Few studies have investigated the impacts of metabolic syndrome (MS) on coronavirus disease 2019 (COVID-19). We described the clinical features and prognosis of confirmed COVID-19 patients with MS during hospitalization and after discharge.
MethodsTwo hundred and thirty-three COVID-19 patients from the hospitals in 8 cities of Jiangsu, China were retrospectively included. Clinical characteristics of COVID-19 patients were described and risk factors of severe illness were analyzed by logistic regression analysis.
ResultsForty-five (19.3%) of 233 COVID-19 patients had MS. The median age of COVID-19 patients with MS was significantly higher than non-MS patients (53.0 years vs. 46.0 years, P=0.004). There were no significant differences of clinical symptoms, abnormal chest CT images, and treatment drugs between two groups. More patients with MS had severe illness (33.3% vs. 6.4%, P<0.001) and critical illness (4.4% vs. 0.5%, P=0.037) than non-MS patients. The proportions of respiratory failure and acute respiratory distress syndrome in MS patients were also higher than non-MS patients during hospitalization. Multivariate analysis showed that concurrent MS (odds ratio [OR] 7.668, 95% confidence interval [CI] 3.062–19.201, P<0.001) and lymphopenia (OR 3.315, 95% CI 1.306–8.411, P=0.012) were independent risk factors of severe illness of COVID-19. At a median follow-up of 28 days after discharge, bilateral pneumonia was found in 95.2% of MS patients, while only 54.7% of non-MS patients presented bilateral pneumonia.
Conclusions19.3% of COVID-19 patients had MS in our study. COVID-19 patients with MS are more likely to develop severe complications and have worse prognosis. More attention should be paid to COVID-19 patients with MS.
Pocos estudios han investigado el impacto del síndrome metabólico (SM) en la enfermedad por coronavirus 2019 (COVID-19). Describimos las características clínicas y el pronóstico de los pacientes con COVID-19 confirmados con SM durante la hospitalización y después del alta.
MétodosSe incluyó de forma retrospectiva a 233 pacientes con COVID-19 de los hospitales de 8 ciudades de Jiangsu (China). Se describieron sus características clínicas y se analizaron los factores de riesgo de enfermedad grave mediante un análisis de regresión logística.
ResultadosDe los 233 pacientes, 45 (19,3%) tenían EM. La mediana de edad de estos pacientes con EM fue significativamente mayor que la de los pacientes sin él (53,0 años frente a 46,0 años; p = 0,004). No hubo diferencias significativas en cuanto a los síntomas clínicos, las imágenes de TC torácica anormales y los fármacos de tratamiento entre los 2 grupos. Hubo más pacientes con EM que tuvieron enfermedades graves (33,3% frente a 6,4%; p < 0,001) y críticas (4,4% frente a 0,5%; p = 0,037) que los pacientes sin EM. Las proporciones de insuficiencia respiratoria y síndrome de dificultad respiratoria aguda en los pacientes con EM también fueron mayores que en los pacientes sin EM durante la hospitalización. El análisis multivariante mostró que la EM concurrente (odds ratio [OR] 7,668; intervalo de confianza [IC] del 95%: 3,062-19,201; p < 0,001) y la linfopenia (OR 3,315; IC del 95%: 1,306-8,411; p = 0,012) eran factores de riesgo independientes de COVID-19 grave. En una mediana de seguimiento de 28 días tras el alta, se encontró neumonía bilateral en el 95,2% de los pacientes con EM, mientras que solo la presentaron el 54,7% de los pacientes sin EM.
ConclusionesEl 19,3% de los pacientes con COVID-19 tenían EM en nuestro estudio. Los pacientes con COVID-19 y EM son más propensos a desarrollar complicaciones graves y tienen peor pronóstico. Se debe prestar más atención a los pacientes con COVID-19 y EM.
Coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is rapidly spreading all over the world.1 As of February 24, 2021, there were 111,762,965 confirmed cases of COVID-19 resulting in 2,479,678 deaths globally.1 However, the severity and prognosis of COVID-19 differ across countries and regions according to previous reports.2–4
A growing body of evidence suggested that elderly patients and patients with comorbidities presented poor outcomes.2,3 More patients with hypertension (23.7% vs. 13.3%, P<0.001) and type 2 diabetes (16.2% vs. 5.7%, P<0.001) developed severe illness than non-hypertension and non-diabetes patients in a large retrospective study from China.2 A retrospective study by matching age and gender found that COVID-19 patients with diabetes had worse outcomes compared to patients without diabetes.5 Wang et al. reported that patients admitted to intensive care units (ICU) presented higher proportions of hypertension and type 2 diabetes compared to non-ICU patients.4 Another study also reported that the proportion of hypertension in deceased patients was significantly higher than recovered patients.3 These results suggested that metabolic factors may be associated with prognosis of COVID-19 patients.
Metabolic syndrome (MS) consists of five determining factors, including obesity, elevated blood pressure, increased triglycerides and cholesterol, and impaired glucose tolerance.6,7 MS is generally regarded as a risk factor for progression of cardiovascular disease and type 2 diabetes.7 However, the impacts of MS on COVID-19 remain unclear. This study aimed to describe the clinical features and prognosis of confirmed COVID-19 patients with MS during hospitalization and after discharge in a multicenter cohort of COVID-19 patients in Jiangsu province, China.
