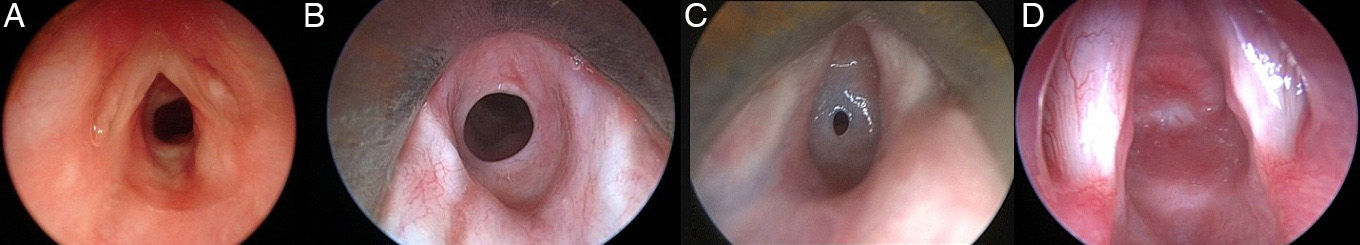
Subglottic stenosis is one of the most common causes of upper airway obstruction. Almost 90% of them result from endotracheal intubation. Therapy depends on the degree of stenosis, among other factors. Therapeutic approaches range from watchful waiting, in mild stenosis, to complex surgery for severe cases. We report our experience on the surgical management of post-intubation subglottic stenosis in children, emphasising the need for recognition and prevention of predisposing factors of post-intubation stenosis.
MethodsWe retrospectively evaluated 71 patients with moderate to severe post-intubation subglottic stenosis, operated in the Respiratory Endoscopy Service in a period of eight years. The clinical variables analysed were age at surgery, degree of stenosis, surgical technique, complications and outcome.
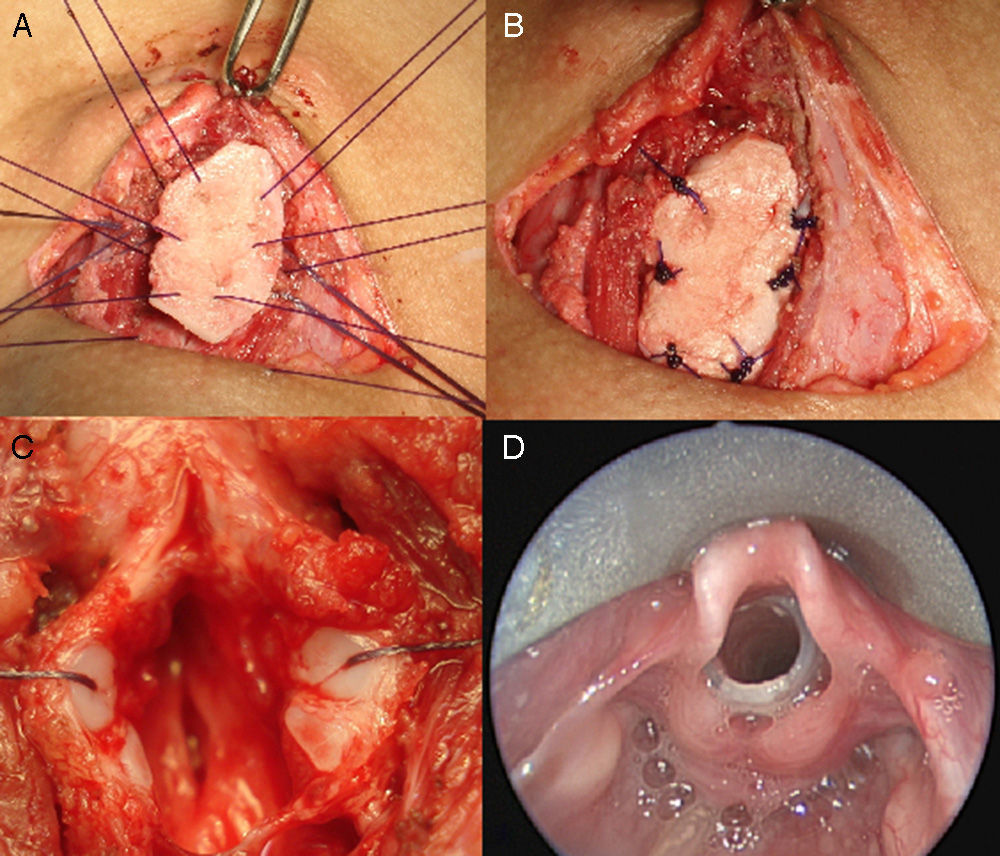
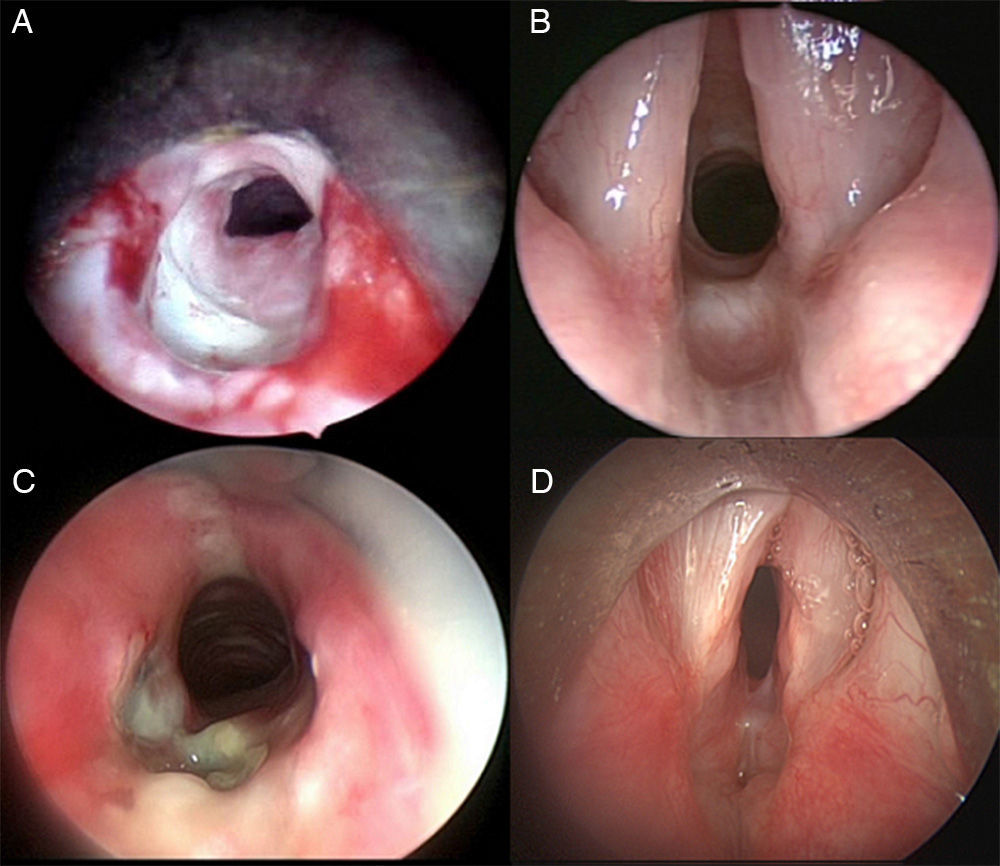
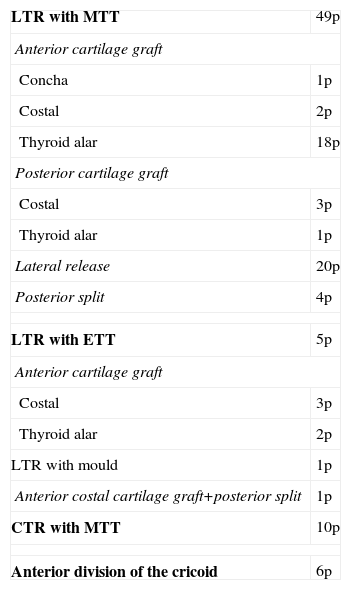
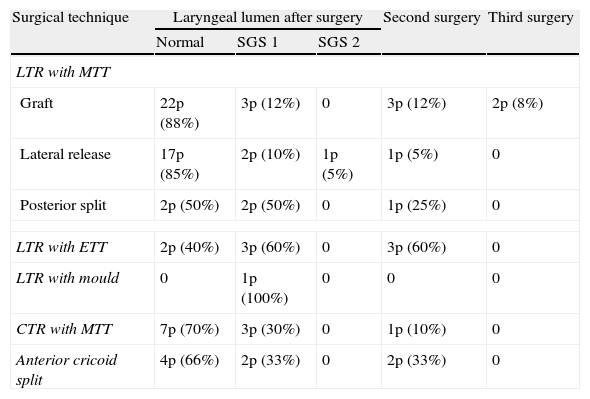
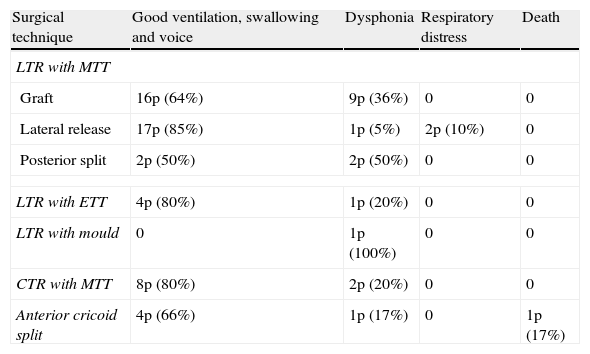
