Bipolar disorder is related to a high level of personal, familial, social and economic burden. There is a need for feasible adjunctive psychological interventions easy to implement in clinical practice in order to enhance aspects that medication alone cannot achieve. This study aims to evaluate the impact of a 12-session adjunctive integrative program designed for patients with bipolar disorder.
MethodsThis is a single-blind prospective, randomized controlled trial involving a total of 132 outpatients with bipolar disorder who will be recruited from the Hospital Clinic of Barcelona. All participants will be randomly assigned to two arms. All the patients will receive treatment as usual (TAU) but in addition the experimental group will receive an integrative approach consisting of 12-sessions of 90min each in which contents of psychoeducation for patients have been combined with a session for family members, and complemented with aspects related to health promotion, mindfulness training, and strategies for cognitive and functional enhancement. The whole sample will be assessed at baseline, after completion (3-months) and at 12 months from baseline regarding demographic and clinical variables, psychosocial and cognitive functioning, wellbeing and quality of life. The primary outcome measure will be improvement in psychosocial functioning.
ConclusionsIf the integrative approach is effective, it would allow clinicians to cover different areas that may be affected by bipolar disorder, by means of a brief intervention that can therefore be easily generalized to clinical practice.
Trial registrationNCT04031560. Date registered July 24, 2019.
El trastorno bipolar se asocia a un nivel elevado de carga personal, familiar, social y económica. Existe la necesidad de intervenciones psicológicas complementarias factibles y fáciles de implementar en la práctica clínica para mejorar aspectos que la medicación no consigue alcanzar. Este estudio tiene como objetivo evaluar el impacto de un programa integral de 12 sesiones complementario al tratamiento farmacológico para pacientes con trastorno bipolar.
MétodosEnsayo clínico aleatorizado, controlado, a simple ciego. Serán reclutados del Hospital Clínic de Barcelona 132 pacientes ambulatorios diagnosticados de trastorno bipolar. Los participantes serán asignados aleatoriamente a dos grupos. Todos los pacientes recibirán el tratamiento habitual (TAU) pero, además, el grupo experimental recibirá un abordaje integral que consistirá en 12 sesiones de 90min en las que se han combinado los contenidos de psicoeducación para pacientes con una sesión para familiares, complementándose con aspectos relacionados con la promoción de la salud, el entrenamiento en atención plena y estrategias para la potenciación cognitiva y funcional. La muestra completa será evaluada al inicio del estudio, a los 3 meses y a los 12 meses de seguimiento respecto a variables sociodemográficas y clínicas, de funcionamiento psicosocial y cognitivo, bienestar y calidad de vida. El principal resultado esperado será la mejoría en el funcionamiento psicosocial.
ConclusionesSi el abordaje integral es efectivo, permitiría a los clínicos cubrir diferentes áreas que pueden verse afectadas por el trastorno bipolar, mediante una breve intervención que, por lo tanto, puede generalizarse fácilmente a la práctica clínica.
Bipolar disorder (BD) is a chronic and recurrent mental condition characterized by mood fluctuations (that range from hypo/mania to depression) and related to a high level of burden1 and mortality rates.2
Although pharmacological treatment is essential in the clinical management of BD,3 adjunctive psychological interventions also play a crucial role to enhance its effect and cover aspects that medication alone cannot reach.4 One issue that has probably contributed to this fact is that a significant proportion of patients with BD present a poor therapeutic adherence.5 Even with maintenance treatment after an index episode survival analyses have shown a 44% risk of having a recurrence within the first year and 70% within the first 5 years.6 Even during remission periods many patients still complain about persistent mood residual symptoms,7 cognitive impairment,8 comorbidities,9 difficulties in psychosocial functioning,10 poor wellbeing and quality of life.11 All these factors combined with the significant role that stress plays as a trigger for mood relapses12 and the impact of the illness on family members,13 justify the introduction of comprehensive psychological approaches that complement pharmacotherapy.
Most psychological interventions designed for BD share different components, but may vary in the emphasis given to them. Main outcomes also differ, with most psychological treatments focusing on the prophylaxis of mood recurrences and few interventions attending other significant aspects such as psychosocial functioning, wellbeing and quality of life.10 Another important concern is that there are understudied subpopulations (e.g. BD type II, patients with psychiatric comorbidities, with subsyndromal symptoms, among others), because they often fulfill exclusion criteria in clinical trials. Furthermore, the duration of therapies is usually around 20 sessions, especially for those psychological treatments that have been proved to be more effective in BD4 such as psychoeducation, family intervention, cognitive-behavioral therapy, interpersonal and social rhythm therapy, and recently, functional remediation.
