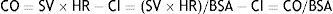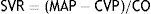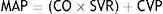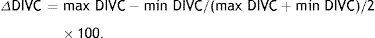The perioperative administration of fluids in critically ill patients is of particular interest to the anaesthetist, as a reduction in mortality rates and the harmful systemic effects of volume has been demonstrated, as long as they are administered in the context of individualised goals.
The aim is to optimise cardiac output. It is therefore necessary to analyse each patient's situation individually, particularly important being the physiological basis, identification of the area of the Frank–Starling curve they are in and the correct clinical application of static and dynamic parameters offered by modern monitoring techniques; the dynamic being the ones that predict with the greatest accuracy.
We carried out a non-systematic review of the passive leg-raising test and its correlation with end-tidal carbon dioxide. This is an affordable dynamic parameter and is easy to determine in the operating theatre as it does not require invasive surgical instruments. It predicts the response to volume on the basis of the increase in cardiac output by identifying patients who are responders to volume according to their ventricular function curve.
La administración de líquidos en el perioperatorio del paciente crítico, es una condición de especial interés para el anestesiólogo, puesto que está demostrado la disminución de la mortalidad y de los efectos deletéreos sistémicos del volumen, siempre y cuando la administración de los mismos se realice en torno a metas.
El objetivo es la optimización del gasto cardiaco, para esto, es necesario individualizar la situación de cada paciente, tomando especial relevancia el conocimiento de las bases fisiológicas, la identificación de la zona de la curva de Frank-Starling en que se encuentra y la correcta aplicación clínica de los parámetros estáticos y dinámicos que nos ofrece hoy día la monitorización, siendo los últimos, los que predicen con mayor certeza.
Se realizó una revisión no sistemática de la prueba de elevación de piernas pasiva y su correlación con el dióxido de carbono exhalado al final de la espiración, siendo este un parámetro dinámico, asequible, de fácil determinación en el quirófano al no requerir instrumentos invasivos y que permite predecir la respuesta a volumen teniendo como eje el incremento del gasto cardiaco mediante la identificación de los pacientes respondedores a volumen de acuerdo a la curva de función ventricular.
Management of perioperative fluid therapy is almost exclusively the responsibility of the anaesthetist. Despite the constant and almost daunting rate at which science is advancing these days, this area continues to be subject to debate and controversy. The lack of consensus initially concerned the availability of the different fluids, i.e. crystalloids or colloids, and which to use depending on the clinical scenario. However, over the last ten years or so, the issue of how much fluid should be administered has also taken on special importance.
There is a wide variability in practice and poor unification of criteria, both institutionally and interpersonally, in the field of anaesthetics. We now know that perioperative morbidity and mortality are strongly linked to the amount of intravenous fluid administered, whether too much or not enough.1,2 Intraoperative fluid volume administration is now calculated according to guidelines and formulas based on body weight, anaesthetic time, blood loss, baseline requirements, diuresis and the subjective perception of the trauma or surgical exposure.3 Fluid therapy should be individualised and integrative according to the patient's requirements, aiming to fulfil goals and objectives in order to achieve haemodynamic homeostasis that reduces complications following surgery.4
Volume expansion is the first-line treatment for acute haemodynamic instability. However, only 50% of patients respond to fluid administration by increasing stroke volume and cardiac index if the capacity for response to preload is not previously assessed5; moreover, the excessive expansion of circulating blood volume can cause harmful effects on the lungs as a result of increased extravascular fluid, so the importance of using reliable parameters that identify which patients will be responders to volume expansion cannot be over-emphasised. Haemodynamic parameters proposed to guide volume management are divided into static and dynamic; the dynamic parameters help identify patients who will benefit from volume expansion by increasing stroke volume (responder patient) while at the same time, avoid useless and potentially harmful treatment (non-responder patient).6
The purpose of this article was to conduct a non-systematic review, including critical analysis, of the available literature, in order to provide the anaesthetist with the best available evidence, with the aim of generating in their minds a clinical/practical correlation of the assessment of cardiovascular response to fluid challenges, using dynamic variables that can be measured in real time in the operating theatre, such as exhaled carbon dioxide after the passive leg-raising test.
