thus, the effects BAS on NAFLD need to be specifically addressed since this therapy might not be beneficial for this condition.
Non-alcoholic fatty liver disease (NAFLD) is now considered the most common form of liver disease worldwide and its presence is associated with an increased liver-related mortality.1 NAFLD encompasses a spectrum of histological abnormalities ranging from simple steatosis to non-alcoholic steatohepatitis (NASH), which is characterized by steatosis plus necroinflammatory changes and various degrees of hepatic fibrosis.2 Patients with NASH are considered to have a more aggressive form of the disease and carry the risk of developing cirrhosis and hepatocellular carcinoma.3
The pathogenesis of NAFLD is not fully understood but it is associated with obesity and insulin resistance (IR).4,5 IR results in alterations on deposits of lipids and lipolysis in insulinosensitive tissues, which induces an increased flow of fatty acids from adipose tissue to the liver causing steatosis.6 The accumulation of triglycerides in the liver determines several alterations, including resistance to insulin action in this organ and oxidative stress that is associated with cell damage, necrosis and inflammation, determining the potential development of NASH and disease progression.6 At present time, lifestyle interventions and behavior therapy, together with drugs used to treat the associated diseases of the metabolic syndrome (hypertension, diabetes, or dyslipidemia), represent the main therapeutic approach available to the clinician.1 In addition, biopsy-proven NASH could benefit from either vitamin E or pioglitazone administration. Of note, these therapies have been mainly tested in non-diabetic patients even though type 2 diabetes mellitus (T2DM) patients are at higher risk of developing hepatic steatosis and NASH compared with non-diabetic individuals.7–9 In fact, in the subgroup of diabetic patients NAFLD tends to be more frequent and more severe.7 Indeed, a good metabolic control of T2DM could be beneficial for both simple steatosis and NASH and therefore optimal antidiabetic strategies should be attempted.
Recently, bile acid sequestration (BAS) using anionic exchange resins such as cholestyramine (CTM) and colesevelam has been shown to be effective improving glycemic control in both T2DM patients and experimental animals.10,11 Resins prevent bile acid reabsorption in the terminal ileum and promote their fecal excretion through the formation of non-absorbable complexes that escape from the efficient transport system located in the ileal enterocyte.11 One of the main mechanism invoked to explain the antidiabetic action of BAS is an increased production of glucagon-like peptide-1 (GLP-1) from enteroendocrine L-cells of the intestinal epithelium through the activation of the G-protein associated receptor TGR5.12 Independently of the mechanisms, theoretically BAS should have a beneficial effect on hepatic steatosis due to its potential effects on insulin resistance or glucose metabolism although this has not been directly tested. The present study assessed the hepatic effects of BAS using CTM on fatty liver development in a genetic rodent model of NAFLD.
Material and MethodsAnimals and treatmentFour-week-old male ob/ob mice (B6.V-Lepob/J) were purchased from Jackson Laboratory (Maine, USA). In selected experiments, chow-fed C57BL6 male mice of 12 weeks of age were used as controls. Obese mice were fed a standard rodent chow (control group) or chow supplemented with 3% w/w CTM from 4 to 12 weeks of age. Each group of animals (n = 4-6) were housed in transparent polycarbonate cages subjected to 12 h light/darkness cycles under a temperature of 21 °C and a relative humidity of 50%. All procedures were performed according to the local ethics review committee on animal experiments. At 12 weeks of age animals were anesthetized with an intraperitoneally administered dose of a mixture of xylazine (10 mg/kg) and ketamine (100 mg/kg). Subsequently, blood samples were taken and livers were removed, frozen in liquid nitrogen and stored at −80 °C until analyzed. Also, before euthanasia, bile collection was carried out after cannulation of the common bile duct through the gallbladder, with a PE-10 polyethylene tube as previously described.13
Histological studiesSections (5-mm thick) from the right lobe of all mice livers were routinely fixed in 10% formalin and embedded in paraffin. Then 4-mm thick sections were stained with hematoxylin/eosin. An investigator blinded to experimental groups evaluated the slides and assigned a score for steatosis, inflammation and fibrosis as described.14 The scores were given as it follows.
Steatosis:
- •
Grade0: none present.
- •
Grade1: steatosis of < 25% of parenchyma.
- •
Grade2: steatosis of 26-50% of parenchyma.
- •
Grade3: steatosis of 51-75% of parenchyma.