Consequently, there is a need for a briefer and integrative intervention which combines the main components of different treatments in order to be simpler and more generalizable, covering wider therapeutic outcomes and improving the prognosis of BD. These characteristics and its consequent lower cost may help to generalize intervention programs developed in specialized centers. Similarly, the inclusion criteria of the patients to whom the intervention will be offered are less restrictive than those of previous clinical trials, thus it would be possible to provide the treatment to a larger number of subjects which would be more representative of the population with BD.
Based on this background, we have recently developed an adjunctive integrative approach that consists of 12, 90-min weekly group sessions for patients with BD.14 This comprehensive and integrative approach combines therapeutic components of broader treatments that the Barcelona Bipolar and Depressive Disorders Unit had previously developed and whose effectiveness has been proved separately, such as group psychoeducation,15,16 family intervention17,18 and functional remediation.19,20 In addition, emphasis in physical health is given and a module of mindfulness training inspired by Mindfulness-Based Cognitive Therapy,21 an adaptation of Mindfulness-Based Stress Reduction,22 has been incorporated since some evidence suggests the benefits of mindfulness on depressive and anxiety symptoms23,24 which are common in BD and negatively affect prognosis.9,25
This project aims to evaluate the efficacy of an adjunctive 12-session integrative program for BD. The primary outcome measure will be improvement in psychosocial functioning. Reduction in number of relapses and hospitalizations as well as in depressive, manic and anxiety symptoms, and an improvement of wellbeing, quality of life, and cognitive performance were considered as secondary outcomes. As a tertiary aim, we will try to identify the predictors of the response to the program. We hypothesized that patients belonging to the experimental arm would have better psychosocial functioning, better well-being and better quality of life. They would also show a better course of the disease, in terms of relapses and hospitalizations, a reduction in symptomatology and better neuropsychological performance compared to the patients included in the control group. It will be possible to characterize the subgroup of patients with a good response to the intervention based on the variables collected.
MethodsDesignThis is a single-blind randomized controlled trial of an integrative approach plus treatment as usual (TAU) versus TAU alone. The study will take place in the Barcelona Bipolar and Depressive Disorders Unit at Hospital Clínic. It will be conducted in accordance with the ethical principles of the Declaration of Helsinki and Good Clinical Practice and has been approved by the Hospital Clinic Ethics and the Research Board (HCB/2017/0432), and has been registered in clinical trials. All participants will be asked to provide written informed consent prior to their inclusion in the study NCT04031560.
ParticipantsOne hundred and thirty two subjects will be recruited (66 in the intervention group and 66 in the control group) from the Barcelona Bipolar and Depressive Disorders Unit at the Hospital Clinic of Barcelona. The main variable used to calculate the sample size was the change in psychosocial functioning, which will be measured by means of the Functioning Short Assessment Test (FAST). Validation and reliability data from the FAST scale, which correlated with the reference functionality scale (GAF), were used for patients presenting with BD, obtaining a mean±SD of 18.55±13.19 points in FAST. Based on these data and considering a significance of α=5% and a statistical power of (1−β) of 90%, we expect to find an improvement in the experimental group versus the control group of 7.8 points on the FAST scale (range 0–72 divided into 24 items). Assuming the possibility of a 10% drop-out rate, 66 patients are needed per branch of intervention.
The sample will have to fulfill the following inclusion criteria: ages from 18 to 60 years, diagnosis of BD type I or II according to DSM-526 criteria, euthymic or with subthreshold symptoms (Hamilton Depression Rating Scale27 <14; Young Mania Rating Scale28 <8) and absence of an acute mood episode in the 3 months prior to the inclusion in the study. Exclusion criteria will be: estimated Intelligence Quotient (IQ) lower than 85, electroconvulsive therapy in the past six months, significant physical or neurologic illness that can affect neuropsychological performance, diagnosis of substance use disorder (SUD) according to DSM-526 criteria (except for tobacco), inability to understand the purposes of the study and absence of participation in the following psychosocial interventions in the past 2 years (group psychoeducation, functional remediation or mindfulness-based interventions).