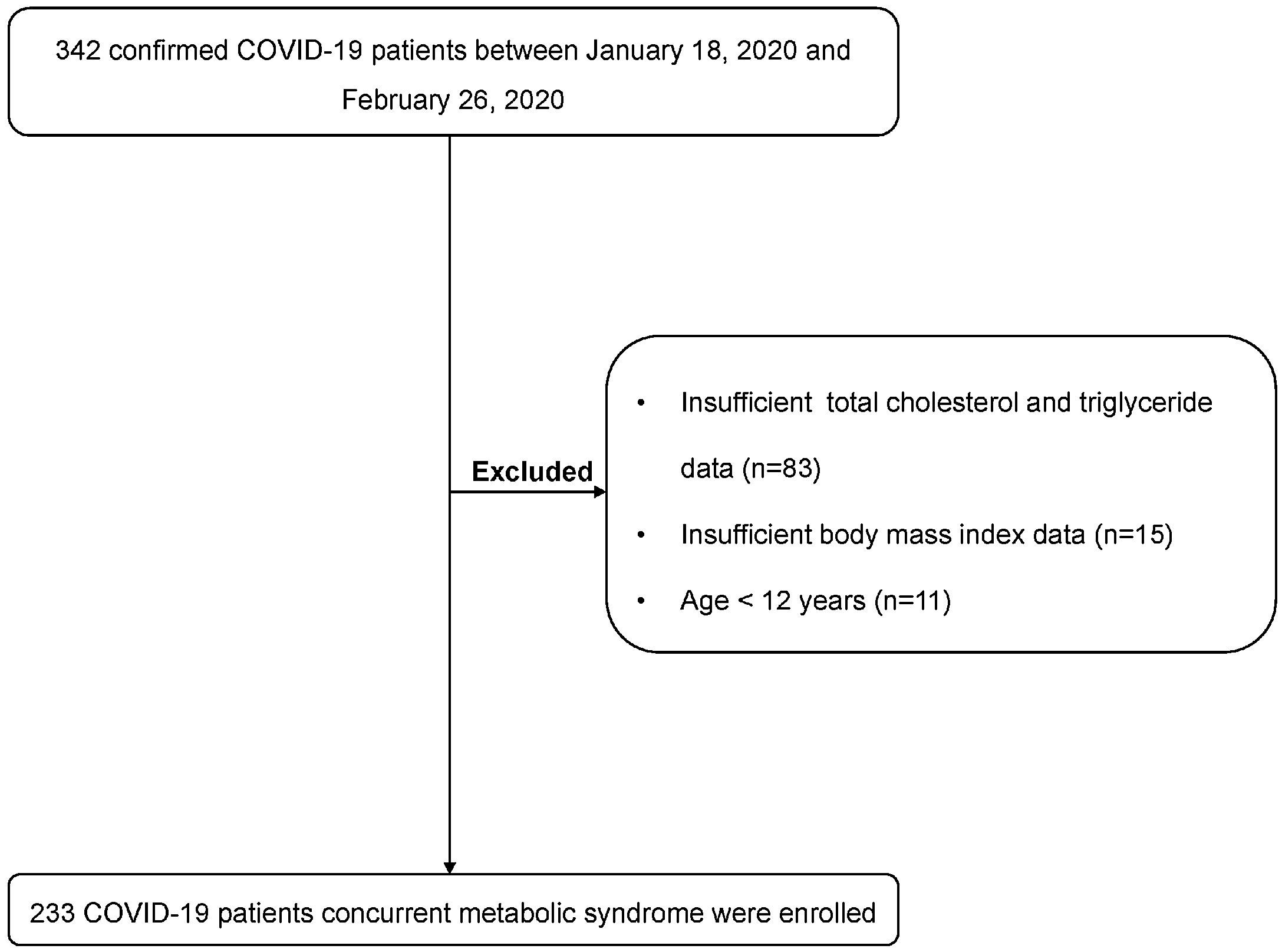
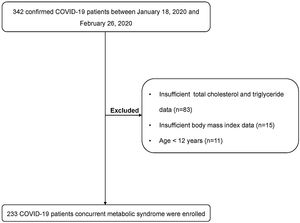
MethodsPatientsThe clinical data of 342 confirmed COVID-19 patients form 10 hospitals in 10 cites in Jiangsu, China between January 18, 2020 and February 26, 2020 were retrospectively collected and reviewed. Confirmed patients were diagnosed based on the criterion of World Health Organization (WHO) interim guidance.8 The diagnostic criteria of acute respiratory distress syndrome (ARDS) was based on previous study.9 To reduce the impact of potential factors, patients under 12 years and pregnant women were excluded.10 In addition, patients with unavailable data of body mass index (BMI), blood pressure, triglycerides (TG), total cholesterol (TC), and fasting blood glucose (FBG) were also excluded. The study was approved by the Ethics Committee of these hospitals and the written informed consent was waived.
ProceduresThe medical records of patients were reviewed by health care workers in each medical center. The characteristics of epidemiology, laboratory, radiology, treatment, and prognosis were collected from medical records. Routine physical examination, including height, weight, systolic pressure and diastolic pressure, were measured on admission. The computational formula of BMI was weight (kg) divided by the square of height (m).7 All data was entered in a computerized database and checked by different researchers for further analysis.