- •
Grade4: steatosis of > 76% of parenchyma.
Inflammation:
- •
Grade0: no inflammatory foci.
- •
Grade1: < 5 inflammatory foci per high power field (hpf).
- •
Grade2: > 5 inflammatory foci/hpf.
Bile flow was measured gravimetrically and total biliary bile acids were quantitated by the 3-alpha-hydroxysteroid dehydrogenase method.15,16 Total biliary phospholipids, cholesterol and glutathione were measured by standard methods.17 Serum cholesterol and triglycerides were measured using kits from Human (Wiesbaden, Germany), serum alanine aminotransferase (ALT) was quantited with Kovalent kit (Río de Janeiro, Brazil), serum bile salts were measured with Randox kit (Antrim, UK) and liver triglycerides were assessed according to Carr, et al.18
Glucose tolerance testFor the glucose tolerance test (GTT) mice were fasted for 12 h with free access to water. Glucose (1 g/kg) was administered intraperitoneally and blood glucose from tail was measured with One Touch glucometer (Johnson & Johnson Medical) at 0, 15, 30, 60, 90 and 120 min as described by Ozcan, et al.19
Quantitative real-time PCRReal-time PCR technique was used to quantify mRNA expression of selected genes and to assess the hepatic response to BAS. We measured the gene expression of cholesterol 7-α-hydroxylase (Cyp7a1) and mRNA levels of the orphan nuclear receptor short heterodimer partner (Shp). We also measured key genes of hepatic bile acid transport (the Na+-taurocholate cotransporting polypeptide Ntcp and the bile salt export pump Bsep), and of both lipid (the transcription factor Srebp-1c) and carbohydrate metabolism (phosphoenolpyruvate carboxykinase, Pepck). Total RNA was isolated from whole-liver tissue using Speed Vacuum Total RNA Isolation System (Promega Corporation, Madison, WI). RNA was quantified by measur-ing absorption at 260 nm on a Nanodrop spectrophotometer ND-1000. First-strand cDNA was synthesized from 1 μg of total RNA with random hexamer primers using the ImProm-II™ Reverse Transcription System (Promega Corporation, Madison, WI). The real-time PCR contained, in a final volume of 20 μL, cDNA, TaqMan® Universal PCR Master Mix 2x and TaqMan® MGB probes, FAM® dye-labeled (Applied Biosystems, CA). Primers designed by Primer Express Software (Applied Biosystems, CA) were used and PCR was carried out in 96-well plates. The relative amounts of all mRNAs were calculated using the comparative threshold cycles (ΔCT) method. 18S mRNA was used as the invariant control for all experiments.
StatisticsAll results are expressed as mean ± standard deviation (SD). A two-tailed non-paired Student’s t-test was used to compare differences between groups. For statistical analysis of glucose tolerance test curves we compared the area under the curve with the nonparametric Mann-Whitney test, considering one tail exact significance. Areas under curves were calculated using the trapezoidal rule. Values were considered significantly different when the P value ≤ 0.05.
ResultsBody and liver weight, serum biochemistry, hepatic triglyceride content and liver histology of chow-fed obese miceOb/ob mice exhibit a greater body and liver weight compared to normal C57BL6 at week 12 of life and increased serum levels of ALT, insulin and cholesterol (Table 1). Liver histology assessment at different time points (4, 7, 8 and 12 week-old mice) showed that steatosis development started at week 8, reaching maximum severity at week 12 as it has been previous described.20 In line with this finding, hepatic triglyceride content in 12 weeks-old obese mice was significantly higher compared to C57BL6 mice (111 ± 19 mg/g liver vs. 12.5 ± 4.5 mg/g liver, p < 0.05).
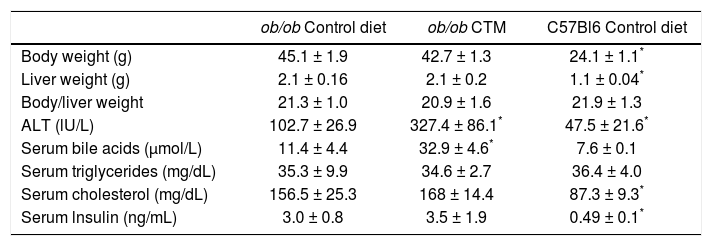
Effects of oral administration of cholestyramine (CTM) on body/liver weight and serum parameters in ob/ob mice.