ProcedureDuring their follow-up appointments at the clinic, all patients who might be potential candidates will be informed about the availability of the study. If they show interest, verbal and written information will be provided and any questions or concerns will be clarified. If the patients confirm their willingness to participate, they will be required to read and sign an informed consent. Participants will be evaluated concerning demographic, clinical variables, neuropsychological performance, psychosocial functioning, wellbeing, quality of life and the rest of the variables detailed below. Subsequently, patients will be randomly assigned (1:1) to two different groups by means of computer-generated random numbers. The experimental group (66 patients) will take part in the integrative program as a complement to pharmacological treatment, whilst the control group (66 patients) will only receive TAU, just usual standard pharmacological treatment without additional group sessions. Psychiatric medication will be recorded during the study and the reasons for modifications will be registered. Subject withdrawal will be considered when they voluntarily discontinue participation, when patients in the experimental group do not attend at least 8 intervention sessions or if patients start an acute episode according to DSM-5 criteria or require psychiatric hospitalization during the group.
Together with the baseline assessment, the whole sample will be re-assessed with all the instruments at 3-months (after the intervention) and at 12 months from the baseline by researchers blinded to the treatment condition. Due to the time required for the evaluation, it will be divided in two days: a first appointment for the collection of sociodemographic information, clinical data (symptoms, recurrences, hospitalizations), psychosocial functioning, quality of life and the rest of variables; and a second one for the neuropsychological evaluation.
Variables and instrumentsA semi-structured interview of the Program's protocol based on the Structured Clinical Interview for DSM26 will be conducted and complemented with clinical record reviews and assessment instruments in order to collect the following variables: gender, age, marital status, educational status and occupation, age at onset of BD, age at first hospitalization, total number and type of previous episodes, number of hospitalizations, history of psychotic symptoms, polarity of the first episode, seasonal pattern, presence of rapid cycling, bipolar subtype (I or II), predominant polarity, psychiatric and physical comorbidities, family psychiatric history, family history of affective disorder, family history of completed suicide, history of suicidal ideation and suicidal behavior, number of suicide attempts, and history of drug misuse. During each assessment, questions will be asked about substance use (type, amount and frequency).
Clinical features will be measured with the 17-item Hamilton Depression Rating Scale (HDRS)27 for the depressive symptomatology and Young Mania Rating Scale (YMRS)28 for manic symptoms. HDRS consists of 17 items; each of them has between three and five possible answers, with a score of 0–2 or 0–4 respectively. The total score ranges from 0 to 52, with higher scores representing more severe depressive symptoms. On the other hand, YMRS consists of 11 items; seven of them are ranked on a scale of 0–4 and four on a scale of 0–8. Total scores range from 0 to 60, with higher scores indicating more severe manic symptoms. The severity of anxiety symptoms will be measured by the Hamilton Anxiety Rating Scale (HAM-A).29 This scale consists of 14 items, each of them defined by a series of symptoms, and measures both psychic anxiety (mental agitation and psychological distress) and somatic anxiety (physical complaints related to anxiety). Each item is scored on a scale of 0–4, with a total score ranging from 0 to 56, where higher scores represent more severe anxiety.
Psychosocial functioning will be assessed through the Functioning Assessment Short Test (FAST).30 The FAST is an interviewer-administered instrument developed to assess the main difficulties in daily life of patients with BD. It is a 24-item scale that provides a total score and also 6 specific subdomain scores (autonomy, occupational functioning, cognitive functioning, financial issues, interpersonal relationships, and leisure time). The overall FAST score ranges from 0 to 72, higher scores indicating greater disability, with a threshold score above 11 representing significant disability.
Affective temperaments will be assessed by means of the Barcelona version of the Temperament Evaluation of the Memphis, Pisa, Paris, and San Diego (TEMPS-A), recently validated in the Spanish population.31 This self-applied instrument consists of 110 items which require a true/false response and assess the presence of five affective temperaments, namely: depressive (items 1–21), cyclothymic (items 22–42), hyperthymic (items 43–63), irritable (items 64–84) and anxious (items 85–110).
Trait impulsivity will be rated through the Barratt Impulsiveness Scale (BIS-11).32 This self-rated questionnaire incorporates three subscales assessing three components of impulsivity: attentional/cognitive, which assess tolerance to complexity and attentional persistence; motor, which explores the tendency to act on the spur of the moment, and non-planning, which measures the lack of future orientation. The 30 items are rated from 1 (rarely/never) to 4 (always). Total score range from 30–120 and greater scores are indicators of a higher level of impulsivity.
