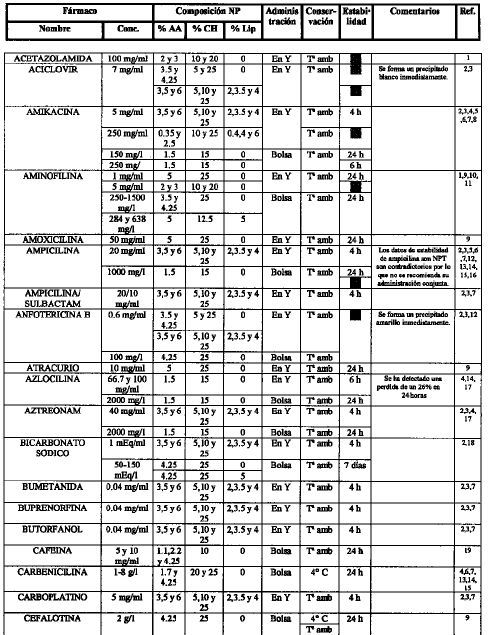
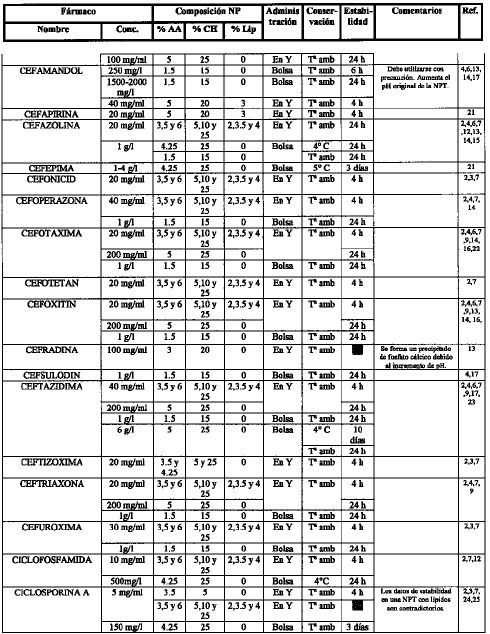
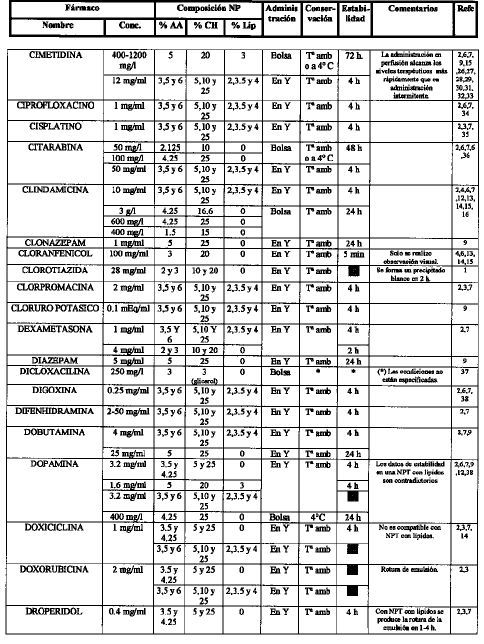
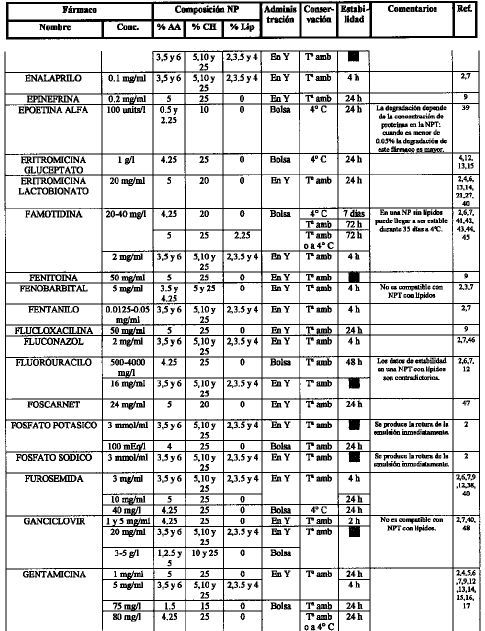
[1]Compatibility of neonatal parenteral nutrient solutions with selected intravenous drugs. Am J Health Syst Pharm 1996;53:2611-3.