| ob/ob Control diet | ob/ob CTM | C57Bl6 Control diet | |
|---|---|---|---|
| Body weight (g) | 45.1 ± 1.9 | 42.7 ± 1.3 | 24.1 ± 1.1* |
| Liver weight (g) | 2.1 ± 0.16 | 2.1 ± 0.2 | 1.1 ± 0.04* |
| Body/liver weight | 21.3 ± 1.0 | 20.9 ± 1.6 | 21.9 ± 1.3 |
| ALT (lU/L) | 102.7 ± 26.9 | 327.4 ± 86.1* | 47.5 ± 21.6* |
| Serum bile acids (μmol/L) | 11.4 ± 4.4 | 32.9 ± 4.6* | 7.6 ± 0.1 |
| Serum triglycerides (mg/dL) | 35.3 ± 9.9 | 34.6 ± 2.7 | 36.4 ± 4.0 |
| Serum cholesterol (mg/dL) | 156.5 ± 25.3 | 168 ± 14.4 | 87.3 ± 9.3* |
| Serum lnsulin (ng/mL) | 3.0 ± 0.8 | 3.5 ± 1.9 | 0.49 ± 0.1* |
Experimental groups are as described in Material and Methods. Data are mean ± SD (n = 4-6).
Feeding 4 week-old ob/ob mice during 8 weeks a chow diet supplemented with CTM was not associated with changes in body weight, liver weight and serum levels of triglycerides, cholesterol and insulin (Table 1). No differences were found in the grades assigned by the pathologist to steatosis or inflammation in livers from the experimental groups. Of note, serum ALT levels were significant increased by three-fold in CTM-fed mice.
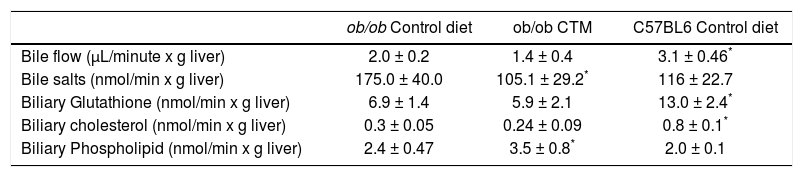
Hepatobiliary parameters were also determined to better explore the effects of experimental diet and given the relationship between obesity, insulin resistance and altered bile secretory function described in the literature by our group and others.21,22 As shown in table 2, 12 weeks-old ob/ob mice exhibited cholestasis with a significantly reduced bile flow. Administration of CTM decreased biliary excretion of bile acids and increased biliary phospholipid excretion. Besides, ob/ob mice showed a significant decrease in biliary glutathione excretion which was not affected by CTM feeding.
Effect of oral administration of cholestyramine (CTM) on hepatobiliary parameters in ob/ob mice.
| ob/ob Control diet | ob/ob CTM | C57BL6 Control diet | |
|---|---|---|---|
| Bile flow (μL/minute x g liver) | 2.0 ± 0.2 | 1.4 ± 0.4 | 3.1 ± 0.46* |
| Bile salts (nmol/min x g liver) | 175.0 ± 40.0 | 105.1 ± 29.2* | 116 ± 22.7 |
| Biliary Glutathione (nmol/min x g liver) | 6.9 ± 1.4 | 5.9 ± 2.1 | 13.0 ± 2.4* |
| Biliary cholesterol (nmol/min x g liver) | 0.3 ± 0.05 | 0.24 ± 0.09 | 0.8 ± 0.1* |
| Biliary Phospholipid (nmol/min x g liver) | 2.4 ± 0.47 | 3.5 ± 0.8* | 2.0 ± 0.1 |
Experimental groups are described in Material and Methods. Data are mean ± SD (n = 4-6).