ResultsIn 84.5% of patients, only 1 surgical approach was required to achieve decannulation. Three surgical techniques were implemented as therapy: laryngotracheal reconstruction, partial cricotracheal resection and anterior cricoid split. Decannulation was achieved in 70 cases. In 71.8%, ventilation, swallowing and voice qualities were good; 23.9% presented dysphonia; and 2.8% presented a mild respiratory distress. One patient died.
ConclusionIn patients with subglottic stenosis, selection of the most accurate treatment is the key to success, reducing the number of surgeries and preventing complications.
La estenosis subglótica es una de las causas más frecuentes de obstrucción laríngea. El 90% resultan de la intubación endotraqueal. La conducta terapéutica dependerá entre otros factores del grado de estenosis que se presente. Variará desde la conducta expectante en los grados leves hasta la cirugía compleja en los graves. Presentamos nuestra experiencia en el manejo quirúrgico de la estenosis subglótica postintubación en niños, y enfatizamos la necesidad de reconocimiento y prevención de los factores predisponentes de la estenosis por intubación.
MétodoSe evaluaron en forma retrospectiva 71 pacientes con estenosis subglótica moderada a severa postintubación, operados en el Servicio de Endoscopia Respiratoria durante un periodo de 8 años. Las variables analizadas fueron la edad al momento quirúrgico, el grado de la estenosis, la técnica quirúrgica utilizada, las complicaciones y los resultados.
ResultadosEl 84,5% de los pacientes requirió un solo tratamiento quirúrgico para lograr la decanulación. Se implementaron 3 técnicas quirúrgicas: reconstrucción laringotraqueal, resección cricotraqueal parcial y división anterior del cricoides. Se logró la decanulación en 70 casos, presentando buena ventilación, deglución y voz el 71,8%, disfonía el 23,9% y dificultad respiratoria leve el 2,8%. Un paciente falleció.
ConclusiónFrente a un paciente con estenosis subglótica, seleccionar el tratamiento apropiado es la llave del éxito, disminuye el número de cirugías y previene las complicaciones.