The high mobility group box 1 proteins (HMGB1) are non-histone nuclear proteins reported to be present at high levels in some autoimmune diseases, such as systemic lupus erythematosus (SLE). Likewise, in contrast to healthy individuals, patients with SLE have a higher prevalence of anti-HMGB1 antibodies, and these levels have also been associated with heightened disease activity. This article will discuss the involvement of these proteins in immunology, and review the evidence supporting their clinical importance in SLE.
Materials and methodsA narrative review was conducted based on a search of the literature up to October 2018, of articles describing the function, structure, prevalence and importance of HMGB1 in different manifestations of SLE. Articles focusing on the presence of HMGB1 and/or its antibodies in patients with SLE or other autoimmune diseases were also reviewed.
ResultsA total of 69 articles were found. These articles were the foundation to define the structure and functions of HMBG1, including its role as a cytokine released by immune cells in inflammatory processes and necrosis. Additionally, a description of its functions in phagocytosis and NETosis - that have an impact on autoimmune diseases, primarily in SLE - was included.
ConclusionHMGB1 proteins and anti-HMGB1 antibodies are elevated in the serum of patients with SLE, in contrast with healthy individuals or non-severe presentations of the disease; this suggests that they may play a role as a biomarker of disease activity.
Las high mobility group box 1 protein (HMGB1, «proteínas de alta movilidad del grupo 1») son proteínas nucleares no histonas cuyos niveles se han documentado elevados en ciertas enfermedades autoinmunes, como el lupus eritematoso sistémico (LES). Igualmente, los pacientes con LES presentan una mayor prevalencia de anticuerpos anti-HMGB1 comparados con individuos sanos, al mismo tiempo que se han relacionado sus niveles con una mayor actividad de la enfermedad. En este artículo se revisará la participación de estas proteínas en la inmunología y se abordará la evidencia que sustenta su importancia clínica en el LES.
Materiales y métodosSe realizó una revisión narrativa basada en la búsqueda de la literature hasta octubre de 2018, de artículos que describieran la función, estructura, prevalencia e importancia de las HMGB1 en diferentes manifestaciones del LES, así como artículos que hayan estudiado la presencia de las HMGB1 o sus anticuerpos en pacientes con LES u otras enfermedades autoinmunes.
ResultadosSe encontraron un total de 69 artículos. Con base en ellos definimos la estructura y funciones de las HMBG1, incluyendo su papel como citocina liberada por células inmunes en procesos inflamatorios y en necrosis. Adicionalmente, describimos sus funciones en la fagocitosis y NETosis, que genera implicaciones en enfermedades autoinmunes, principalmente en el LES.
ConclusiónLas proteínas HMGB1 y los anticuerpos anti-HMGB1 se encuentran elevados en suero de pacientes con LES comparados con individuos sanos o con formas no severas de la enfermedad, evidenciando que estas pueden comportarse como un biomarcador de actividad de la enfermedad.
Systemic Lupus Erythematous (SLE) is the prototype of autoimmune systemic diseases and is characterized by clinical manifestations ranging in severity from a mild skin and joint disease, to more severe conditions such as renal, neurological and lung involvement, that in some cases may lead to death. This is associated with autoimmune serology findings, of which the most relevant are the presence of antinuclear antibodies, anti-double stranded DNA, anti-Sm.1 The pathogenesis of SLE is still unknown and it is believed to have a multifactorial origin, with a convergence of genetic, environmental, hormonal, epigenetic, and immunoregulatory factors that lead to the development of the disease.2
Over the last few years, it has been shown that the impaired clearance of apoptotic cells is one of the most relevant aspects in the pathophysiology of SLE, since it has been proven that the accumulation of apoptotic bodies is one of the key components in the onset and later development of SLE.3,4 It has been shown in murine and human models, that protein deficiencies such as C1q complement factor and serum P amyloid protein, which are involved in opsonization and also in phagocytosis and apoptotic cell clearance, lead to the development of lupus-like characteristics such as autoantibody production and proliferative glomerulonephritis.5,6 Likewise, there has been evidence in humans of an accumulation of apoptotic cells, including high levels of apoptotic T-lymphocytes and neutrophils in the peripheral blood.7,8 Moreover, apoptotic lymphocyte cells are also seen in the lymph nodes of patients with SLE.9 In such environment, any uncleared apoptotic cells turn into cells that will lose their membrane integrity, and hence lead to the release of intracellular structures that may behave as autoantigens.3 One such example are the nucleosomes, that bound to the high mobility group box 1 proteins (HMGB1), not only trigger immune but also inflammatory responses, which are relevant for the pathogenesis of SLE.10 This narrative review describes the functions of HMGB1 and discusses their role in the pathogenesis of SLE.