DefinitionsCentral obesity (waist circumference ≥90cm in male or ≥80cm in female) is one of the diagnostic criteria of MS.7 However, waist circumference data were not available in our study. The diagnosis of obesity was based on BMI index.11–13 The diagnostic criterions of metabolic syndrome were as follows according previous studies: (1) Obesity, BMI≥28kg/m2; (2) TG, >150mg/dL or use of triglyceride lowering medication; (3) TC, >200mg/dL or use of cholesterol lowering medication; (4) Blood pressure, systolic pressure>130mmHg and/or diastolic pressure>85mmHg or use of blood pressure lowering medication; (5) FBG, >5.6mmol/L or use of diabetes medication.6,7,11,14 The presence of any three of the above five criterions was considered as metabolic syndrome. In addition, previous study reported that a determinant effect of age>50 years on prognosis of COVID-19 patients.15 Therefore, the age of 50 years was used as a threshold to analyze the association between age and severe illness in our study. Severe illness of COVID-19 was defined according to the current guideline as follows: (1) respiratory frequency ≥30/min, (2) pulse oximeter oxygen saturation≤93% at rest, (3) oxygenation index≤300mmHg.16 Critical illness of COVID-19 was defined as follows: (1) respiratory failure and requiring mechanical ventilation, (2) shock, (3) with other organ failure that requires ICU care.16 The poor prognosis was defined as developed respiratory failure, ARDS, severe illness, critical illness, or admission to ICU during hospitalization in this study.
Follow-upCOVID-19 patients were followed up for 3–6 weeks after discharge. SARS-CoV-2 nucleic acid in throat swab samples, blood routine examination, biochemical examination, and chest CT were tested during follow-up. Additionally, symptoms of patients were also recorded.
Statistical analysisContinuous variables were described as means (standard deviations) or medians (interquartile range (IQR)). Categorical variables were showed as the counts and percentages. Two-sample t tests or Mann–Whitney U were used for continuous variables, and Chi-square tests or Fisher's exact tests were used to compare the categorical variables. Binary logistic regression was used to analyze the risk factors of severe illness. Variables with P values<0.05 in the univariate analysis were further entered into a multivariate logistic regression analysis. P<0.05 was considered as statistically significant. Age, gender, smoking, lymphopenia and leukopenia were reported to be associated with the severity of COVID-19.17 Therefore, these variables were also adjusted in the multivariate logistic regression. SPSS version 22.0 software (SPSS Inc., Chicago, IL, United States) was used for the data analysis.
ResultsClinical characteristics of patients with COVID-19The flow diagram of the enrolled patients is presented in Fig. 1. 109 patients were excluded. Two hundred and thirty-three patients were included for the final analysis. The median age of patients was 47.0 (IQR 35.0–57.0) years and 87 (37.3%) of patients were over 50 years. 56.7% patients were male. The median BMI of patients was 24.4 (IQR 22.5–26.6) kg/m2 and 14.6% patients were obese. The median of systolic pressure and diastolic pressure were 128 (IQR 119–136) mmHg and 83.0 (IQR 76.0–89.5) mmHg, respectively. Thirty-eight (16.3%) patients had a history of hypertension, and 18 (7.7%) patients had a history of type 2 diabetes on admission, respectively. Other comorbidities included chronic liver diseases (15 [6.4%]), chronic lung diseases (10 [4.3%]), cardiovascular diseases (5 [2.1%]), and cerebrovascular diseases (3 [1.3%]). 54.9% patients had a contact history with suspected or confirmed patients. The median time from symptom onset to admission was 5.0 (IQR 2.0–9.0) days (Table 1).
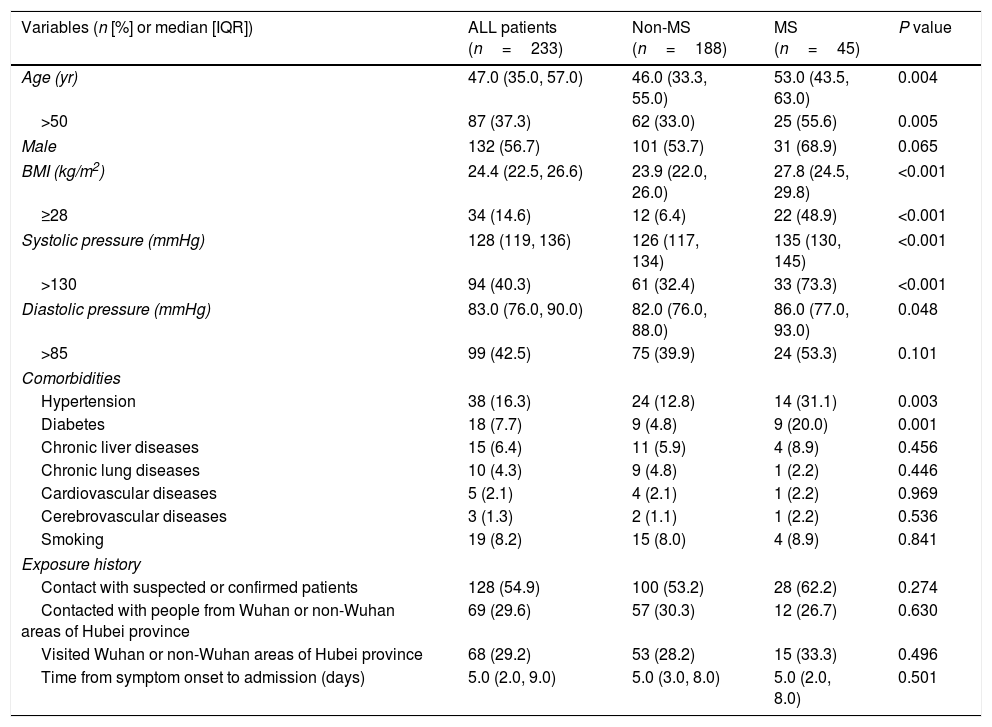
Demographic and epidemiologic characteristics of COVID-19 patients with and without metabolic syndrome.