Cardiac output and determinantsCardiac output (CO) is the amount of blood flowing into the circulation per minute. Arithmetically, it can be expressed as the product of stroke volume (SV) and heart rate (HR); under normal conditions, it ranges from 4.5 to 6l/min. The cardiac index (CI) is a much more specific measurement as it takes into account body surface area, with normal values in the range of 2.5–3.5l/min/m2:
SV is the amount of blood ejected by the ventricles in each contraction; the factors that determine it are: preload, afterload and contractility.PreloadThis is defined as the maximum degree of stretching or tension of the myocardial fibres before the onset of ventricular contraction and is determined by average sarcomere length at the end of diastole.7
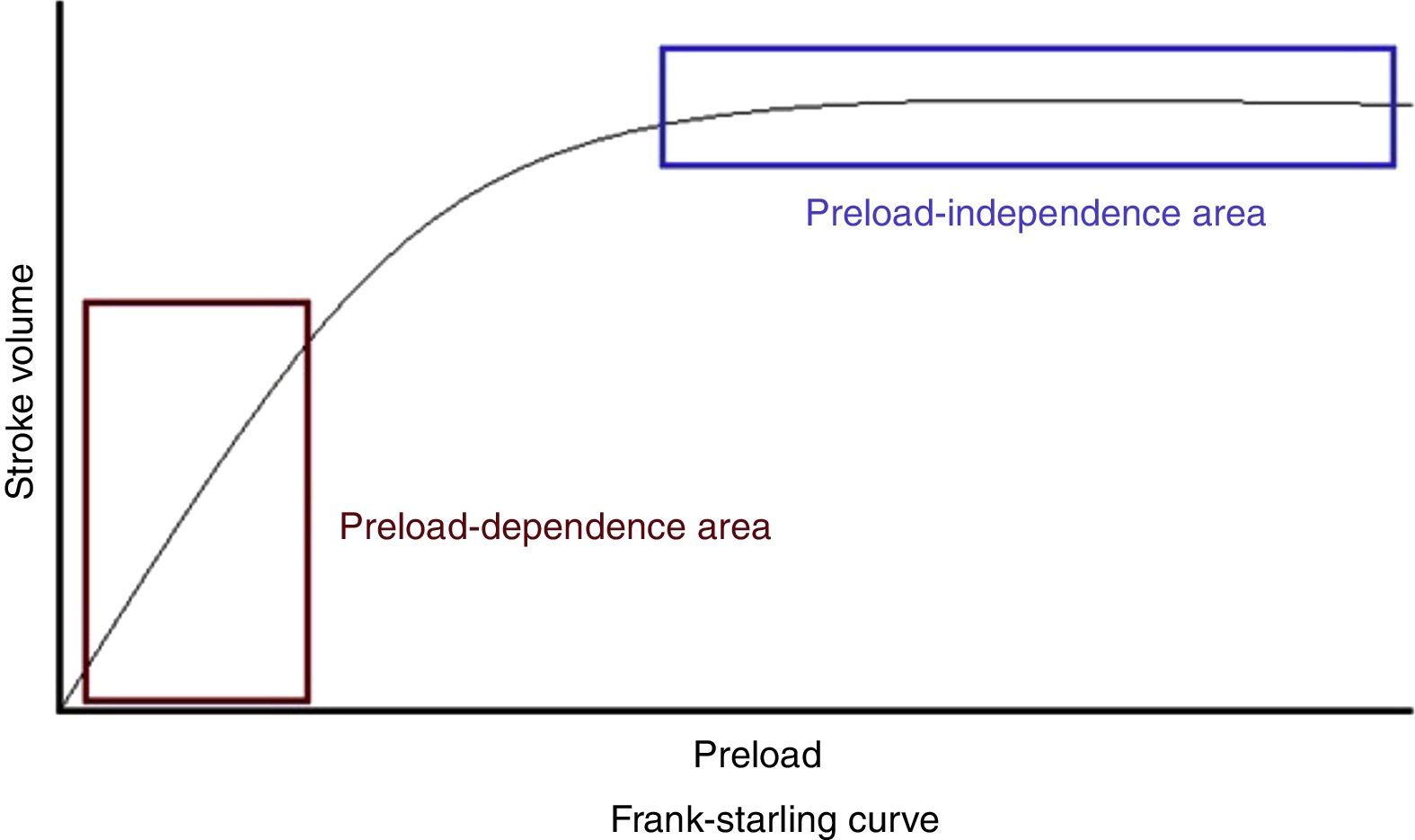
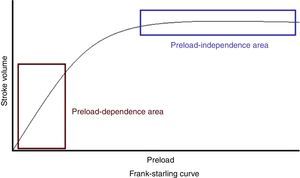
According to the Frank–Starling law or ventricular function curve, there is a direct relationship between the degree of fibre elongation in diastole and subsequent myocardial fibre shortening in systole. The stretching of the fibre leads to elongation of the sarcomere, thus increasing the number of sites of interaction between actin and myosin. Clinically, it could be defined as the end-diastolic dimension of the ventricle. At a constant heart rate, cardiac output is directly proportional to the preload up to a certain point, after which, although ventricular filling pressures may increase, the CO will not increase further and, under certain circumstances, may fall.7
Within the ventricular function curve (Figure 1), two stages can be defined: a first in which the increase in preload correlates linearly with an increase in SV (preload-dependent area); and a second in which the increased preload hardly correlates with increased cardiac output (preload-independent area).8
Preload, therefore, has a direct relationship with ventricular filling, whose main determinant is the venous return to the heart.