MethodsLiterature search methodsA narrative review was conducted based on articles published until October 2018. The following were among the databases consulted: SciELO, Clinical Trials, Clinics Review Articles, Academic Search Ultimate, Medline, Embase and Google Scholar. The search included articles written in English and Spanish. The Medline search was conducted through PubMed, using the MeSH terms: HMGB1; antibodies; systemic lupus erythematosus; antigen; autoimmunity. Then, the terms were linked to the Boolean connector AND.
Selection of articles and data miningAt the end of the search, the articles were stored in an Excel database. Duplicate articles were excluded and the selection process of the articles relevant to this publication was initiated. Any articles that included keywords in the title or the abstract were considered. Every article had to be compliant with the inclusion criteria, and finally a consensus was reached among all the authors to standardize and review the database.
Inclusion criteria- none-
Types of studies: cohort studies, cases and controls, randomized and non-randomized trials, subject reviews, and experimental studies.
- none-
Type of population: adult patients with SLE in whom the presence of HMGB1 or its antibodies had been analyzed.
- none-
Intervention: studies describing the function, the structure, the prevalence and the importance of HMGB1 in different manifestations of SLE.
- none-
Articles for which full text access was not available.
- none-
Case reports.
- none-
Duplicate articles.
After the initial search, 69 articles were found, most of them in Medline and Google Scholar. When excluding the articles to which full access was not available, as well as any duplicate articles, 67 articles were finally included.
Following is a description of the highlights from the literature review.
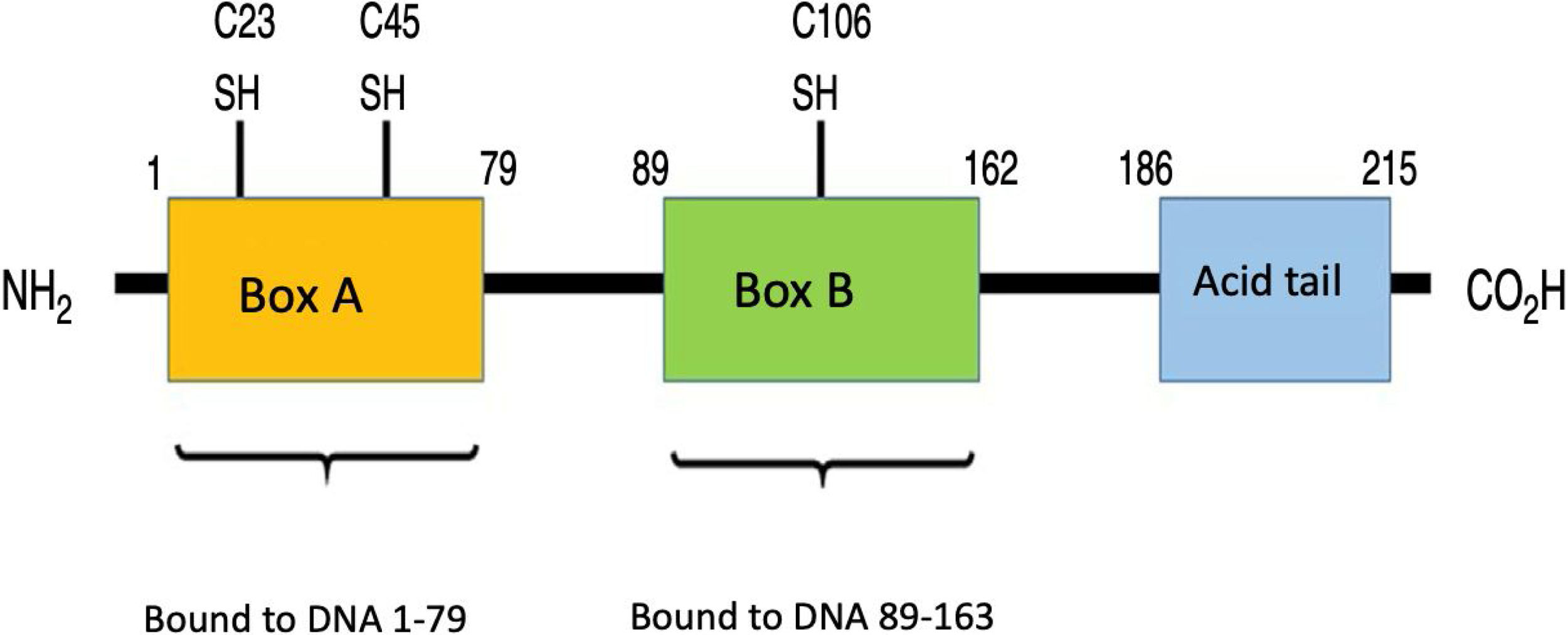
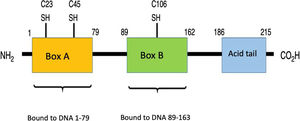
What are HMGB1proteins?HMGB1 proteins, also known as amphoterine, belong to the non-histone family of proteins known as HMG, which are found in the nucleus of eukaryotic cells.11,12 These proteins bind to several types of DNA, such as the double-stranded DNA (dsDNA), the single stranded DNA, the distorted DNA, and the nucleosomes, participating in transcriptional regulation via 3 mechanisms: the first one is nucleosome binding, behaving like a chromatin fluidifier, which facilitates nucleosome remodeling; the second