The increasing expertise of transnasal endoscopic surgery has recently expanded its indications to include the management of tumours affecting the skull base. We report our experience with endoscopic management of these tumours, emphasising the indications and surgical technique used.
Material and methodA retrospective analysis was performed of patients treated by an endoscopic endonasal approach (EEA) in our department from 2004 until 2011.
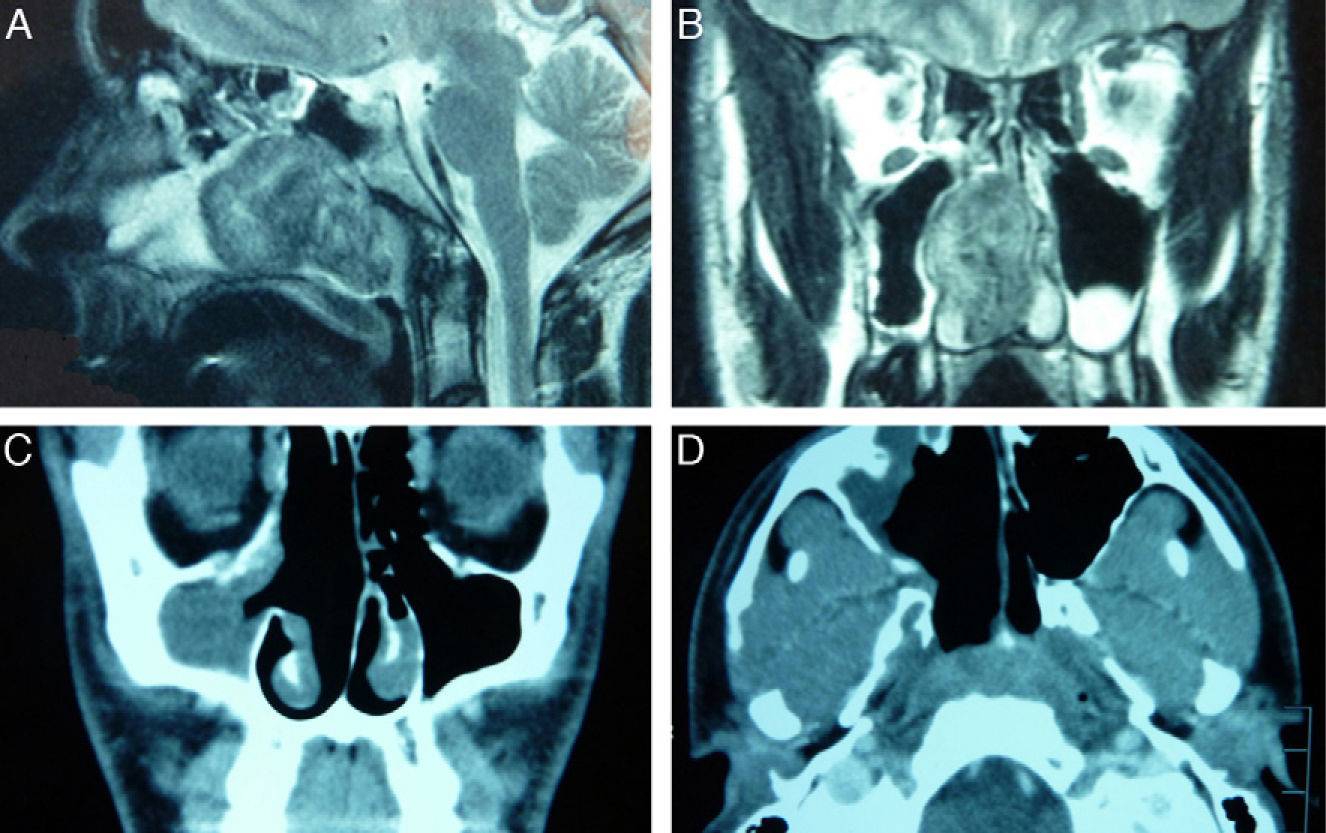
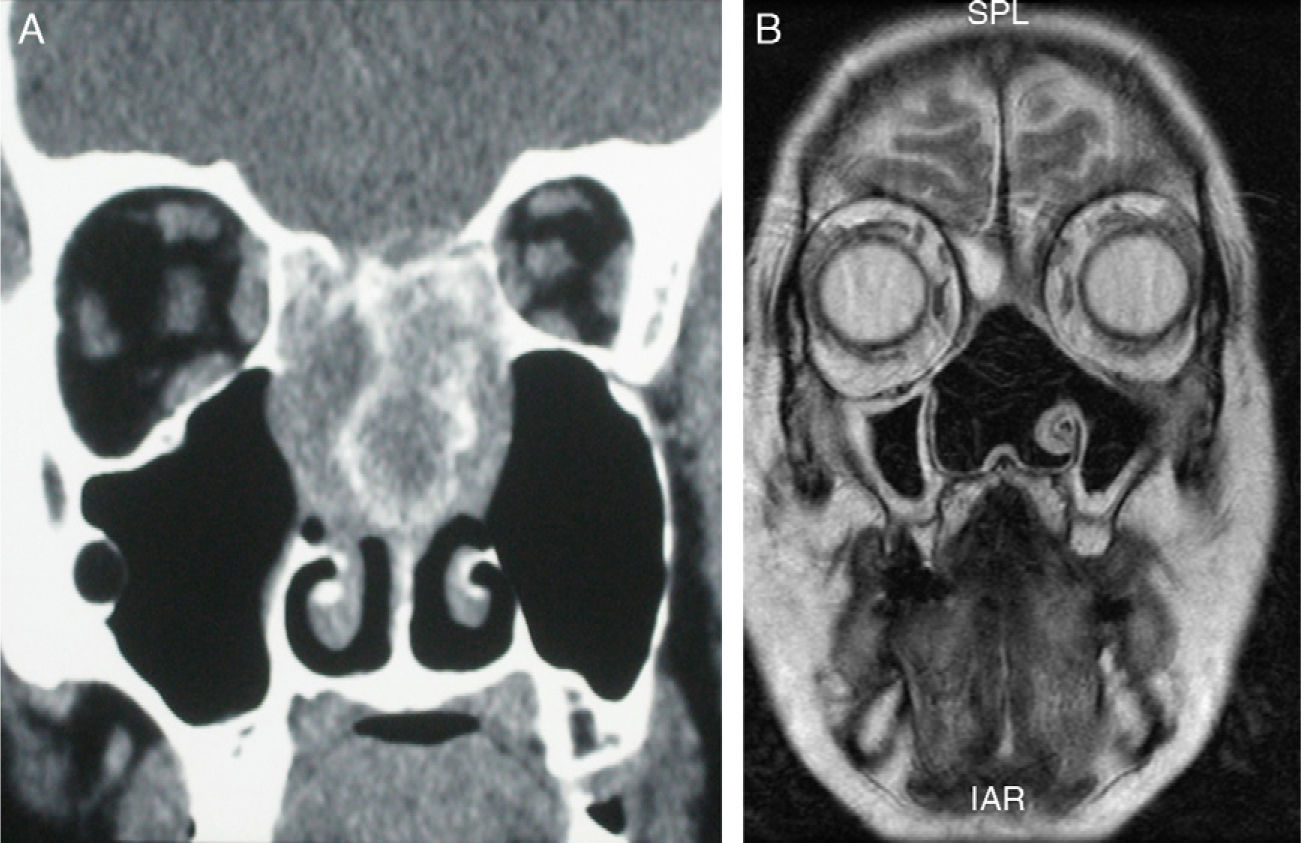
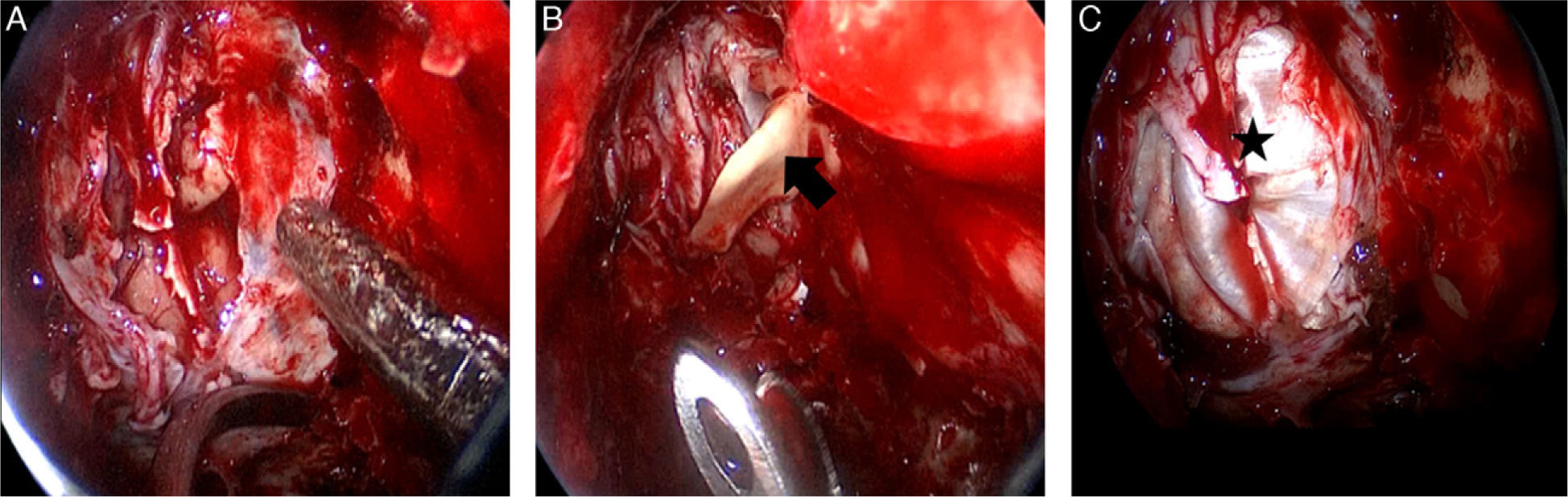