[2]Compatibility of medications with 3-in-1 parenteral nutrition admixtures. JPEN 1999;23:67-74.
[3]Handbook of injectable drugs, 10.a ed. Bethesda, MD: ASHP; 1997.
[4]Issues in contemporary drug delivery. Part V: total parenteral nutrition. J Pharm Technol 1992;8:6-17.
[5]De Juana P, Bermejo T, Morell A, García B, Elviro J, Benlloch M..
Estabilidad de antibióticos administrados en Y con una mezcla de nutrición parenteral enriquecida en aminoácidos de cadena ramificada. Parte I: amikacina y gentamicina..
Nutr Hosp, 8 (1993), pp. 30-7
[6]Guía práctica para la administración de fármacos con la nutrición parenteral. Nutr Hosp 1991;6:345-55.
[7]Compatibility of parenteral nutrient solutions with selected drugs during simulated Y-site administration. Am J Health Syst Pharm 1997;54:1295-300.
[8]Visual compatibility of 30 additives with a parenteral nutrient solution. Am J Hosp Pharm 1979;36:511-3.
[9]Visual compatibility of total parenteral nutrition solution (Synthamin 17 Premix®) with selected drugs during simulated Y-site injection. Aust J Hosp Pharm 1994;24:167-70.
[10]Stability of aminophylline injection in three parenteral nutrition solutions. Am J Hosp Pharm 1983;40:428-32.
[11]Intravenous aminophylline: in vitro stability in fat-containing TPN. Ann Pharmacother 1992;26:127-8.
[12]Compatibility of antibiotics and other drugs in total parenteral nutrition solutions. Can J Hosp Pharm 1983;36:43-51.
[13]Piggyback compatibility of antibiotics with pediatric parenteral nutrition solutions. JPEN 1985;9:220-3.
[14]Compatibility considerations in parenteral nutrient solutions. Am J Hosp Pharm 1984;41:893-911.
[15]Incompatibilidades entre medicamentos y mezclas de nutrición parenteral. Farm Clín 1985;2:12-20.
[16]Analysis of antibiotic stability in a parenteral nutrition solution. Pediatr Infect Dis 1985;4:387-9.
[17]Antibiotic stability in a pediatric parenteral alimentation solution. J Pediatr 1998; 112:813-7.
[18]Compatibility and availability of sodium bicarbonate in total parenteral nutrient solutions. Am J Hosp Pharm 1985;42:2718-20.
[19]Stability of caffeine injection in intravenous admixtures and parenteral nutrition solutions. DCIP 1989;23:466-7.
[20]Stability of cefepime admixed with vancomycin, metronidazole, ampicillin, clindamycin, tobramycin, netilmicin, TPN solution, and PD solution. Bristol-Myers Squibb Company, Syracuse, New York, October, 1992.
[21]Compatibility of total nutrient admixtures and secondary antibiotic infusions. Am J Hosp Pharm 1985;42:362-3.
[22]Stability of ampicillin, piperacillin, cefotaxime, netilmicin and amikacin in a L-amino acid solution prepared for total parenteral nutrition of newborn infants. APMIS 1988;96:329-32.
[23]Wade CS, Lanpasona V, Mullins RE, Parks RB..
Stability of ceftazidime and amino acids in parenteral nutrient solutions..
Am J Hosp Pharm, 48 (1991), pp. 1515-9
[24]Compatibility of comonly used bone marrow transplant drugs during Y-site delivery. Am J Health Syst Pharm 1997;54:181-4.
[25]Stability of cyclosporine in dextrose 5%, NaCI 0.9%, dextrose/amino acid solution, and lipid emulsion. Ann Pharmacother 1992,26:172-5.