one causes the bending of the DNA promoter site, increasing the affinity of the binding protein to TATA, allowing for a more efficient selection of factors such as TFIIB or TFIIA (essential for DNA polymerase II recognition); finally, the third mechanism is the ability of HMGB1 to establish a protein-protein interaction with specific transcription factors (such as p53, p73 and NF-kB, inter alia). Upon binding, the HMGB1 may fold the DNA and stabilize their target associated protein, or promote the selection of other proteins.13 Moreover, it is coded in human chromosome 13q12-13 and comprises 3 different domains: Box A, Box B, and C-terminal acidic tail. The latter contains a section of around 30 continuous glutamic and aspartic acid residues.14 Box A plays the role of competitive HMGB1 antagonist and has the ability to inhibit the activity of another HMGB1 molecule. Both Box A and Box B are important for DNA binding as previously described, and are positively charged, while the C-terminal tail is negatively charged, giving the molecule a bipolar charge.15,16Fig. 1 illustrates the structure of HMGB1.
HMGB1 structure. Human protein HMGB1 has 125 amino acid residues and comprises 3 domains: one Box A, one Box B and one acid tail at the terminal carboxyl end. There are 3 redox-sensitive cysteine residues at positions 23, 45 and 106 that regulate the HMGB1 function in response to oxidative stress. The binding domains to the RAGE receptor are at position 150–183 and those binding to TLR are between residue 80–108 (no shown in the picture).
It has also been shown that HMGB1 may be released into the extracellular environment, where they play different roles. One of the best recognized roles is the damage-associated molecular pattern, which is passively released by cells that die from necrosis or immune apoptosis (i.e., NETosis), but not as the result of physiological apoptosis, recognized by Toll like receptors (TLR) 2 and 4, activating an inflammatory response.17,18 Similarly, these proteins may be actively secreted by some types of immune cells such as macrophages, dendritic cells, and natural killer lymphocytes, in response to an infection, a tissue injury or any other inflammatory stimulus.18 In 1999, Wang et al. discovered the ability of the macrophage to secrete HMGB1 as a late mediator of inflammation and sepsis, noticing its release after the peak of proinflammatory mediators such as tumor necrosis factor (TNF) and