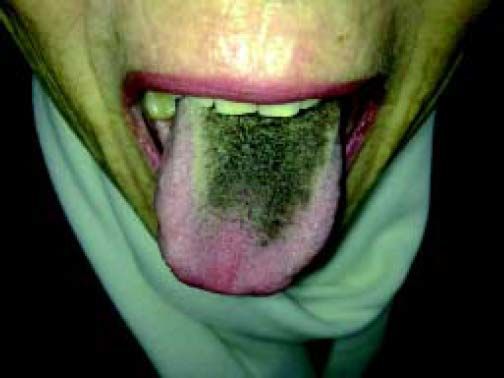
A 64-year-old woman with history of chronic hepatitis related to Hepatitis C virus [(HCV), genotype 1b] infection received treatment with pegylated interferon (PEG-IFNα2a) and rivabirin noticed a black coloration of her tongue during the thirty-third week of treatment (Figure 1). She had no other relevant past medical history and was a nonsmoker. She did not refer any pain, discomfort or other symptoms in the oral cavity. Physical examination showed a black hyperpigmentation and elongation of the phylliform papillae on the dorsal surface of the tongue. After appropriate diagnosis and counseling, pigmentation attenuated and later disappeared allowing completion of therapy.
Black hairy tongue (BHT), also called lingua villosa nigra, is a benign disorder characterized by hypertrophied and elongated phylliform papillae on the surface of the tongue, caused by defective desquamation.1,2 Its clinical expression is an abnormal black coating of the dorsal surface of the tongue, usually in the posterior third. Associated involvement of the skin and oral mucosa has been described.3 Hyperpigmentation is usually asymptomatic, although symptoms such as altered taste, nausea, and halitosis have been reported.4,5
The prevalence of BHT in general population is variable, with higher rates described in oral medicine patients.1 Tobacco use is the more frequent aggravating or causative factor. Excessive alcohol intake, xerostomia, poor oral hygiene, oxidizing mouthwashes, and a number of drugs have been also associated to this disorder. Among medications antibiotics, antipsychotics, proton pump inhibitors, immunosupresors, antifungals and oral contraceptives are mentioned in different reports. They most likely share the potential to cause xerostomia.1
Many types of adverse skin reactions, mainly of a lichenoid type, have been described during therapy with PEG-IFN for chronic HCV infection, with a synergistic effect of combination therapy with ribavirin. Eczema, erythema and rash are common.6 BHT has been reported as a rare adverse reaction of PEG-IFN therapy for HCV, with nearly 20 cases described in literature. Reported cases include predominantly dark-skinned patients,3,4,6–8 with few reports on Caucasian patients.9–11 The temporal relationship between the onset of the treatment and the appearance of the pigments spots range from 1 to 10 months.3 It appears not to be related to age, gender, HCV genotype, and doses of PEG-IFN or ribavirin, duration of treatment or treatment outcome.3 Although the exact mechanism underlying oral hyperpigmentation in patients treated with PEG-IFN and ribavirin is still unknown, it has been postulated that increased local melanin production. In fact, increased amounts of melanin have been shown in the basal layer of the tongue epithelium.4 Of note, melanocyte expression of melanocyte stimulating hormone receptors seems to be up-regulated by interferon.6
Differential diagnosis of BHT includes Addison disease, Peutz-Jeghers syndrome, Laugier-Hunziker syndrome, amalgam tattoo, malignant melanoma, nevi, lichen planus pigmentosus, and adverse reactions to other medications.
Therapy should include recommendations for meticulous oral hygiene and modification of any predisposing factors. Brushing or scraping of the tongue can be recommended. More aggressive treatment with topical retinoids, salicylic acid, or surgical excision could be considered. This last approach is usually not necessary, whereas slow improvement occurs in most cases with discontinuation of IFN-based therapy.12
Although PEG-IFN therapy use for HCV is expected to decrease in the near future due to the introduction of new antivirals, this treatment modality will still be employed in a subset of patients and in countries with difficult access to the new agents. Since BHT could be of great concern for patients, clinicians should be aware of this rare and benign adverse reaction to PEG-IFN-based treatment.
Abbreviations- •
bht: black hairy tongue.
- •
hcv: hepatitis C virus.
- •
ifn: interferon.
- •
msh: melanocyte stimulating hormone.
- •
peg-ifn: pegylated interferon.
None.