| Variables (n [%] or median [IQR]) | ALL patients (n=233) | Non-MS (n=188) | MS (n=45) | P value |
|---|---|---|---|---|
| Age (yr) | 47.0 (35.0, 57.0) | 46.0 (33.3, 55.0) | 53.0 (43.5, 63.0) | 0.004 |
| >50 | 87 (37.3) | 62 (33.0) | 25 (55.6) | 0.005 |
| Male | 132 (56.7) | 101 (53.7) | 31 (68.9) | 0.065 |
| BMI (kg/m2) | 24.4 (22.5, 26.6) | 23.9 (22.0, 26.0) | 27.8 (24.5, 29.8) | <0.001 |
| ≥28 | 34 (14.6) | 12 (6.4) | 22 (48.9) | <0.001 |
| Systolic pressure (mmHg) | 128 (119, 136) | 126 (117, 134) | 135 (130, 145) | <0.001 |
| >130 | 94 (40.3) | 61 (32.4) | 33 (73.3) | <0.001 |
| Diastolic pressure (mmHg) | 83.0 (76.0, 90.0) | 82.0 (76.0, 88.0) | 86.0 (77.0, 93.0) | 0.048 |
| >85 | 99 (42.5) | 75 (39.9) | 24 (53.3) | 0.101 |
| Comorbidities | ||||
| Hypertension | 38 (16.3) | 24 (12.8) | 14 (31.1) | 0.003 |
| Diabetes | 18 (7.7) | 9 (4.8) | 9 (20.0) | 0.001 |
| Chronic liver diseases | 15 (6.4) | 11 (5.9) | 4 (8.9) | 0.456 |
| Chronic lung diseases | 10 (4.3) | 9 (4.8) | 1 (2.2) | 0.446 |
| Cardiovascular diseases | 5 (2.1) | 4 (2.1) | 1 (2.2) | 0.969 |
| Cerebrovascular diseases | 3 (1.3) | 2 (1.1) | 1 (2.2) | 0.536 |
| Smoking | 19 (8.2) | 15 (8.0) | 4 (8.9) | 0.841 |
| Exposure history | ||||
| Contact with suspected or confirmed patients | 128 (54.9) | 100 (53.2) | 28 (62.2) | 0.274 |
| Contacted with people from Wuhan or non-Wuhan areas of Hubei province | 69 (29.6) | 57 (30.3) | 12 (26.7) | 0.630 |
| Visited Wuhan or non-Wuhan areas of Hubei province | 68 (29.2) | 53 (28.2) | 15 (33.3) | 0.496 |
| Time from symptom onset to admission (days) | 5.0 (2.0, 9.0) | 5.0 (3.0, 8.0) | 5.0 (2.0, 8.0) | 0.501 |
IQR, interquartile range; MS, metabolic syndrome; BMI, body mass index.
Forty-five (19.3%) of 233 patients had MS in our study. The age (median, 53.0 years vs. 46.0 years, P=0.004), BMI (median, 27.8kg/m2 vs. 23.9kg/m2, P<0.001), systolic pressure (median, 135mmHg vs. 126mmHg, P<0.001), and diastolic pressure (median, 86.0mmHg vs. 82.0mmHg, P=0.048) in patients with MS were significantly higher than non-MS patients. The proportion of male gender was comparable between two groups. The proportions of a hypertension history (31.1% vs. 12.8%, P=0.003) and type 2 diabetes (20.0% vs. 4.8%, P=0.001) in patients with MS were significantly higher than non-MS patients. Other comorbidities were not significantly different between two groups. In addition, symptoms at onset of illness were also comparable between MS patients and non-MS patients (Table 1).
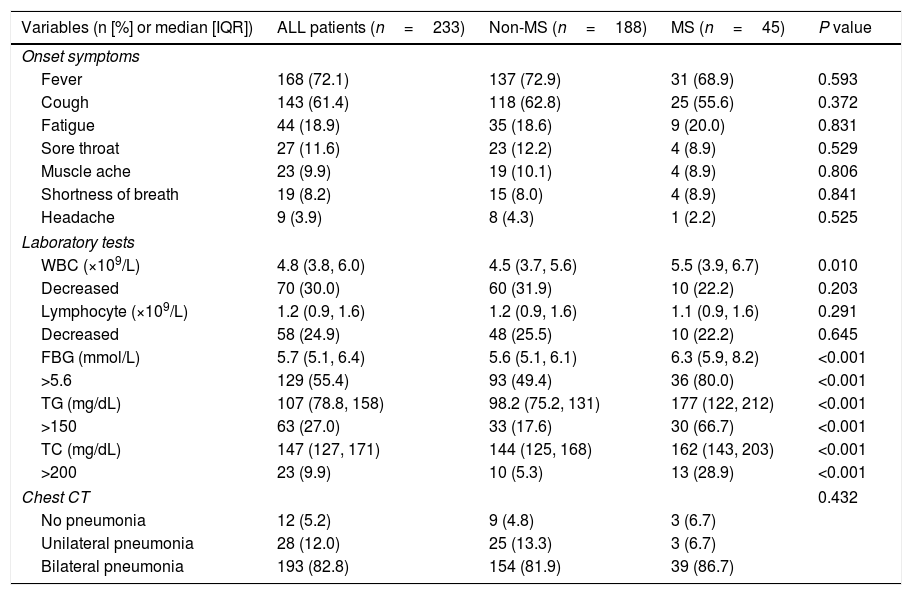
Symptoms, laboratory tests, and chest CT finding of patients with COVID-19 on admissionThe most common symptoms were fever (72.1%) and cough (61.4%), followed by fatigue (18.9%), sore throat (11.6%), muscle ache (9.9%), shortness of breath (8.2%), and headache (3.9%). The median white blood cells (WBC) and lymphocytes were 4.8 (IQR 3.8–6.0)×109/L and 1.2 (IQR 0.9–1.6)×109/L, respectively. The proportions of leukopenia and lymphopenia were 30.0% and 24.9%, respectively. The median FBG, TG, and TC were 5.7 (IQR 5.1–6.4) mmol/L, 107 (IQR 78.8–158) mg/dL, and 147 (IQR 127–171) mg/dL, respectively. The proportions of patients with FBG>5.6mmol/L, TG>150mg/dL, and TC>200mg/dL were 55.4%, 27.0%, and 9.9%, respectively. 221 (94.8%) of 233 patients presented abnormal chest CT images (Table 2).