An increase in venous return will produce an increase in cardiac output in a healthy heart and central venous pressure (CVP) will remain within normal limits. In contrast, a diseased heart cannot cope with all the blood arriving and this translates into an increase in right atrial pressure (CVP) with decreased venous return due to the drop in the pressure gradient between the venous system and the right atrium, as well as a decrease in cardiac output.
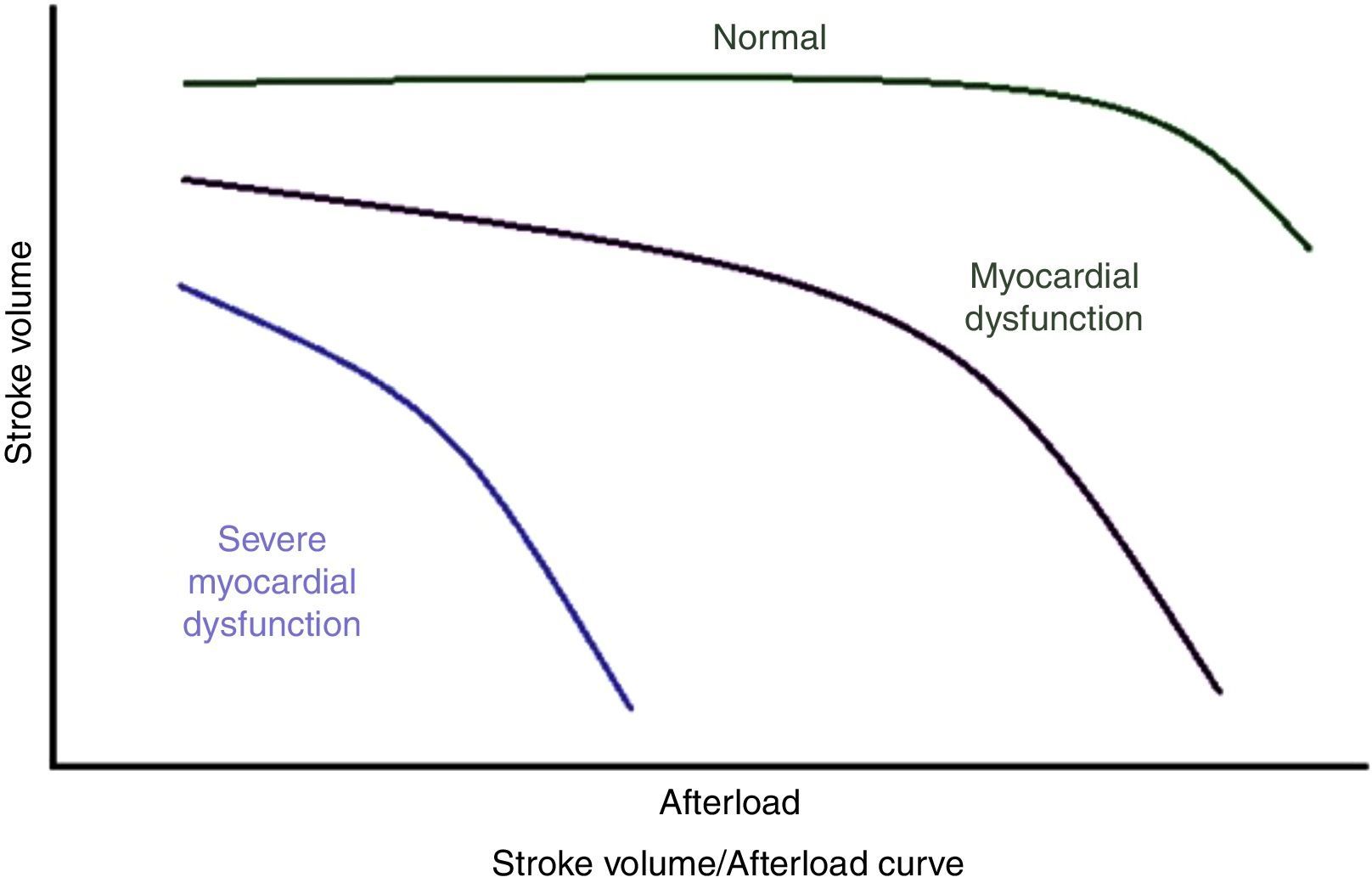
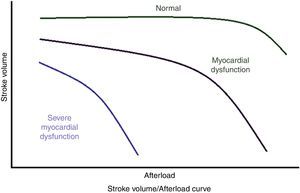
AfterloadConsidered as the force that opposes the ventricular wall during systole or arterial impedance to the ejection of stroke volume; also defined as the stress on the ventricular wall in systole and the load the ventricular wall has to support for ejection of the SV. Because the left ventricle (LV) is associated with the systemic circulation via the passage through the aortic valve, stroke volume and the pressure generated by the LV can be affected by the compliance and resistance of the arterial system.9
Afterload is the only factor inversely proportional to output; a healthy heart tolerates reasonable increases in resistance without reducing output (Figure 2), but after a certain limit, CO is inversely proportional to the afterload; in contrast, when the myocardium is diseased or when it is depressed by anaesthetic drugs, small increases in afterload lead to large reductions in output. Resistance is the determining factor in output when there is loss of inotropy, while the preload is the predominant factor in output in a normal heart.10
The factors determining ventricular pressure during systole are: the strength of ventricular contraction; the compliance of the walls of the aorta; and systemic vascular resistance. For practical purposes in medicine, systemic vascular resistance (SVR) tends to be equated with afterload, and can be calculated from the following formula:
From this formula it follows that the mean arterial pressure (MAP) depends directly on cardiac output (CO) and peripheral vascular resistance, as follows:ContractilityContractility or inotropy refers to the intrinsic ability of the cardiac cell that defines the force and speed of ventricular contraction and it is dependent on the availability of intracellular calcium (Ca). Under normal conditions, about 70% of Ca enters the sarcoplasmic reticulum via the Ca/ATPase pathway and almost 30% is sent to the extracellular space by the Na+/Ca pump.11 The use of inotropes promotes the release of or increase in intracellular Ca and sensitisation to it.
Although contractile activity is influenced by hormonal, neural, pharmacological and electrophysiological mechanisms, the heart has intrinsic mechanisms by which it can adapt CO to haemodynamic changes.12
Haemodynamic response to volume expansionThere are several techniques for determining whether or not fluid resuscitation is necessary, although we need to be aware that there are some conditions that diminish its utility, such as the need for mechanical ventilation, the presence of cardiac arrhythmias and right ventricular dysfunction.
We must not forget that just because there is a response to the volume expansion, it does not necessarily mean that volume was required. The need for fluid administration will be determined by the presence of signs of haemodynamic instability and organ dysfunction.6
Respiratory variations in venae cavae diameters: In mechanical ventilation, respiratory variations in the diameter of the inferior vena cava (ΔDIVC) have been shown to predict response to fluids in patients with circulatory failure.
During the inspiratory phase, increased pleural pressure is transmitted into the right atrium, reducing venous return and dilating the inferior vena cava (IVC). In contrast, during exhalation, reducing intrathoracic pressure favours venous return and decreases the diameter of the IVC. These oscillations in the diameter of the IVC are most evident in hypovolaemic patients and appear to die down in hypervolaemic patients when the IVC is less compliable.