IL-1, highlighting the role of these proteins as late mediators in these conditions and potential severity indicators.19–21 As already mentioned, HMGB1 proteins are now recognized as cytokines because they act as mediators of the systemic inflammatory response (selecting and facilitating the activation of macrophages and other inflammatory cells, stimulating IL-6 and TNF production); they are secreted by activated immune cells, activate the prototypical inflammatory response of immune and endothelial cells, and finally, transduce cell signals through the TLR receptors and the receptor for advanced glycation end products (RAGE), which are so far the key HMGB1-activated receptors. RAGE signaling results in the activation of the nuclear-κB factor pathway, and in the transduction of signals through the extracellular regulated kinase (ERK) and p38, which promotes the production of inflammatory cytokines (TNF, IL-6 and interferon gamma [IFN-γ]). Furthermore, HMGB1-dependent TLR2 and TLR-4 activation results in the activation of the κB nuclear factor through a myeloid differentiation 88 protein -dependent mechanism (MyD88). There is however considerable interest in determining a different type of receptors activated by this protein, in order to learn more about these molecules as inflammation mediators.18
HMGB1 expression and cell secretionHMGB1 proteins are expressed by practically all cells, except those with no nucleus, such as erythrocytes, and a few intestinal cells. It must be highlighted that these proteins are exclusively found in the nucleus. As already mentioned, these proteins may be released in two ways into the extracellular space: actively, through immune cells (such as macrophages) in response to an inflammatory stimulus playing a cytokine role; and passively, through cells undergoing necrosis, releasing intracellular proteins such as HMGB1.22
The active secretion of HMGB1 proteins by immune cells such as macrophages and dendritic cells is controlled by a previous acetylation process comprising two steps.22 The first is the passage of HMGB1 outside the nucleus which requires acetylation in most of the 43 cysteine residues distributed throughout the molecule inside the nucleus, avoiding the interaction between the proteins and the nuclear protein import complex so as to prevent their return to the nucleus. However, the mechanism whereby an activated immune cell induces HMGB1 acetylation for its secretion is jet to