Clinical characteristics of COVID-19 patients with and without metabolic syndrome.
| Variables (n [%] or median [IQR]) | ALL patients (n=233) | Non-MS (n=188) | MS (n=45) | P value |
|---|---|---|---|---|
| Onset symptoms | ||||
| Fever | 168 (72.1) | 137 (72.9) | 31 (68.9) | 0.593 |
| Cough | 143 (61.4) | 118 (62.8) | 25 (55.6) | 0.372 |
| Fatigue | 44 (18.9) | 35 (18.6) | 9 (20.0) | 0.831 |
| Sore throat | 27 (11.6) | 23 (12.2) | 4 (8.9) | 0.529 |
| Muscle ache | 23 (9.9) | 19 (10.1) | 4 (8.9) | 0.806 |
| Shortness of breath | 19 (8.2) | 15 (8.0) | 4 (8.9) | 0.841 |
| Headache | 9 (3.9) | 8 (4.3) | 1 (2.2) | 0.525 |
| Laboratory tests | ||||
| WBC (×109/L) | 4.8 (3.8, 6.0) | 4.5 (3.7, 5.6) | 5.5 (3.9, 6.7) | 0.010 |
| Decreased | 70 (30.0) | 60 (31.9) | 10 (22.2) | 0.203 |
| Lymphocyte (×109/L) | 1.2 (0.9, 1.6) | 1.2 (0.9, 1.6) | 1.1 (0.9, 1.6) | 0.291 |
| Decreased | 58 (24.9) | 48 (25.5) | 10 (22.2) | 0.645 |
| FBG (mmol/L) | 5.7 (5.1, 6.4) | 5.6 (5.1, 6.1) | 6.3 (5.9, 8.2) | <0.001 |
| >5.6 | 129 (55.4) | 93 (49.4) | 36 (80.0) | <0.001 |
| TG (mg/dL) | 107 (78.8, 158) | 98.2 (75.2, 131) | 177 (122, 212) | <0.001 |
| >150 | 63 (27.0) | 33 (17.6) | 30 (66.7) | <0.001 |
| TC (mg/dL) | 147 (127, 171) | 144 (125, 168) | 162 (143, 203) | <0.001 |
| >200 | 23 (9.9) | 10 (5.3) | 13 (28.9) | <0.001 |
| Chest CT | 0.432 | |||
| No pneumonia | 12 (5.2) | 9 (4.8) | 3 (6.7) | |
| Unilateral pneumonia | 28 (12.0) | 25 (13.3) | 3 (6.7) | |
| Bilateral pneumonia | 193 (82.8) | 154 (81.9) | 39 (86.7) | |
IQR, interquartile range; MS, metabolic syndrome; WBC, white blood cells; FBG, fasting blood glucose; TG, triglyceride; TC, total cholesterol.
The proportions of onset symptoms such as fever and cough were similar between patients with MS and without MS. The proportions of leukopenia and lymphopenia were also comparable between two groups. Patients with MS showed higher levels of FBG (median, 6.3mmol/L vs. 5.6mmol/L, P<0.001), TG (median, 177mg/dL vs. 98.2mg/dL, P<0.001), and TC (median, 162mg/dL vs. 144mg/dL, P<0.001) than non-MS patients. There was no significant difference in the proportion of abnormal chest CT images between two groups (Table 2).
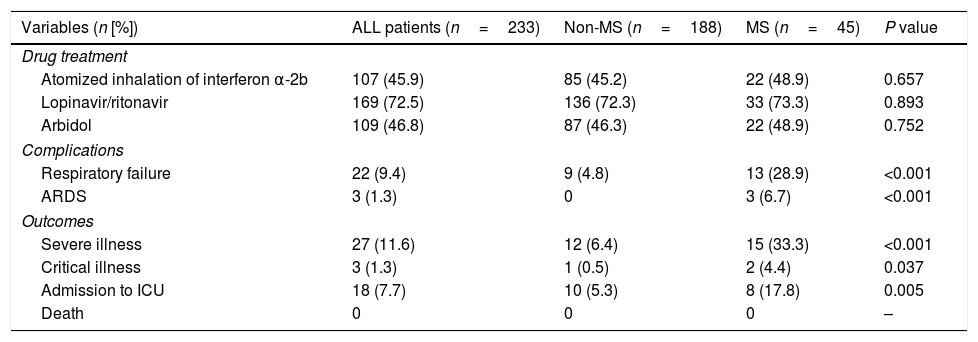
Treatment and clinical prognosis of patients with COVID-19The proportions of patients treated with atomized inhalation of interferon α-2b, lopinavir/ritonavir, or arbidol were 45.9%, 72.5%, and 46.8%, respectively. During hospitalization, 22 (9.4%) patients developed respiratory failure and 3 (1.3%) patients progressed to acute respiratory distress syndrome (ARDS). 179 (76.8%) patients were discharged, and 18 (7.7%) patients were transferred to the ICU. 27 (11.6%) patients had severe illness and 3 (1.3%) patients had critical illness during hospitalization. However, no patient was deceased in our study (Table 3).