[26]Estabilidad de las mezclas de NP y su compatibilidad de las mezclas de NP y su compatibilidad con fármacos. Nutr Hosp 1989;4:6-11.
[27]Parenteral nutrient admixtures as drugs vehicles: theory and practice in the critical care setting. DTIC 1991;25:276-82.
[28]Histamine receptor antagonists and lipid stability in total nutrient admixtures. JPEN 1994;18:308-12.
[29]Role of histamine (H2)-receptor antagonists in total parenteral nutrition patients. Am J Med 1987;83:53-7.
[30]Stability of cimetidine in total parenteral nutrition. J Clin Nutr Gastroenterol 1987;2:40-3.
[31]Mitrano F, Baptista R..
Stability of cimetidine HCI and copper sulfate in a TPN solution..
DICP, 23 (1989), pp. 429-30
[32]Stability of cimetidine hydrochloride in parenteral nutrition solutions. Am J Hosp Pharm 1982;39:484-5.
[33]Stability of cimetidine hydrochloride in a total nutrient admixtures. Am J Hosp Pharm 1985;42:2208-10.
[34]Balet A, Cardona D, Pastor C, Márquez M..
Estabilidad in vitro de la amikacina y ciprofloxacina administradas en «Y» con dos nutriciones parenterales totales (NPT) de diferentes pH..
Nutr Hosp, 5 (1990), pp. 108-11
[35]Stability of cisplatin and etoposide in intravenous admixtures. Am J Hosp Pharm 1989;46:1400-4.
[36]Stability of citarabine in a parenteral nutrient solution. Am J Hosp Pharm 1985;42:592-4.
[37]American McGaw, Irvine, Ca; 1985.
[38]Compatibility of total nutrient admixtures and secondary cardiovascular medications. Am J Hosp Pharm 1985;45:777-8.
[39]Stability of human recombinant epoetin alfa in commonly used neonatal intravenous solutions. Ann Pharm 1996;30:466-8.
[40]Medication delivery in the short-bowel syndrome. JPEN 1993;17:180-5.
[41]Stability of famotidine 20 and 40 mg/l and amino acids in total parenteral nutrient solutions. Am J Hosp Pharm 1989;46:2321-5.
[42]Stability of famotidine 20 and 50 mg/l in total nutrient admixtures. Am J Hosp Pharm 1989;46:2326-8.
[43]Stability of famotidine 20 and 40 mg/l in total nutrient admixtures. Am J Hosp Pharm 1989;46:2329-32.
[44]Long-term stability of famotidine 20 mg/l in a total parenteral nutrient solution. Am J Hosp Pharm 1989;46:2333-5.
[45]Stability of famotidine in a 3-in-1 total nutrient admixtures. DICP 1990;24:232-5.
[46]Stability of fluconazole and amino acids in parenteral nutrient solutions. Am J Hosp Pharm 1992;49:1459-62.
[47]Visual compatibility of foscarnet with other injectable drugs during simulated Y-site administration. Am J Hosp Pharm 1990; 47:2075-7.
[48]Stability of ganciclovir sodium and amino acids in parenteral nutrient solutions. Am J Hosp Pharm 1994;51:503-8.
[49]Pharmacologic levels of heparin do not destabilise neonatal parenteral nutrition. JPEN 1998;22:311-4.
[50]Stability of heparin anticoagulant activity over time in parenteral nutrition solutions. JPEN 1996;20:219-21.
[51]Compatibility of hydromorphone hydrochloride in large-volume parenterals. Am J Hosp Pharm 1982;39:307-8.
[52]Stability of imipenem and cilastatina sodium in total parenteral nutrient solutions. JPEN 1990;14:306-9.
[53]Stability and activity of intravenous immunoglobulin with neonatal dextrose and total parenteral nutrient solutions. Ann Pharmacother 1994;28:1014-7.
[54]Visual compatibility of indometacin sodium trihydrate with drugs given to neonates by continous infusion. Am J Hosp Pharm 199l;48:2442-3.