be fully understood. The second process consists of the migration of the cytosolic and acetylated HMGB1 into the cytoplasmic secretory vesicles for their subsequent release into the extracellular space.23–25 The mRNA expression encoding the HMGB1 proteins is positively regulated by certain cytokines such as IFN-γ, TNF, and transforming growth factor beta; this has been shown in cultured THP-1 macrophage cell lines and in human peripheral blood monocytes. Likewise, these factors also induce intracellular redistribution and secretion of HMGB1 proteins.26,27
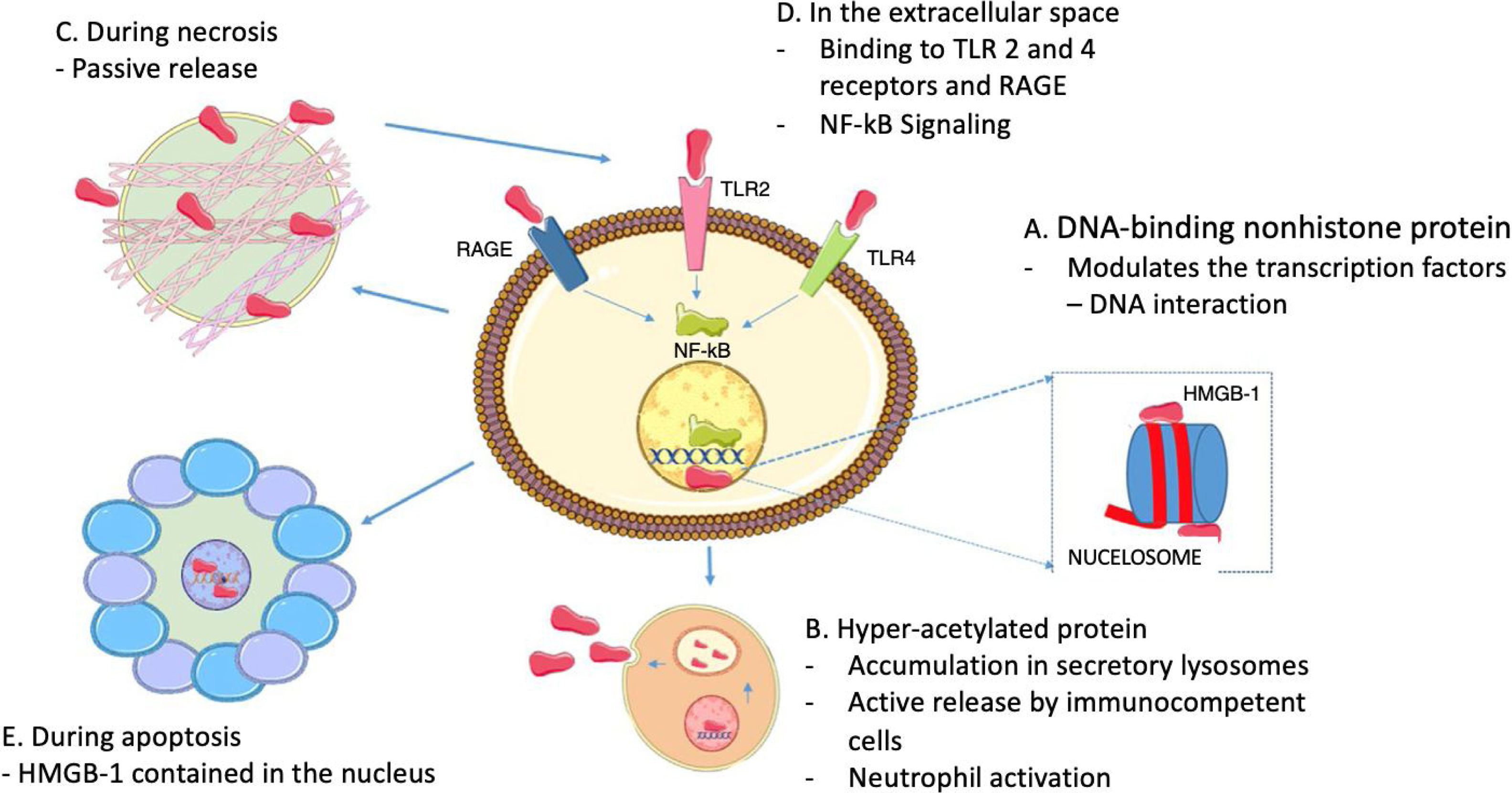
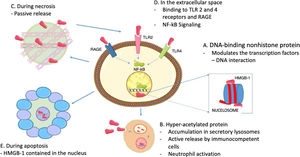
In terms of the role of HMGB1 as cytokine, it does not only depend on its active release through immune cells, because HMGB1 proteins are usually bound to chromatin, so if a cell turns necrotic, the HMGB1 proteins passively spread into the extracellular space.22 It must be highlighted that this does not happen when cells die by apoptosis, since the HMGB1 bind to the cruciform DNA (DNA structures formed by inverted repeats reinforced by supercoiled DNA), which forms after DNA excision (through topoisomerase ii) at nucleosome intervals, or bind to hypoacetylated proteins inside the apoptotic cell nucleus.28 This difference in the mode of cell death may be the basis for the different abilities of the necrotic and apoptotic cells to activate an inflammatory response. Fig. 2 summarizes the intranuclear and extranuclear roles of HMGB1 proteins, and their release modes.
HMGB1 Associated functions. A. Inside the nucleus, HMGB1 proteins are loosely and temporarily associated with the nucleosomes and are important in controlling DNA transcription and for the interaction with the nuclear matrix. B. Active HMGB1 secretion through immunocompetent cells such as macrophages and dendritic cells, following bacterial product activation such as endotoxins, proinflammatory cytotoxins; hyperacetylated HMGB1 accumulation in secretory lysosomes, cell membrane fusion and subsequent release into the extracellular space is required for secretion. C. During necrosis, HMGB1 proteins are passively secreted, generating inflammatory processes. D. When present in the extracellular fluid, HMGB1 signaling through RAGE and TLR 2 and 4 results in the activation of survival signals involving the nuclear factor-kB (NF-kB). E. During apoptosis, HMGB1 proteins are sequestered inside the nucleus hence preventing their release.