The ΔDIVC is calculated as the difference between the maximum value and the minimum value of IVC diameter in a respiratory cycle divided by the mean of these values:
A ΔDIVC value ≥ 12% predicts response to volume administration with a positive predictive value of 93% and negative predictive value of 92%. In the superior vena cava (SVC), during the inspiratory phase, the increase in pleural pressure decreases the transmural pressure, reducing the diameter of the SVC. If the intravascular pressure (CVP) is sufficiently low, the transmural pressure can become negative and the SVC may collapse completely during inspiration, as in hypovolaemic patients. A SVC collapsibility index >36% can discriminate responders (defined as those with ≥11% increase in cardiac index after volume expansion) with a sensitivity of 90% and a specificity of 100%.6Filling pressures: Central venous pressure (CVP) and pulmonary capillary wedge pressure (PCWP) reflect the end-diastolic pressure of the right and left ventricles respectively. These parameters are called static and have never proved sufficient to assess an adequate response to fluid resuscitation. Despite that, it is accepted that very low values (CVP<5mmHg or PCWP<7mmHg) can be considered predictive of positive response to volume expansion. None of the parameters that estimate filling pressures by Doppler echocardiography are sufficiently reliable to predict response to volume expansion.6
Volumes and static areas: provide a better estimate of preload than pressures, but are not good predictors of response to volume expansion. Lack of response has been identified in right ventricular end-diastolic volume index (RVEDVI) with RVEDVI>140ml/m2 and adequate response with values <90ml/m2.
In terms of the left ventricular end-diastolic area (LVEDAI), the available data do not distinguish between responders and non-responders.
Measurement of the diameter of the inferior vena cava (DIVC) by ultrasound at the subxiphoid window level in end-expiratory phase has shown that a very low value (DIVC<12mm) predicts positive response to volume expansion and a very high value (>20mm), no response.6
Variation in right atrial pressure (ΔRAP): In theory, the right ventricle is preload-dependent, so it should decrease with each inspiratory effort with the drop in intrathoracic pressure. The absence of an inspiratory decrease would indicate that the heart is working in the flat part of the ventricular pressure-volume loop, and that volume expansion would not increase cardiac output. Magder et al. reported a correlation between RAP reduction >1mmHg in the inspiratory phase and the response to volume expansion, but the findings have not been reproduced in subsequent studies.6
Parameters derived from variations in arterial pulse wave pressure: The parameters derived from the arterial pressure curve have been shown to be poor predictors of response to volume expansion in patients with spontaneous breathing; in patients on mechanical ventilation, good correlation was shown between a variation in pulse oximetry plethysmographic wave amplitude (ΔPpleth) >15% and a variation in pressure of pulse (VPP) of >13% after fluid loading, with sensitivity of 87% and specificity of 100%. The Valsalva – variation of pulse pressure (ΔVPP), defined as the percentage of variation between the highest pulse pressure during the first phase of the Valsalva manoeuvre and the lowest pulse pressure during the second phase of the Valsalva manoeuvre, when an increase in intrathoracic pressure occurs with the consequent reduction in venous return and the consequent reduction in stroke volume, predicts a positive response to volume expansion when it increases by >52% during the manoeuvre, with a sensitivity of 91% and a specificity of 95%.6
Variation in arterial pressure with the end-expiratory occlusion manoeuvre: If during the inspiratory phase of mechanical ventilation, increased intrathoracic pressure decreases venous return and right ventricular preload, an end-expiratory occlusion manoeuvre (as used to measure intrinsic PEEP) would facilitate venous return and increase preload. This is done by applying an expiratory pause of 15s and observing changes in arterial pulse pressure and cardiac index. Responders are those patients showing an increase >15% in the cardiac index after volume administration.6
Passive leg-raising testThere are several tests that allow us to evaluate the likelihood of a patient responding to an intravenous fluid challenge. These tests assess static preload parameters such as pulmonary artery wedge pressure (PAWP) and central venous pressure (CVP); it has been shown through multiple studies that these parameters are poor predictors for response to volume.13,14
Unlike the static measurements, dynamic parameters, such as the variability of systolic pressure (VSP), variability of pulse pressure (VPP) and variability of stroke volume (VSV), are the result of heart-lung interaction and show the preload-dependence of the heart, providing a functional assessment of cardiac output in the context of transient changes in preload and an instant image or idea of heart function; these parameters have proven to be good indicators of the need for volumetric therapy.15,16
The dynamic measurements do not quantify the preload or provide information about blood volume, but estimate the cardiac response to changes in preload, which helps determine which area of the ventricular function curve both ventricles are working in. These variables are not indicated for predicting response to volume in clinical settings where there is no heart–lung interaction, as in patients with arrhythmias or who are breathing spontaneously.17
Regardless of the method used for quantification, dynamic preload-dependent parameters stand out for being the reversible and transient expression in cardiac output or stroke volume of a change in cardiac preload. The magnitude of this change helps to estimate the degree of preload-dependence; i.e. the larger the variation, the greater the degree of preload-dependence and, therefore, the greater the expected increase with volume administration.