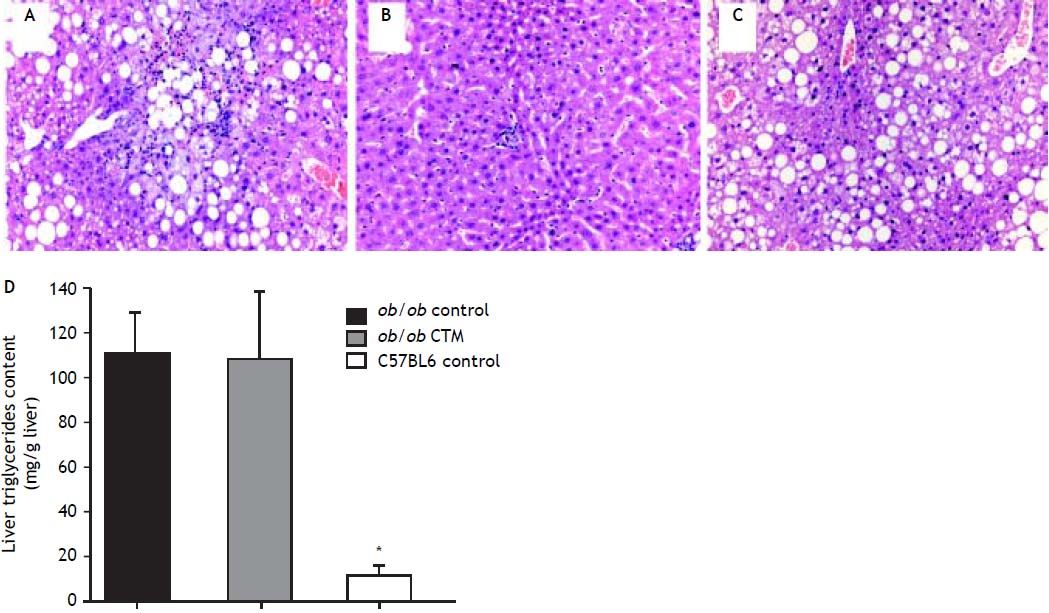
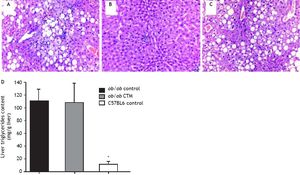
Representative histological evaluation of liver samples after 8 weeks of experimental diet revealed that administration of CTM had no effect on steatosis development in obese ob/ob mice (Figure 1). Determination of hepatic triglyceride content revealed that obese mice had a nine-fold increase in this variable compared to non-obese mice. CTM administration had no influence on liver triglycerides (Figure 1).
Effects of cholestyramine (CTM) administration on liver histology and hepatic triglyceride content in ob/ob mice. Upper panel: 12-week-old ob/ob mice (A) developed marked steatosis compared to chow-fed, age-matched C57Bl6 mice (B). CTM administration during 8 weeks did not prevent liver steatosis development in obese ob/ob mice (C) (haematoxylin-eosin stain, 20 x). Lower panel: D. Hepatic triglycerides content (mg/g liver) in chow-fed and CTM-fed ob/ob mice compared with a chow-fed control non-obese (C57BL6) mice. Results are as mean ± SD (n = 4-6).
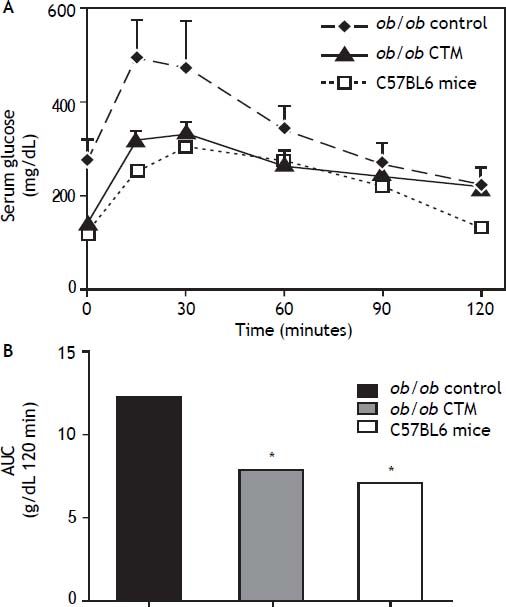
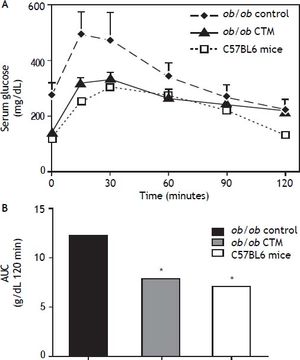
Figure 2 shows results obtained in studies of glucose tolerance (GTT) in different experimental groups. Analysis of the area under the curve of the different groups shows that ob/ob mice exhibited a marked intolerance to glucose compared with the control group (one-tail significance p = 0.01) and CTM was able to significantly reduce this parameter after 8 weeks of treatment (p = 0.01).