Biological rhythms interview of assessment in neuropsychiatry (BRIAN)33 will be used to evaluate problems related to the maintenance of circadian rhythm regularity. The 18 items of the scale are divided into 4 specific areas: sleep, activities, social rhythm and eating pattern. All items are scored from 1 (no difficulties) to 4 (serious difficulties). The total BRIAN scores range from 1 to 72, where higher scores suggest severe circadian rhythm disturbances.
Adherence will be evaluated by an adherence-focused interview with the patient combined with information from their medical records, in collaboration with the patient's psychiatrist if needed. Lithium and valproic acid blood levels will be also collected, and psychiatric medication and reasons for changes will be recorded during the study.
Penn State worry Questionnaire-11 (PSWQ-11)34 will be used to evaluate the worry severity. The scale includes a direct 11 Likert-type items (ranging from “nothing” to “a lot” with scores ranging from 1 to 5), to assess a general tendency to worry or worry-trait. Total score range from 16–80 with the algorithm of total scores: low worry (16–39), moderate worry (40–59), and high worry (60–80).
Subjective cognitive deficits will be assessed through Cognitive Complaints in Bipolar Disorder Rating Assessment (COBRA).35 This self-reporting rating scale consists of the assessment of subjective cognitive impairment in daily activities experienced by patients with BD. The COBRA scale includes 16 items that allow patients to report assessments of subjective cognitive dysfunction related to executive function, processing speed, working memory, verbal learning and memory, attention and concentration, and mental tracking. All items are rated using a 4-point scale: 0=never, 1=sometimes, 2=often, and 3=always. A total COBRA score will be calculated by adding the scores of each item, with higher scores reflecting higher levels of subjective cognitive deficits.
Wellbeing will be assessed through the Spanish version of the WHO (Five) Well-being Index (WHO-5)36; a self-administered scale that contains five positively phrased Likert-type items. The respondent is asked to rate how well each of the five statements applies to him or her over the last two weeks. Each item is rated on a six-point Likert scale ranging from 5 (all of the time) to 0 (none of the time). The scores are summed to obtain a total raw score ranging from 0 to 25 (0 is absence of well-being and 25 is maximal well-being). The instructions for the scale recommend multiplying the raw score by 4, hence the highest possible score is 100 on this scale.
Quality of life was assessed with the Quality of Life in Bipolar Disorder scale (QoL.BD),37 the first quality of life questionnaire specifically designed for people with BD. This measure was developed specifically by assessing 12 factor-analytically derived domains (Physical, Sleep, Mood, Cognition, Leisure, Social, Spirituality, Finance, Household, Self-esteem, Independence, and Identity) and two optional domains (Work and Education) from the parent instrument (the 56-item Qol.BD). This scale is characterized by a response format (5-point likert strongly disagree-strongly agree) in which higher scores indicate greater quality of life.
Mindfulness capacity will be assessed by the Five Facet Mindfulness Questionnaire (FFMQ)38 and the Mindful Attention Awareness Scale (MAAS).39 FFMQ is a 39-item questionnaire scored on a Likert scale ranging from 1 (never or rarely true) to 5 (very often or always true). It evaluates 5 facets: (1) observation: ability to notice internal and external experiences such as sensations, emotions or thoughts; (2) description: ability to label experiences with words; (3) conscious action: ability to act consciously and not be on autopilot mode; (4) do not judge internal experience: capacity for non-evaluative attitude toward thoughts and emotions; (5) non-reactivity to internal experience: ability to allow the free flow of thoughts and emotions without being trapped by them or rejecting them. MAAS evaluates in a global way the capacity of an individual to be attentive and aware of the experience of the present moment in daily life. The questionnaire consists of 15 items and is scored according to a Likert scale, which ranges from 1 (almost always) to 6 (almost never). High scores indicate better mindfulness skills.
Finally, a self-administered questionnaire of 31 items of Knowledge of Bipolar Disorder (available upon request) will be measured.