It has been shown that HMGB1 proteins may develop complexes when binding to molecules such as chemokine CXCL12, liposaccharide, IL-1, CpG sequences, dsDNA, and nucleosomes.3 This characteristic increases up to 100-fold the macrophage response by forming complexes with liposaccharide and Pam3CSK4 – a synthetic triacylated lipoprotein – hence inducing the production of cytokines via TLR4, binding to the lipopolysaccharide, and via TLR-2, with Pam3CSK4.29,30 Furthermore, HMGB1 proteins are an essential component of DNA-containing immune complexes, capable of stimulating the production of cytokines through a TLR9-MyD88 pathway involving the multivalent RAGE receptor.29 Likewise, the binding of HMGB1 to the class CpG motifs, significantly increases the production of cytokines through TLR9 and RAGE. Moreover, some autoimmune diseases are mediated by the formation of immune complexes involved in the damage and mediation of the local inflammatory response of certain organs. A case in point is SLE, an autoimmune disease in which organ damage – the kidney, for example – is mediated by immune complexes.1 It has also been shown that immune complexes with DNA and HMGB1 may induce the production of anti-dsDNA autoantibodies in patients with SLE.30 It is also probable that complexes formed by HMGB1 may play a role in the pathophysiology of lupus nephritis (LN), since it has been shown that patients experiencing this complication exhibit an increased HMGB1 expression as compared to other renal diseases; so they behave as a proinflammatory mediator in autoantibody-induced kidney damage in patients with LN.31
It has also been shown that by stimulating endothelial cells with immune complexes in patients with SLE, there is an increase in the production of proinflammatory cytokines such as IL-6 and IL-8. Moreover, RAGE, VCAM-1 and ICAM-1 showed a higher expression on the surface of endothelial cells, in the presence of immune complexes. These effects could be blocked by adding the inhibitory A box of HMGB1 or soluble RAGE, indicating the significant role of HMGB1 proteins in mediating endothelial inflammation.32 This shows that, depending on the effector cells and other components involved, HMGB1 proteins play a regulatory role over the immune complexes.
HMGB1 and their role in phagocytosisPhagocytosis is an immune process in which the innate immune response cells, macrophages, and neutrophils phagocyte or “ingest” microorganisms, in addition to cell debris generated through mechanisms such as apoptosis and necrosis (this process is also called spherocytosis or necroptosis, respectively). In both cases, their role is essential in the resolution of inflammation.33 Similarly, the disruption of apoptotic debris in spherocytosis has been associated with the pathogenesis of SLE, leading to secondary cell necrosis and to exposure of the intracellular contents to the surrounding tissue, which may also induce an inflammatory response.34 Furthermore, serum molecules such as complement factors are important in phagocytosis and are reduced in patients with SLE.35 It has been said that HMGB1 proteins are a potential contributing factor to the reduction of phagocytosis through various mechanisms.36,37 During apoptosis, the phosphatidylserine molecules found inside the cell express in the cytoplasmic membrane and act as “ingestion” signals that are recognized by a broad range of receptors at the phagocyte membrane, such as integrins, allowing for the identification and removal of apoptotic cells.38 HMGB1 proteins may bind to the phosphatidylserine present on the surface of apoptotic neutrophils, inhibiting one of the most important signals for the macrophage to phagocyte this cell.36 This was shown in vitro, where macrophages previously incubated with HMGB1, experience a reduced ability to phagocyte apoptotic neutrophils, since the HMGB1 proteins compete for binding to integrin αvβ3 with milk fat globule-epidermal growth factor 8, which behaves as an opsonin, hence preventing the recognition and elimination of apoptotic neutrophils.39 In fact, recent studies have shown the participation of the RAGE receptor as a potentiator of phagocytosis through its integration with phosphatidylserine.40 Similarly, RAGE deficient macrophages have a decreased phagocytic ability. The inhibitor effect of HMGB1 on phagocytosis was attributed to the C-terminal tail, since after it is eliminated, there is a considerable effect reduction and loses its ability to bind to RAGE, which would potentiate its inhibitory effect in phagocytosis.37
In addition to the extracellular effect of HMGB1 proteins in inhibiting phagocytosis, its intracellular role as inhibitor of phagocytosis has been shown in experimental models, through its interaction with the focal adhesion kinase complex (FAK) and Src kinase (FAK/Src), which participates in intracellular signaling allowing for phagocytosis.41 Specifically, Banerjee et al. showed HMGB1 binding to Src kinase of the FAK/Src complex, preventing its interaction with FAK, and simultaneously blocking the intracellular cascade, which results in the inhibition of phagocytosis.42 The macrophages activated through the classical pathway (M1) present a proinflammatory profile with phagocytic activity and production of proinflammatory cytokines such as IL-1 or IL-12, while the alternatively activated macrophages (M2) present an anti-inflammatory profile evidenced by the production of anti-inflammatory cytokines such IL-10 and growth transforming factor beta. Today we know that HMGB1 proteins are capable of inducing the differentiation of M2 macrophages towards an M1 profile, negatively influencing the phagocytosis of apoptotic cells. The latter process could contribute to explain why the circulating monocytes of patients with SLE show a stronger M1 (inflammatory) phenotype, as compared to healthy individuals.43