Effects of cholestyramine (CTM) administration on glucose tolerance in ob/ob mice. A. Plasma glucose concentrations during the intraperitoneal glucose tolerance test (1 g/kg) following fasting for 12 h in C57BL6, control ob/ob and ob/ob treated with CTM mice. Results are means ± SD (n = 4-6). B. Areas under curves were calculated using the trapezoidal rule. * One-tail significance p = 0.01.
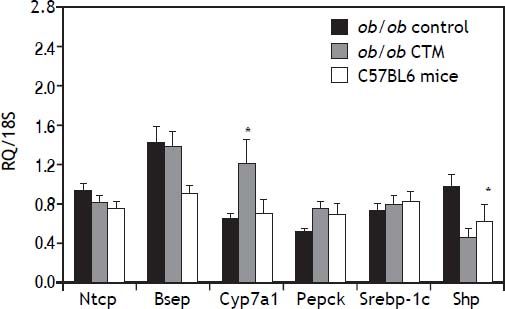
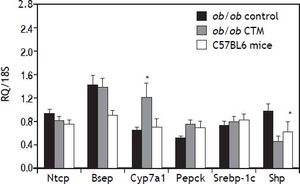
Assessment of the expression of genes that are known to be regulated by BAS such as CYP7A1 and SHP is shown in figure 3. As expected, CTM feeding was associated with a marked induction of Cyp7a1 and to a reduction of Shp mRNA levels, which is consistent with an effect of deactivation of the nuclear receptor FXR determined by a decrease in the bile acid pool size. On the other hand, CTM did not influence the expression of genes involved in hepatic bile acid transport (Ntcp and Bsep), lipogenesis (Srebp-1c) or carbohydrate metabolism (Pepck).
Effects of cholestyramine (CTM) administration on hepatic expression of selected genes: Na+-taurocholateco-transporting polypeptide (Ntcp); bile salt export pump (Bsep); Cholesterol 7 a-hydroxylase (Cyp7a1); sterol regulatory element binding transcription factor 1 (Srebp-1c); Phosphoenolpyruvate carboxykinase (Pepck); short heterodimeric partner (Shp). Results are means ± SE (n=4-6). * p<0.05 compared to ob/ob control.
The use of BAS using resins such as CTM or colesevelam has been proposed as a potential new approach to the management of T2DM based in both clinical and experimental studies showing that BAS is able to lower plasma glucose and glycosylated hemoglobin levels as well as to improve tissue glucose metabolism.23,25 However, long-term effects of BAS in T2DM patients are still insufficiently characterized. Also, specifically with regard to NAFLD, the use of BAS has not been well addressed. The latter is relevant since the prevalence of NAFLD in T2DM patients is considerable with a higher prevalence of NASH, the aggressive form of the disease.26 Thus, BAS therapeutic potential as well as safety in this specific patient population needs to be explored. In this context, the present study evaluated the hepatic effects of CTM on NAFLD development in leptindeficient ob/ob mice, a widely used obese mouse model of NAFLD.
The results of this work show that BAS did not produce any positive effects on liver steatosis development in the ob/ob mice. Neither histology nor hepatic triglyceride content was influenced by CTM administration. Our data is in agreement with observations made in other animal studies.27,28 Indeed, Herrema, et al.27 reported that BAS are able to induce the lipogenic pathway in an FXR- and LXRα-dependent manner leading to the increase of liver triglyceride droplets. Other studies have observed beneficial effects of BAS on hepatic steatosis in genetically diabetic mice.29,30 These differences could be explained in part by differences in the experimental models. It has been established that BAS produce malabsorption of dietary fatty acids in the intestine due to impaired micelle formation.28 Since Matsumoto, et al. and Kobayashi, et al. fed mice a high-fat diet, CTM might have prevented the absorption of the fat content of the diet and therefore an important part of the caloric input. On the other hand, it could be possible that the effect of CTM in diabetic mice fed chow diet, as used in the Herrema, et al. and Meissner, et al. reports and in the current study, is lost since carbohydrates are the major part of the composition of this diet. It is known that high intake of carbohydrates in obese and diabetic patients may lead to increase liver steatosis through the activation of de novo lipogenesis in hepatocytes.31 Thus, it is suggested that the action of BAS in liver steatosis may depend on the macronutrients composition of the diet. The use of genetic rather than dietary models is likely more accurate to test the effects of BAS on hepatic steatosis.