Neurocognitive performance will be evaluated through a comprehensive neuropsychological battery that lasts approximately 180min and is composed of different tasks assessing seven cognitive domains: (1) Estimated premorbid IQ, which will be evaluated with the Wechsler Adult Intelligence Scale version III (WAIS-III) vocabulary subtest.40 (2) The processing speed index, which consists of two subtests of the WAIS-III, the digit-symbol coding and symbol search.40 (3) Executive function, which will be tested by set shifting, planning, and response inhibition using the Computerized Wisconsin Card Sorting Test (WCST),41 the Stroop Color-Word Interference Test,42 the phonemic (F-A-S) and categorical (animal naming) components of the Controlled Oral Word Association Test,43 and the Trail Making Test, part B (TMT-B).44 (4) Visual memory and verbal learning/memory, which will be assessed with the Rey-Osterrieth Complex Figure for visual memory,45 and the California Verbal Learning Test (CVLT)46 for verbal memory. (5) The working memory index, which will be tested with three subtests of the WAIS-III40 Arithmetic, Digits, and Letter-number sequencing. (6) Attention, which will tested with the Continuous Performance Test-II, version 5 and the Trail Making Test, part A (TMT-A).47 (7) Social cognition which will be assessed through Mayer-Salovey-Caruso Emotional Intelligence Test (MSCEIT),48 Reading the Mind in the Eyes Test (RMET),49 Hinting Task,50 Ambiguous Intentions Hostility Questionnaire (AIHQ)51 and the Iowa Gambling Test (IGT).52
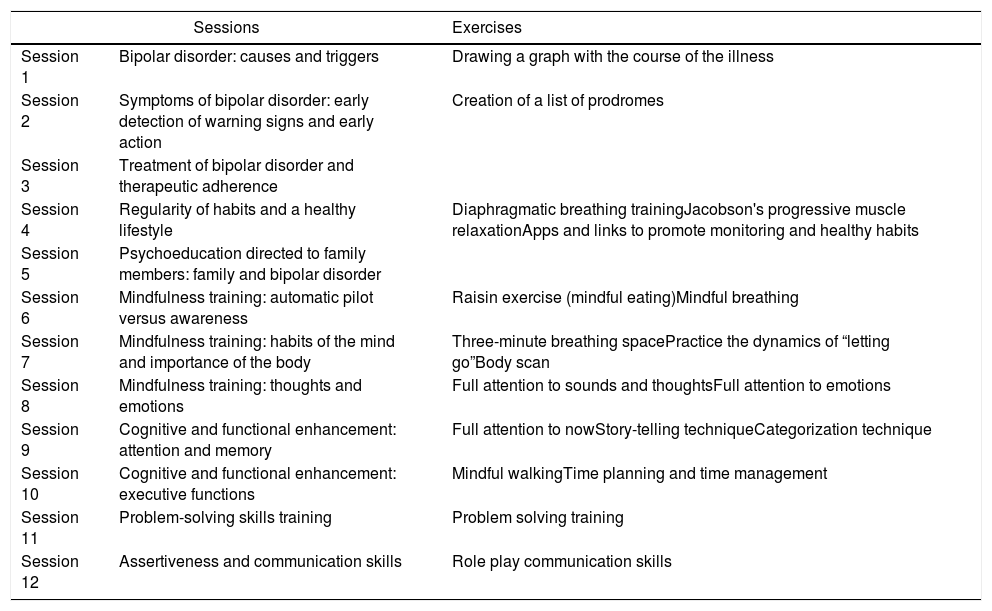
InterventionsThe adjunctive integrative approach will consist of 12 weekly group sessions, each one lasting 90min, over a period of 3 months provided in the outpatient clinic plus TAU. Six intervention groups with 11 patients in each will be conducted. As mentioned in the introduction, the integrative approach incorporates, among others, therapeutic components of broader psychological approaches that the Barcelona Bipolar and Depressive Disorders Unit has previously developed and whose efficacy has been evaluated separately. Therefore, some contents of psychoeducation for patients have been combined with a session for family members only (in which a maximum of two relatives per patient will be admitted, without patients being present), and complemented with aspects related to health promotion, mindfulness training, and strategies for cognitive and functional enhancement. The foundations of the approach and the contents of each session have been extensively explained in Reinares et al. (2019).14Table 1 summarizes the sessions of the program. The groups will be conducted by two psychologists with clinical experience in the management of BD, group dynamics and with the different components of the treatment. The sessions will be structured by dedicating the first few minutes to holding an informal conversation in which possible incidents and doubts that may have arisen since the last encounter are commented on, practical exercises are reviewed or the mindfulness practice introduced (from the first session of this module onwards). Subsequently, the topic of the day will be addressed, covering the main objectives of each session with flexibility while encouraging participation and discussion. The formulation of questions that guide the contents, rounds and the use of exercises will encourage the participation of every member of the group. After each session, a summary of the topic worked on will be delivered. Each thematic block will be complemented with additional information in the form of mobile applications, links, audios, weekly practice logs, and literature so that members can go deeper into the practice of the implemented components. In contrast, the TAU group will not receive any type of add-on psychotherapy, just usual standard psychiatric treatment.