HMGB1 and NETosisNETosis is a neutrophil defense mechanism aimed at immobilizing and destroying the microorganisms that are too large to phagocytose. The process consists of the development of extracellular traps called NET, containing intracellular components of the neutrophils, such as antimicrobial peptides, histones, nucleosomes, oxygen reactive species, enzymes such as myeloperoxidase, antimicrobial peptide LL-37, and HMGB1 proteins, inter alia, enclosed into a DNA mesh which allows for the immobilization and destruction of the microorganism and the death of the neutrophil. Over the las few years, this defense mechanisms has been associated with the pathophysiology of SLE, since it has been shown that this type of neutrophil cell death may be a source of autoantigens for the development of autoantibodies, due to a deficient clearance of the apoptotic bodies.3,44–46
Usually NET traps degrade by circulating DNasa-1, but the ability to reduce them is impaired in patients with SLE and is associated with the activity of the disease.47 The NETosis process is generated by different stimuli, including pathogens, proinflammatory cytokines, activated platelets, endothelial cells, anti-neutrophil cytoplasmic antibodies, and immune complexes. As a reference, several trials have shown that any impairment in the expression and regulation of the factors involved in NETosis, facilitates the development of SLE. It is noteworthy, that HMGB1 proteins promote the continuous induction of NET through TLR2 and TLR4-dependent mechanisms; for instance, inducing the release of proinflammatory cytokines.3,48 Likewise, Garcia-Romo et al. confirmed that in SLE, HMGB1 proteins rise during NETosis,49 a process in which NADPH oxidase has also been involved.50 Another effect of NETosis is the stimulation of plasmacytoid dendritic cells to produce interferon type I (IFN-α), which plays an important role in the pathogenesis of SLE.45,46
HMBG1 in autoimmune diseasesHMGB1 in patients with SLESince the HMGB1 are elevated in SLE, there is particular interest in establishing the role this cytokine may play in the pathogenesis of the disease, in addition to using the anti-HMGB1 antibodies as markers for disease activity and severity. Furthermore, to consider the possibility of converting this pathway into a therapeutic target. Following is a description of the evidence supporting this argument.4,51–54
HMGB1 proteins may serve as an autoantigen, leading to the production of anti-HMGB1 antibodies.55–57 These antibodies were originally described as a sort of anti-cytoplasm antibodies of “perinuclear” pattern neutrophils in ulcerative colitis.55 However, their presence was later shown in other types of autoimmune diseases such as SLE; so these anti-HMGB1 correlate with HMGB1serum levels, and also with the disease activity measured with the SLE activity index; in contrast, the relationship is inversely proportional with the serum levels of complement proteins C3 and C4.51–53,56 It has also been shown that HMGB1 proteins potentiate the production of antinuclear antibodies that recognize nucleosomes and dsDNA.10,30 Another observation has been that the HMGB1 bound to DNA-containing immune complexes, induce the production of IFN-α by plasmacytoid dendritic cells and the activation of autoreactive B cells, contributing to the pathogenesis of SLE.29
Additionally, the epitope mapping of the anti-HMGB1 antibodies conducted showed that most of them react against the protein,56 indicating that these antibodies do not play a neutralizing role when directed against an antagonistic region. In contrast, in murine models for other diseases with absent anti-HMGB1 antibodies, i.e., sepsis, the administration of box A with its antagonistic effect on HMGB1 levels is beneficial in terms of clinical outcomes.58
Lastly, the redox state plays a key role in HMGB1 functionality. However, not many studies have been conducted on the detection of the redox state of HMGB1 in serum samples of patients with SLE. One of these trials with a small sample of patients showed that oxidized HMGB1 is present in the serum of these patients.59 Hence, further studies are needed, with larger series of patients, in order to determine which HMGB1 isoforms are present in the serum of patients with SLE, and hence be able to establish a clinical association with the presence of these proteins.
HMGB1 in lupus nephritisWhen comparing the serum of patients with SLE, with and without renal involvement, the HMGB1 serum levels are higher in the former, probably indicating that this protein plays a role in the development of LN.52,53 Moreover, kidney biopsies of patients with LN have shown a strong expression of HMGB1 at cytoplasmic and extracellular sites.54,60 However, HMGB1 is detected in the urine of patients with this condition, but not in control subjects, and was positively correlated with serum levels, in addition to proteinuria and the score in the Systemic Lupus Erythematous Activity Index, and was inversely proportional to the C3 complement levels.60 In view of the above, HMGB1 proteins may potentially be used as markers for SLE activity and severity, and in particular, for renal compromise. However, further studies are needed to confirm this association.