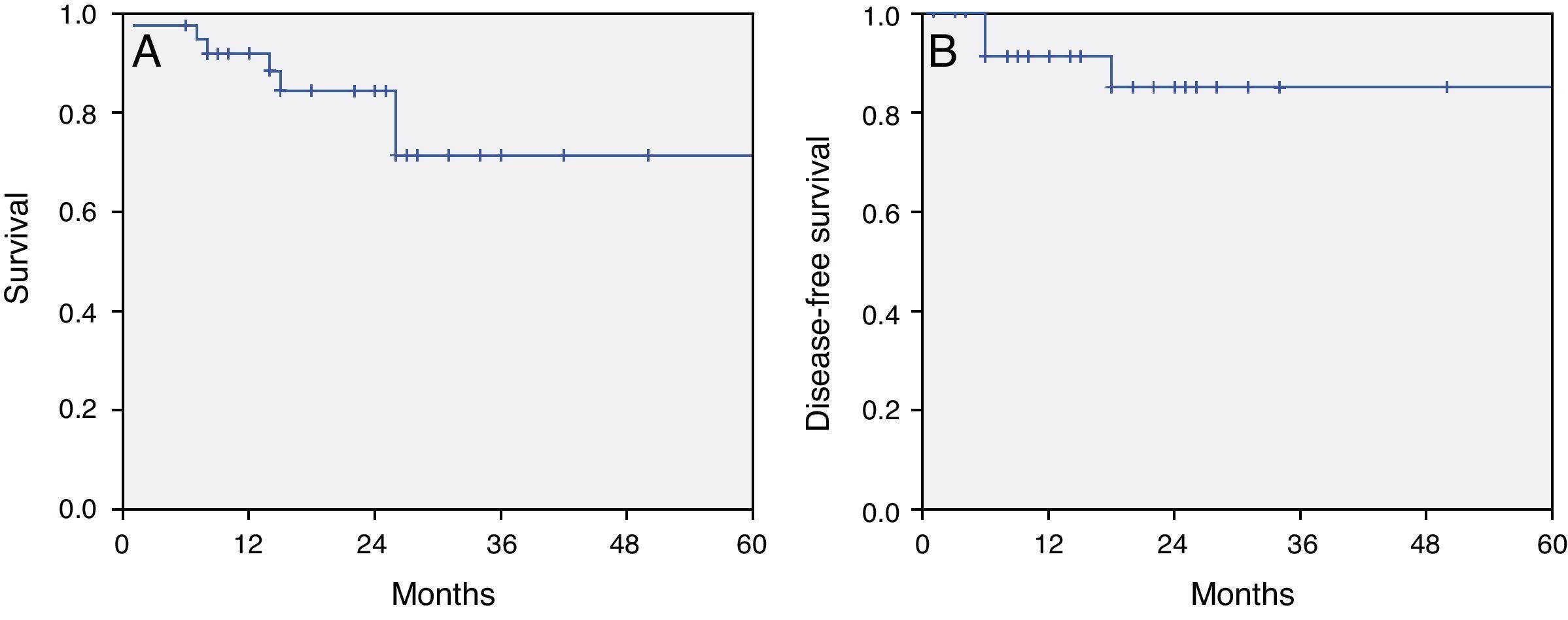
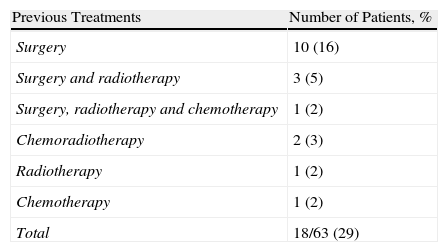
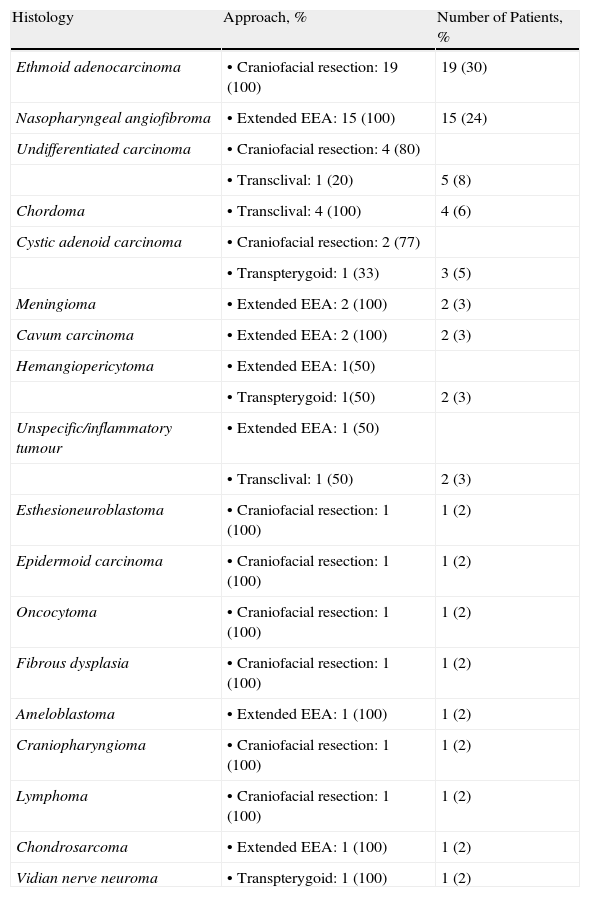
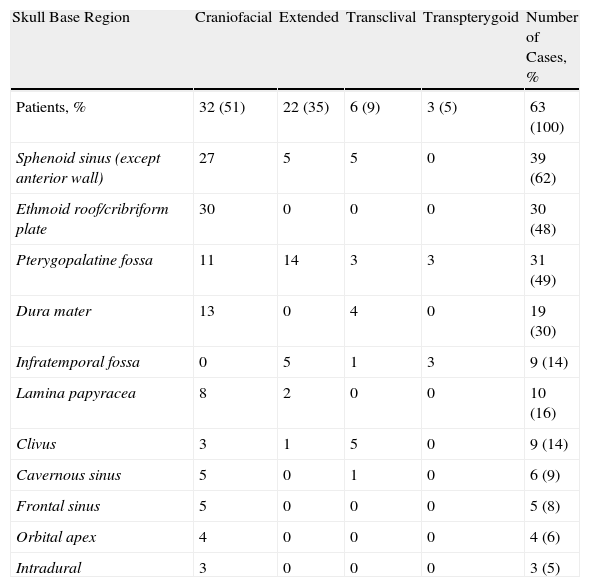
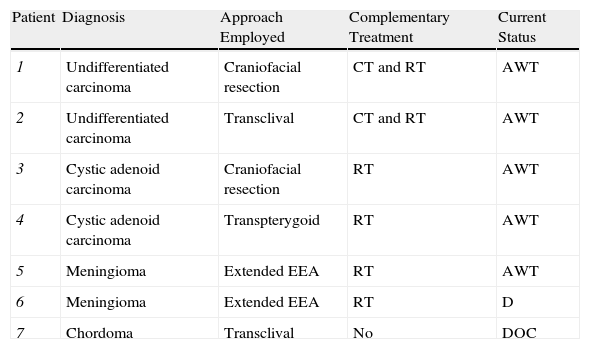
ResultsSixty-three patients were analysed. We performed an endoscopic craniofacial resection in 32 patients (51%), an expanded EEA in 22 (35%), a transclival approach in 6 (9%) and a transpterygoid approach in 3 (5%). The most frequent benign tumour was nasopharyngeal angiofibroma (24%), while adenocarcinoma (30%) was the most common among malignancies. Mean follow-up