Session program of the integrative approach.
| Sessions | Exercises | |
|---|---|---|
| Session 1 | Bipolar disorder: causes and triggers | Drawing a graph with the course of the illness |
| Session 2 | Symptoms of bipolar disorder: early detection of warning signs and early action | Creation of a list of prodromes |
| Session 3 | Treatment of bipolar disorder and therapeutic adherence | |
| Session 4 | Regularity of habits and a healthy lifestyle | Diaphragmatic breathing trainingJacobson's progressive muscle relaxationApps and links to promote monitoring and healthy habits |
| Session 5 | Psychoeducation directed to family members: family and bipolar disorder | |
| Session 6 | Mindfulness training: automatic pilot versus awareness | Raisin exercise (mindful eating)Mindful breathing |
| Session 7 | Mindfulness training: habits of the mind and importance of the body | Three-minute breathing spacePractice the dynamics of “letting go”Body scan |
| Session 8 | Mindfulness training: thoughts and emotions | Full attention to sounds and thoughtsFull attention to emotions |
| Session 9 | Cognitive and functional enhancement: attention and memory | Full attention to nowStory-telling techniqueCategorization technique |
| Session 10 | Cognitive and functional enhancement: executive functions | Mindful walkingTime planning and time management |
| Session 11 | Problem-solving skills training | Problem solving training |
| Session 12 | Assertiveness and communication skills | Role play communication skills |
Baseline distribution descriptive analysis of the variables will be carried out. Both groups will be compared at baseline and during the follow-up (3 and 12 months). Repeated-measures ANOVA will be used to compare the two arms (integrative treatment versus TAU) on the primary and secondary outcomes collected in different evaluations over time. Effect sizes will be calculated to quantify the effect of the intervention between groups. Multivariate regression models will be used to establish the relationship between the treatment response and the variables collected. Statistical analyses will be conducted using the software Statistical Package for Social Sciences version 23.0. The statistical level of significance will be set at α=0.05.
DiscussionTo our knowledge this is the first study evaluating an adjunctive psychological treatment integrating different evidence-based approaches in a single brief comprehensive program. Considering the chronicity and high disability associated with BD, it is crucial to identify and to implement effective and affordable strategies. Due to the long length of previous evidence-based psychological treatments, shorter adaptations have sometimes been used but often not rigorously tested. This 12-session intervention would, therefore, cover a clinical need using a rigorous methodology.
Although main outcomes vary between studies, in general, adjunctive psychological treatments have focused on the prevention of recurrences and hospitalizations.4 However, the improvement of other clinical variables such as subsyndromal symptoms, and the reduction of psychosocial impairment and cognitive deficits, as well as the enhancement of wellbeing and quality of life, which are crucial for the subjects suffering from the illness, have hardly been studied.10 Some treatments have been designed to address specific targets, i.e. cognition53 and psychosocial functioning,19,20 but there is a lack of treatments aimed at integrating different therapeutic approaches to cover varied aims. It is worth mentioning some multi-component or care packages studies that have shown a reduction in manic symptoms54,55 and an improvement in social role function and quality of life.55 These works not only differ from our proposed package in the length of the intervention (2 years) but also in the contents (which consisted of a structured group psychoeducational program, monthly telephone monitoring of mood symptoms and medication adherence, feedback to treating mental health providers, facilitation of appropriate follow-up care, and as-needed outreach and crisis intervention). An improvement in symptoms and psychosocial functioning was also observed with 20 sessions of psychoeducation together with cognitive-behavioral therapy.56 A randomized clinical trial to assess the efficacy of a group intervention combining acceptance and commitment therapy and psychoeducation approaches, which primary outcome is wellbeing, is ongoing.57
An integrative intervention such as the one we want to test,14 would allow us to encompass different needs and outcomes by selecting the main components of distinct interventions previously tested with the aim of having a shorter but potentially effective adjunctive approach to BD. The psychological treatments available to date have also focused on study populations that met very restrictive inclusion criteria. In contrast, the integrative approach is aimed at people with BD in pharmacological treatment who are stable but also for patients with subsyndromic symptoms, which is the rule rather than the exception in BD. For this program, people with other associated psychiatric illnesses (anxiety disorder, etc.) will not be excluded, with the exception of active drug abuse/dependence.