The passive leg raising (PLR) manoeuvre is a quick, reversible, easy-to-perform test which has proven to be a good indicator of response to volume; the best indicator of response to PLR is an increase in the SV or CO. Aortic pulse pressure is directly proportional to left ventricular SV, and if there are no changes in arterial compliance, pulse pressure should indicate an increase in SV. Boulain et al. found an association between changes in radial pulse pressure and stroke volume (r=0.77, p<0.001) during PLR.18
Passively raising the legs to an angle of 45° with respect to the bed for at least 1min leads to rapid movement of blood, equivalent to about 300ml, from the lower limbs and abdomen to the chest cavity. This produces an increase in venous return, cardiac preload and, if both ventricles are working over the preload-dependent segment of the Frank–Starling curve, a significant transient increase in stroke volume and, consequently, in cardiac output.19,20
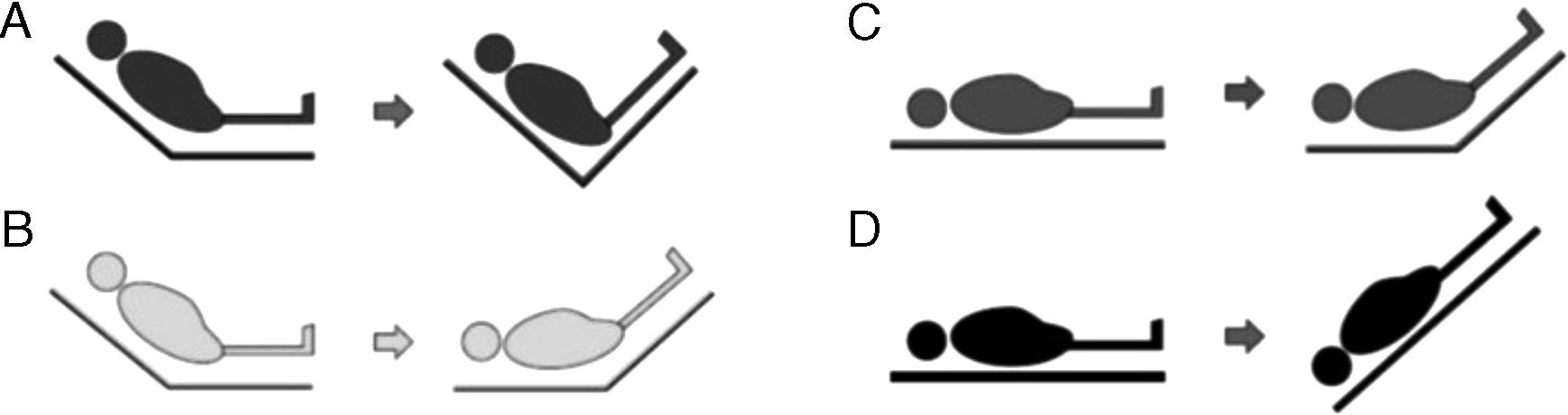
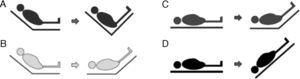
Four PLR methods have been described (Figure 3):
- A.
From Fowler's position, the legs are raised without lowering the trunk. This manoeuvre involves less risk of aspiration and elevation of intracranial pressure (ICP), but generates less volume expansion since the splanchnic blood volume is not included.
- B.
From Fowler's position, the legs are raised and the trunk is lowered to 0°. This is the method recommended by the experts, but it involves a greater risk of aspiration and increase in ICP.
- C.
From supine decubitus, the legs are raised 45° without moving the trunk. This was the first method to be studied, and provides less volume without avoiding the risks.
- D.