[55]Availability of insulin from total parenteral nutrition solutions. JPEN 1990;14:262-4.
[56]Potential incompatibility of insulin and octreotide in total parenteral nutrient solutions. Am J Hosp Pharm 1989;46:1128.
[57]Biological activity of recombinant interleukin-2 in intravenous admixtures containing antibiotic, morphine sulfate, or total parenteral nutrient solution. Am J Hosp Pharm 1992;49:608-12.
[58]Antibiotic stability in solutions used for intravenous nutrition and fluid therapy. Pediatrics 1973;51:1016-26.
[59]Compatibility and stability of electrolytes, vitamins and antibiotics in combination with 8% amino acids solution. Am J Hosp Pharm 1978;35:33-44.
[60]Flagyl i.v. drug-drug physical compatibility. Am J IV Ther Clin Nutr 1981;8:9-16.
[61]Stability of penicillins in total parenteral nutrient solution. Am J Hosp Pharm 1987;44:1625-8.
[62]Stability of midazolam hydrochloride in parenteral nutrient solutions. Am J Hosp Pharm 1993;50:285-8.
[63]Stability of morphine and meperidine hydrochloride in a parenteral nutrient formulation. Am J Hosp Pharm 1985;42:1087-94.
[64]Stability of nizatidine in total nutrient admixtures. Am J Hosp Pharm 1991;48:1507-10.
[65]Stability of ondasetron hydrochloride in a total parenteral nutrient admixtures. Am J Health Syst Pharm 1995; 52:1557-8.
[66]Stability of propofol with parenteral nutrient solutions during simulated Y-site injection. Am J Health-Syst Pharm 1995; 52:192-6.
[67]Factors influencing the stability of ranitidine in TPN mixtures. Clin Nutr 1995; 14:171-6.
[68]Stability of ranitidine and thiamine in parenteral nutrition solutions. Nutrition 1997;13:547-53.
[69]In vitro evaluation of the stability of ranitidine hydrochloride in total parenteral nutrient mixtures. Am J Hosp Pharm 1990; 47:1574-9.
[70]Stability of ranitidine hydrochloride in total parenteral nutrient solution. Am J Hosp Pharm 1985;42;590-3.
[71]Stability of somatostatin in total parenteral nutrition. Pharm Week Scien Edit 1990;12240-2.
[72]Estabilidad de somatostatina en unidades nutrientes parenterales. Nutr Hosp 1991;6:98-101.
[73]Stability of tacrolimus injection in total parenteral nutrition solution. J Pharm Technol 1996;12:58-61.
[74]Stability and compatibility of teicoplanin in parenteral nutrition solutions used in pediatrics. Clin Nutr 1999; 18:159-65.
[75]Estabilidad de la teofilina en bolsas de nutrición parenteral unicapa y multicapa. Nutrición Hospitalaria (en prensa).
[76]Compatibility of thiotepa (lyophilized) with selected drugs during simulated Y-site administration. Am J Health-Syst Pharm 1996; 53:1041-5.
[77]The stability of amikacin, gentamicin, and tobramicin in total nutrient admixtures. JPEN 1989;13:505-9.
[78]Stability of urokinase in parenteral nutrition solutions. Nutr Supp Serv 1985;5:41-3.
[79]Martins A, McDougal A, Hamilton D, Igwemezie L, McErlane K..
In vitro assessment of vancomycin HCl compatibility after coinfusion with a specialised amino acid formulation..
JPEN, 15 (1991), pp. 536-9
[80]Stability of vancomycin hydrochloride in total parenteral nutrient solutions. Am J Hosp Pharm 1989;46:2055-7.
[81]Stability and delivery of vancomycin hydrochloride when admixed in a total parenteral nutrition solution. JPEN 1989;13:63-4.
[82]Vancomycin stability in heparin and total parenteral nutrition solutions: novel approach to therapy of central venous catheter-related infections. JPEN 1992;16:268-74.