Treatment and prognosis of patients with and without metabolic syndrome.
| Variables (n [%]) | ALL patients (n=233) | Non-MS (n=188) | MS (n=45) | P value |
|---|---|---|---|---|
| Drug treatment | ||||
| Atomized inhalation of interferon α-2b | 107 (45.9) | 85 (45.2) | 22 (48.9) | 0.657 |
| Lopinavir/ritonavir | 169 (72.5) | 136 (72.3) | 33 (73.3) | 0.893 |
| Arbidol | 109 (46.8) | 87 (46.3) | 22 (48.9) | 0.752 |
| Complications | ||||
| Respiratory failure | 22 (9.4) | 9 (4.8) | 13 (28.9) | <0.001 |
| ARDS | 3 (1.3) | 0 | 3 (6.7) | <0.001 |
| Outcomes | ||||
| Severe illness | 27 (11.6) | 12 (6.4) | 15 (33.3) | <0.001 |
| Critical illness | 3 (1.3) | 1 (0.5) | 2 (4.4) | 0.037 |
| Admission to ICU | 18 (7.7) | 10 (5.3) | 8 (17.8) | 0.005 |
| Death | 0 | 0 | 0 | – |
ARDS, acute respiratory distress syndrome; MS, metabolic syndrome; ICU, intensive care unit.
The proportions of patients treated with atomized inhalation of interferon α-2b, lopinavir/ritonavir, or arbidol were similar between two groups. More MS patients developed respiratory failure (28.9% vs. 4.8%, P<0.001), ARDS (6.7% vs. 0%, P<0.001), severe illness (33.3% vs. 6.4%, P<0.001) or critical illness (4.4% vs. 0.5%, P=0.037) compared to non-MS patients. More COVID-19 patients with MS were admitted to ICU compared to non-MS patients (17.8% vs. 5.3%, P=0.005) (Table 3).
We further analyzed the clinical characteristics and prognosis of patients with different numbers of MS components (Table S1). The median age were 36.5 years, 41.0 years, 49.0 years, 52.3 years, and 53.0 years in patients with 0, 1, 2, 3, 4–5 components of MS, and the proportions of male were 50.0%, 47.5%, 60.5%, 68.4%, and 71.4%, respectively. The proportions of abnormal chest CT images among these groups were comparable, and more than 90% patients had unilateral or bilateral pneumonia on admission in these groups. None of patients developed respiratory failure, ARDS, severe illness, critical illness, or were transferred to ICU in patients without MS component. However, with the increasing numbers of MS components, the proportions of respiratory failure, ARDS, severe illness, critical illness, or admission to ICU showed an increasing trend.
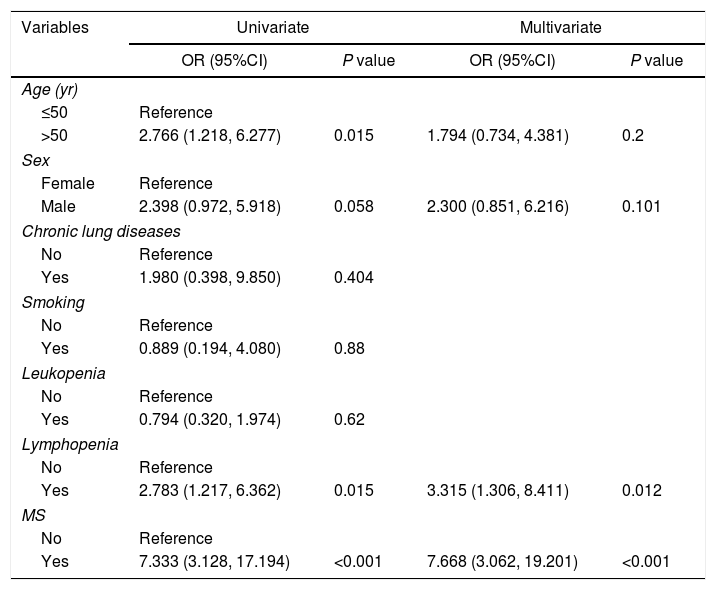
Risk factors of severe coronavirus disease 2019Univariate analysis presented that age>50 years (odds ratio [OR], 2.766; 95% confidence interval [CI], 1.218–6.277; P=0.015), lymphopenia (OR, 2.783; 95% CI, 1.217–6.362; P=0.015), and concurrent MS (OR, 7.333; 95% CI, 3.128–17.194; P<0.001) were associated with severe illness. Further multivariate analysis showed lymphopenia (OR, 3.315; 95% CI, 1.306–8.411; P=0.012), and concurrent MS (OR, 7.668; 95% CI, 3.062–19.201; P<0.001) were the independent risk factors of developing severe illness (Table 4).
Univariate and multivariate analysis of risk factors for severe coronavirus disease 2019.
| Variables | Univariate | Multivariate | ||
|---|---|---|---|---|
| OR (95%CI) | P value | OR (95%CI) | P value | |
| Age (yr) | ||||
| ≤50 | Reference | |||
| >50 | 2.766 (1.218, 6.277) | 0.015 | 1.794 (0.734, 4.381) | 0.2 |
| Sex | ||||
| Female | Reference | |||
| Male | 2.398 (0.972, 5.918) | 0.058 | 2.300 (0.851, 6.216) | 0.101 |
| Chronic lung diseases | ||||
| No | Reference | |||
| Yes | 1.980 (0.398, 9.850) | 0.404 | ||
| Smoking | ||||
| No | Reference | |||
| Yes | 0.889 (0.194, 4.080) | 0.88 | ||
| Leukopenia | ||||
| No | Reference | |||
| Yes | 0.794 (0.320, 1.974) | 0.62 | ||
| Lymphopenia | ||||
| No | Reference | |||
| Yes | 2.783 (1.217, 6.362) | 0.015 | 3.315 (1.306, 8.411) | 0.012 |
| MS | ||||
| No | Reference | |||
| Yes | 7.333 (3.128, 17.194) | <0.001 | 7.668 (3.062, 19.201) | <0.001 |
MS, metabolic syndrome; OR, odds ratio; CI, confidence interval.