As there is a need to generalize intervention programs developed in specialized centers to daily practice, this integrative package has the distinctive feature of being sufficiently brief and feasible to be implemented easily in the regular practice, reaching a large amount of people who otherwise would not have access to evidence-based interventions thus maximizing healthcare effects.
The implementation of adjunctive psychosocial strategies may help to change the outcome of the illness and avert potentially irreversible harm to patients with BD. Our proposal would considerably improve the scope of the therapeutic approaches that already exist for the management of the illness, assuming there would be better care for this population. In addition, if we can identify response predictors to the treatment, it will be an important contribution for future research and clinical practice. This would allow the clinician to consider individual variables and personal needs when the treatment is selected or to modify the treatment based on the characteristics of the population if needed, as recent studies have shown.58
There are some potential limitations of this project such as the lack of a third condition arm as an active control treatment. Another limitation will be the difficulty to control the pharmacological treatments that patients will receive although any changes in medication will be registered and taken into account in the statistical analysis. In addition, the estimation of dropouts could be higher than expected; however, a sample size of 132 patients (66 patients in each branch) is similar or even higher than the sample size used in most studies of psychological treatments in bipolar disorder. Finally, the sample will be recruited from a specialized mental healthcare center in BD, which could limit the generalizability of the results.
Despite these potential limitations, if the intervention that we propose is effective, it would represent a useful comprehensive approach that could be generalized beyond specialized centers and implemented in services with fewer resources. At the same time, it could be offered to a larger number of patients, responding to a clinical, social and economic demand, thus optimizing socio-health resources with a clear clinical applicability and a significant improvement for people living with BD and their caregivers. This study could also shed some light on the length and intensity of psychological treatments, which may vary among patients, and is still a matter of debate.
ConclusionThis study aims to evaluate a brief group intervention based on the combination of contents from different programs that have previously shown an improvement for patients with BD, expanded with other elements in order to achieve wider therapeutic targets. This could translate into a clear advantage for the management of the illness, considering the elevated global burden of this disease. If the integrative approach proves to be useful, it would be an innovative, cost-effective adjunctive tool for BD, being short and feasible enough to be implemented in centers with fewer resources and benefiting patients who would otherwise not have access to evidence-based interventions, therefore maximizing healthcare effects and reducing suffering.
Ethics approval and consent to participateThis study is approved by Hospital Clinic Ethics and Research Board (Comité Ético de Investigación Clínica: CEIm) from the Hospital Clínic Barcelona, project number HCB/2017/0432. Protocol version 1, 10th May 2017. This study will be conducted in accordance with the ethical principles of the Declaration of Helsinki and Good Clinical Practice.
Consent for publicationAll participants will be asked to provide written informed consent prior to their inclusion in the study.
Availability of data and materialsThe investigators are currently collecting data. This is an ongoing study. Hence, preliminary results have not yet been published.
The datasets used and/or analyzed during the current study will be available from the corresponding author on reasonable request.
FundingThis study has been funded by Instituto de Salud Carlos III through the project PI17/00941.
Conflict of interestThe authors declare that they have no competing interests related to this manuscript.
This work was supported by the Spanish grant FIS PI17/00941 from Instituto de Salud Carlos III. The authors would like to thank the support of the Spanish Ministry of Science, Universities and Innovation, Instituto Carlos III integrated into the Plan Nacional de I+D+I (cofounded by European Regional Development Fund/European Social Fund) “investing in your future”, the CIBER of Mental Health (CIBERSAM); the Comissionat per a Universitats I Recerca del DIUE de la Generalitat de Catalunya to the Bipolar Disorders Group (2017 SGR 1365), the CERCA Programme/Generalitat de Catalunya, and Instituto de Salud Carlos III for funding through project PI17/0094. Dr. Bonnín would like to thank de Departament de Salut de la Generalitat de Catalunya for the PERIS grant (SLT002/16/00331).