was 26months (range: 6–84months). The complication rate was 5% and resection was complete in 56 cases (89%). The 5-year overall-survival was 71% in patients with malignant tumours and the effectiveness was 100% in benign tumours.
ConclusionOur results support that endoscopic surgery, when properly planned, represents a valid alternative to standard surgical approaches for the management of skull base tumours.
La progresiva ampliación de las indicaciones de la cirugía endoscópica nasal ha permitido que sea utilizada como vía de abordaje en el tratamiento de tumores que afectan la base del cráneo. Presentamos nuestra experiencia en el tratamiento endoscópico de estos tumores.
Material y métodoSe presenta una serie retrospectiva de los tumores tratados en nuestro servicio entre los años 2004 y 2011 mediante un abordaje endoscópico endonasal (AEE).
ResultadosFueron analizados 63 pacientes. En 32 pacientes (51%) se realizó una resección craneofacial endoscópica, en 22 (35%) se realizó un AEE ampliado, en 6 (9%) un abordaje transclival y en 3 (5%) un abordaje transpterigoideo. El tumour benigno más frecuentemente fue el angiofibroma nasofaríngeo (24%) y el adenocarcinoma (30%) fue el más frecuente entre los malignos. El seguimiento medio fue de 26 meses (rango: 6 a 84 meses). La tasa de complicaciones fue del 5% y la resección fue completa en 56 casos (89%). La supervivencia en los pacientes con tumores malignos fue del 71% a los 5 años y la efectividad fue del 100% en los tumores benignos.
ConclusionesLos resultados obtenidos permiten afirmar que, la cirugía endoscópica ampliada a la base del cráneo, con indicaciones precisas, es una alternativa válida a los tradicionales abordajes abiertos.