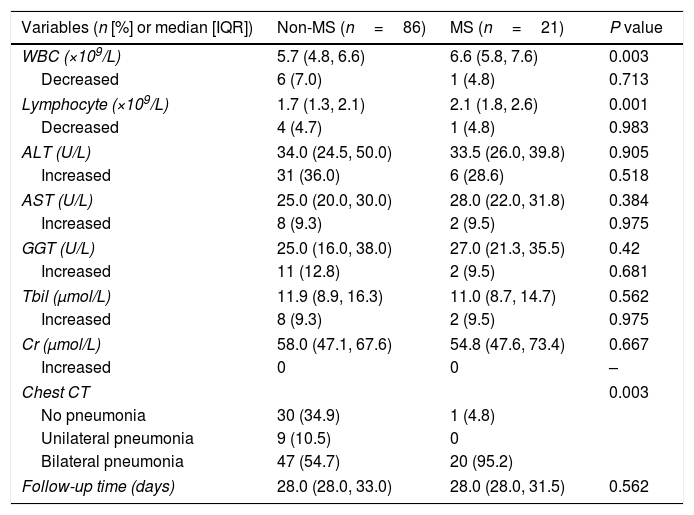
Among these COVID-19 patients, 107 patients (86 non-MS patients and 21 MS patients) with available follow-up data after discharge were analyzed. The median follow-up days of non-MS patients and MS patients were 28 (IQR 28–33) days and 28 (IQR 28–31.5) days, respectively. The laboratory and chest CT examinations during follow-up are shown in Table 5. The median WBC and lymphocyte counts were 6.6 (IQR 5.8–7.6)×109/L and 2.1 (IQR 1.8–2.6)×109/L in MS patients, which were significantly higher than that of non-MS patients. However, the proportions of leukopenia and lymphopenia were comparable between non-MS patients and MS patients. The median alanine transaminase (ALT), aspartate aminotransferase (AST), gamma-glutamyl transferase (GGT), total bilirubin (Tbil), and creatinine (Cr) levels in MS patients were 33.5 (IQR 26.0–39.8)U/L, 28.0 (IQR 22.0–31.8)U/L, 27.0 (IQR 21.3–35.5)U/L, 11.0 (IQR 8.7–14.7)μmol/L, and 54.8 (IQR 47.6–73.4)μmol/L, respectively. The proportions of elevated ALT, AST, GGT, Tbil, and Cr were 28.6%, 9.5%, 9.5%, 9.5%, and 0%, respectively. The median levels and abnormal proportions of ALT, AST, GGT, Tbil, and Cr were comparable between MS patients and non-MS patients. The proportion of patients with bilateral pneumonia was significantly higher in MS patients than non-MS patients (95.2% vs. 54.7%, P=0.003). We also analyzed the clinical symptoms of patients during follow-up. The results shown that only four patients (2.7%) remained had cough, three of whom were in non-MS group and one in MS group. The remaining patients had no significant symptoms.
Laboratory and radiological examinations of COVID-19 patients with Non-MS and MS at follow-up.
| Variables (n [%] or median [IQR]) | Non-MS (n=86) | MS (n=21) | P value |
|---|---|---|---|
| WBC (×109/L) | 5.7 (4.8, 6.6) | 6.6 (5.8, 7.6) | 0.003 |
| Decreased | 6 (7.0) | 1 (4.8) | 0.713 |
| Lymphocyte (×109/L) | 1.7 (1.3, 2.1) | 2.1 (1.8, 2.6) | 0.001 |
| Decreased | 4 (4.7) | 1 (4.8) | 0.983 |
| ALT (U/L) | 34.0 (24.5, 50.0) | 33.5 (26.0, 39.8) | 0.905 |
| Increased | 31 (36.0) | 6 (28.6) | 0.518 |
| AST (U/L) | 25.0 (20.0, 30.0) | 28.0 (22.0, 31.8) | 0.384 |
| Increased | 8 (9.3) | 2 (9.5) | 0.975 |
| GGT (U/L) | 25.0 (16.0, 38.0) | 27.0 (21.3, 35.5) | 0.42 |
| Increased | 11 (12.8) | 2 (9.5) | 0.681 |
| Tbil (μmol/L) | 11.9 (8.9, 16.3) | 11.0 (8.7, 14.7) | 0.562 |
| Increased | 8 (9.3) | 2 (9.5) | 0.975 |
| Cr (μmol/L) | 58.0 (47.1, 67.6) | 54.8 (47.6, 73.4) | 0.667 |
| Increased | 0 | 0 | – |
| Chest CT | 0.003 | ||
| No pneumonia | 30 (34.9) | 1 (4.8) | |
| Unilateral pneumonia | 9 (10.5) | 0 | |
| Bilateral pneumonia | 47 (54.7) | 20 (95.2) | |
| Follow-up time (days) | 28.0 (28.0, 33.0) | 28.0 (28.0, 31.5) | 0.562 |
IQR, interquartile range; WBC, white blood cells; PLT, platelet; ALT, alanine transaminase; AST, aspartate aminotransferase; GGT, gamma-glutamyl transferase; Tbil, total bilirubin; Cr, creatinine; CT, computed tomography; MS, metabolic syndrome.