Change from supine decubitus to the Trendelenburg position. Involves a greater risk of complications.
A number of studies have shown the usefulness of this test for estimating the response to volume. An increase of ≥10% in cardiac output in the first 60–90s of the leg raising manoeuvre predicts, with sensitivity and specificity of over 90%, the capacity to raise CO with the administration of intravenous fluids.21
It is important to mention that, for correct assessment, a continuous or fast-response haemodynamic monitoring system is required that allows rapid detection of the haemodynamic changes induced during the manoeuvre.22 An increase in stroke volume >12% measured by either PiCCO, oesophageal Doppler or transthoracic echocardiogram during this manoeuvre has been able to predict an increase in stroke volume of >15% after volume expansion, with high sensitivity and specificity.23 In this context, an increase in aortic flow of >10% measured by oesophageal Doppler ultrasound during the leg raising manoeuvre predicts an increase in aortic flow of >15%, with sensitivity and specificity values 90%.23
However, one disadvantage of PLR is that it requires a direct measurement of CO, not available in most clinical areas, whether emergency department, intensive care unit or operating theatre, where they constantly have the dilemma of whether administering fluids for haemodynamic compensation is what the patient needs.
In this context, measurement of end-tidal carbon dioxide (ETCO2), which reflects the production of tissue CO2 (VCO2), alveolar ventilation and CO (in practical terms, the pulmonary blood flow), could be an alternative method for assessing the effects of the test when evaluation of the cardiac index (CI) is not available. In fact, the amount of CO2 exhaled is proportional to the CI in stable respiratory and metabolic conditions,24 and changes in exhaled CO2 have reflected changes in CO.25,26 Therefore, provided that ventilation is unaltered and cell metabolism is stable, ETCO2 could allow continuous and non-invasive estimation of changes in CI during PLR.
In January 2016, Monnet et al.27 and, in May of the same year, Cherpanath et al.,28 conducted two multicentre studies in which they carried out searches in databases such as MEDLINE, EMBASE and Cochrane, finding 21 and 23 clinical studies respectively, identifying two studies that mentioned CO2 as surrogate for CO for predicting haemodynamic response to raising of the lower limbs.
Monge García et al.29 conducted a clinical trial with 37 patients on mechanical ventilation with acute circulatory failure. The patients were monitored with CardioQ-ODM® oesophageal Doppler and the PLR manoeuvre was applied for 2min; response to fluids was defined according to increase in CO≥15% (responders) after volume expansion. The increase in CO and ETCO2 induced by the PLR test correlated strongly (R2=0.79, p<0.0001). The areas under the ROC curve for PLR showed an increase in CO and ETCO2 (0.97±0.03SD, 95% CI: 0.85–0.99 and 0.94±0.04 SD; 95% CI: 0.82–0.99 respectively) with no significant differences. An increase ≥5% in ETCO2 or ≥12% in CO during PLR predicted fluid responsiveness with a sensitivity of 90.5% (95% CI: 69.9–98.8%) and 95.2% (95% CI: 76.2–99.9%) respectively, and a specificity of 93.7% (95% CI: 69.8–99.8%).
Monnet et al., determined the role of ETCO2 in 65 patients under controlled mechanical ventilation, with or without arrhythmia; they found that an increase in exhaled end-tidal CO2 of over 5% with PLR predicts an increase in CI of over 15%, with a sensitivity of 71% and a specificity of 100%, with significant AUC (0.93) and moderate correlation (R2=0.45).30,31
These two trials have the drawbacks of a limited number of patients and a weak positive correlation (R2=0.49) and moderate positive correlation (R2=0.79) respectively; further studies with larger numbers of patients are needed to evaluate these parameters in greater depth. Nevertheless, we would highlight the importance of opening up a new affordable perspective for our patients.29,30
ConclusionsPassive leg raising is a manoeuvre that can determine accurately and reversibly which patients are in the preload-dependent area of the Frank–Starling curve; an increase in ETCO2≥5% after the manoeuvre predicts an increase in CO≥12% and ≥15% in CI, with adequate sensitivity and specificity. This positions the test as a simple-to-apply, supremely useful tool for the anaesthetist in perioperative fluid therapy, although more research is needed to provide a definitive recommendation.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that no patient data appear in this article.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflict of interestThe authors declare that they have no conflict of interests.