Noteworthy, 8 weeks of CTM treatment produced a significant increase in serum levels of ALT and bile acids implying the occurrence of liver injury in ob/ob mice. These observations are in line with a recent report in humans where colesevelam treatment increased serum ALT levels, being this outcome associated with a modest increase in liver fat content when measured by a sensitive magnetic resonance technique.32 These authors suggest that BAS may lead to a compensatory increase in bile acid synthesis and fatty acid synthesis in the liver although we did not observed changes in these two compensatory mechanisms judging by gene expression analysis. A recent study in mice had shown that CTM feeding tended to increase hepatic levels of cytotoxic bile acids such as chenodeoxycholic acid and lithocholic acid and also decrease hepatic levels of the hydrophilic bile acid beta-muricholic acid.33 These changes in the bile acid pool might be related to liver damage in an injury-prone organ such as the steatotic liver.34 Thus, if long-term CTM administration is harmful in the setting of hepatic steatosis warrants further study.
The current work also found a significant improvement in glucose tolerance in CTM-treated mice in agreement with previous reports. In this regard, it has been proposed that BAS may act increasing the metabolic glucose clearance by peripheral tissues while hepatic glucose output remains unaffected.28 Also, some studies have demonstrated that BAS are able to induce the expression of GLP-1 by stimulation of L-cells in the terminal ileum or colon.35,36 Finally, it has been recently suggested that increased plasma cholecystokinin concentrations induced by BAS treatment may cont ribute to postprandial glucose control via a delay in gastric emptying.37 These mechanisms could explain the beneficial effects observed by CTM in the present study on reducing blood glucose levels. Why the improvement in GTT does not translate in fatty liver improvement is unclear. We aimed to explore this assessing the hepatic gene expression of selected relevant genes. CTM treatment evoked the expected effects on mRNA levels of Cyp7a1 and Shp, likely due to a deactivation of the nuclear receptor FXR. Decreased activation of SHP could result in relative activation of liver receptor homolog-1 and liver-x receptor a, which ultimately results in increased triglyceride synthesis. Of note, no changes were seen in the expression of major bile acid transporters Ntcp and Bsep after CTM feeding.
Among the limitations of the present study it should be kept in mind that, in spite of the agreement with some human data,32 these findings cannot be directly extrapolated to humans since mice and men exhibit multiple differences in biliary physiology.38,39 Also, giving the emerging role of bile acids in energy metabolism the explanation of the dissociation between the positive impact on glucose metabolism and the lack of effect in the liver could be related to effects in other organs such as brown adipose tissue and skeletal muscle which could have a role in amelioration of glucose metabolism.40
In summary, BAS with CTM reduces glucose plasma levels in obese, diabetic mice. However, the results obtained in this work demonstrate that BAS does not ameliorate hepatic steatosis and can also exert some deleterious effects in the liver in some circumstances. Thus, our findings and those from Le, et al.32 suggest that BAS might not be beneficial for the treatment of NAFLD and its long-term safety in this regard needs to be addressed. This is particularly important considering the proposed role of BAS in the treatment of diabetic patients a population at-risk of liver complications due to the increased prevalence of NAFLD and its potentially progressive form NASH.26,41
Abbreviations- •
ALT: serum alanine aminotransferase.
- •
BAS: bile acid sequestration.
- •
BSEP: bile salt export pump.
- •
CTM: cholestyramine.
- •
CYP7A1: cholesterol 7-alpha-hydroxylase.
- •
GLP-1: glucagon-like peptide-1.
- •
GTT: glucose tolerance test.
- •
IR: insulin resistance.
- •
NAFLD: non-alcoholic fatty liver disease.
- •
NASH: non-alcoholic steatohepatitis.
- •
NTCP: Na/Taurocholate Cotransporting Polypeptide.
- •
SHP: small heterodimer partner.
- •
SREBP-1C: sterol regulatory element-binding protein-1c.
- •
T2DM: type 2 diabetes mellitus.
- •
TGR-5: G protein-coupled bile acid receptor 1.
This study was carried out with support of grants from the Fondo Nacional de Investigación Científica y Tecnológica (FONDECYT, project 1110455) and the Comisión Nacional de Investigación, Ciencia y Tecnología (CONICYT, project ACT 79).