Several impact factors of severity and prognosis of COVID-19 have been reported which included age, gender, comorbidities, etc.2–4 Nevertheless, few studies reported the impacts of MS on COVID-19. MS is a global epidemic and the complications of MS are diverse.7 The common consequences including cardiovascular disease and type 2 diabetes.7 In our study, 19.3% of COVID-19 patients had MS. The global prevalence of MS was ranged 20–35% in general population.18,19 The prevalence of MS was consistent with general population in our study, which suggested that MS may not a susceptible factor of COVID-19.
Consistent with previous studies, the most common symptoms were fever and cough in our study.20,21 There were no significant differences in clinical features between with MS patients and non-MS patients. About one third of patients presented leukopenia and lymphopenia on admission, while the proportions of leukopenia and lymphopenia were comparable between two groups. Also, the abnormal images of chest CT were not significantly different between two groups. These results indicated that concurrent MS may not associate with clinical manifestation of COVID-19.
Although no patient died in our study, 27 (11.6%) patients had severe illness and 18 (7.7%) patients were admitted to the ICU. Compared with non-MS patients, more MS patients developed severe illness and were admitted to the ICU. In addition, the proportions of respiratory failure and ARDS patients in MS patients were also significantly higher than non-MS patients. Despite the laboratory examinations were comparable between non-MS patients and MS patients, chest CT findings were more severe in MS patients during follow-up. These results implied that MS may be a risk factor of adverse outcomes of COVID-19. Further logistic regression analysis also demonstrated that concurrent MS increased the risk of severe illness in our study. Although previous studies found elderly patients had a worse prognosis,3,20 age was not associated with severe illness in our study. The possible interpretation may be that the age of patients was younger in our study than other studies.3,20 The treatment drugs, including atomized inhalation of interferon α-2b, lopinavir/ritonavir, or arbidol were comparable between MS patients and non-MS patients.
Currently, the mechanisms of the impact of MS on COVID-19 are not yet clear. Bijani et al. reported that MS was an independent risk factor of hypoxemia in influenza A (H1N1),10 which was associated with the severity of diseases. Similar results were observed in our study which found that more MS patients had respiratory failure and ARDS than non-MS patients during hospitalization. As an important determinant of MS, obesity could affect the progress of the many diseases.22,23 Excessive adipose accumulation may affect energy metabolism, neuroendocrine function, and immune function.24 Previous studies found that obesity could result in functional disorder of the body defense mechanisms. The mechanism may be interpreted as adipose accumulation induce the chronic aggravation of the pro-inflammatory responses of Th-1 type.25,26 Animal experiment also demonstrated that obesity could decreased the expression of pro-inflammatory cytokines and the cytotoxicity of natural killer cells.27 Several studies of influenza also found obesity was a risk factor for incidence of severe complications and mortality.28–30 Blood glucose level was another determinant of MS. Previous study reported that high blood glucose level was an independent risk factor of mortality and morbidity in patients with severe acute respiratory syndrome (SARS).31 Elevated blood glucose might reflect the severity of viral infection with multisystem involvement, which may increase the risk of hypoxia and mortality in patients with SARS.31 Our study also analyzed impacts of the numbers of MS components on COVID-19 patients which showed the severity of COVID-19 increased with the numbers of MS components.
Although some studies have reported the impacts of metabolic factors on COVID-19 and other virus infection-related respiratory diseases,3,4,10 association of concurrent MS with the prognosis of COVID-19 needs to be explored. COVID-19 patients with MS are more likely to develop severe complications. However, no patient was deceased in our study. Thus, the impacts of MS on the mortality of COVID-19 deserve further investigation. Furthermore, more than 90% patients remained had bilateral pneumonia in MS patients, which is significantly higher than non-MS patients during follow-up. Thus, long-term follow-up is necessary for these patients.
Our study has some limitations. First, our findings might be limited by the small sample size. However, by including consecutive COVID-19 patients in 10 designated hospitals from 10 cities, we consider our study population is much representative of cases diagnosed and treated in Jiangsu, China. Second, the association of MS and fatal outcome could not be analyzed in our study. Third, we only included hospitalized COVID-19, while those who were asymptomatic or had mild cases and treated at home were not included in our study. Thus, our study may represent the more severe COVID-19 patients. Fourth, this study used BMI and total cholesterol as alternative criteria of waist circumference and high-density lipoprotein cholesterol since these indexes were not routine tests in COVID-19 patients. Thus, the MS patients might be underestimated in our study. However, previous studies also used BMI and total cholesterol as alternative criteria for the diagnosis of MS.11,12 Fifth, the blood glucose and blood pressure values used in this study were detected at admission, which cannot reflect daily state of patients due to the stress situation. Sixth, about a third of patients were excluded due to the lack of BMI, TC, and/or TG and only 107 patients with available follow-up data after discharge were analyzed, which may result in selection bias. Finally, this study was retrospective, the impact of MS on COVID-19 needs to be validated in prospective studies.
In conclusion, 19.3% of the COVID-19 patients had MS in our study. COVID-19 patients with MS are more likely to develop severe complications and have worse prognosis. Therefore, more attention should be paid to COVID-19 patients with MS.
FundingYangzhou Key R&D Program (Social Development) (YZ2020101). China Postdoctoral Science Foundation for COVID-19 (2020T130049ZX).
Data availabilityThe datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Conflicts of interestThe authors have declared that no conflicts of interest exist.