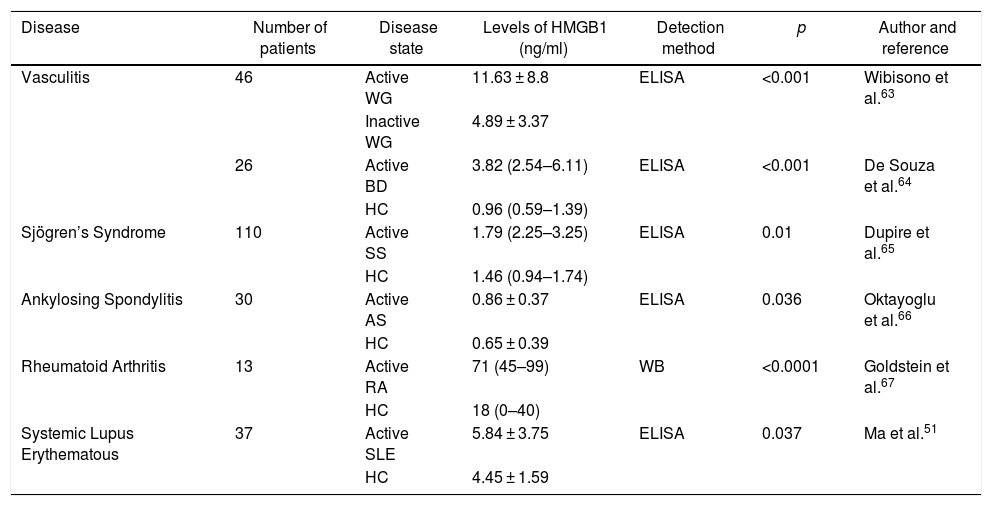
HMGB1 in other autoimmune diseasesHMGB1 may be elevated in other autoimmune diseases such as rheumatoid arthritis, Sjögren’s syndrome and certain types of vasculitis, although the role they may play is not yet clear.53,54,61 A recent study found that patients with antiphospholipid syndrome had significantly increased HMGB1, RAGE and TNF-α serum levels, as compared against healthy controls. This supports the proinflammatory activity of HMGB1 in different autoimmune diseases other than SLE.62Table 1 illustrates the HMBG1 serum levels found in other rheumatic conditions.
Serum levels of HMGB1 in different rheumatic diseases.
| Disease | Number of patients | Disease state | Levels of HMGB1 (ng/ml) | Detection method | p | Author and reference |
|---|---|---|---|---|---|---|
| Vasculitis | 46 | Active WG | 11.63 ± 8.8 | ELISA | <0.001 | Wibisono et al.63 |
| Inactive WG | 4.89 ± 3.37 | |||||
| 26 | Active BD | 3.82 (2.54–6.11) | ELISA | <0.001 | De Souza et al.64 | |
| HC | 0.96 (0.59–1.39) | |||||
| Sjögren’s Syndrome | 110 | Active SS | 1.79 (2.25–3.25) | ELISA | 0.01 | Dupire et al.65 |
| HC | 1.46 (0.94–1.74) | |||||
| Ankylosing Spondylitis | 30 | Active AS | 0.86 ± 0.37 | ELISA | 0.036 | Oktayoglu et al.66 |
| HC | 0.65 ± 0.39 | |||||
| Rheumatoid Arthritis | 13 | Active RA | 71 (45–99) | WB | <0.0001 | Goldstein et al.67 |
| HC | 18 (0–40) | |||||
| Systemic Lupus Erythematous | 37 | Active SLE | 5.84 ± 3.75 | ELISA | 0.037 | Ma et al.51 |
| HC | 4.45 ± 1.59 |
RA: rheumatoid arthritis; HC: healthy controls; AS: ankylosing spondylitis; BD: Behcet disease;
WG: Wegener’s granulomatosis; HMGB1: high mobility group box 1 proteins; SLE: systemic lupus erythematous; SS: Sjögren’s syndrome; WB: Western Blot. The HMGB1 levels are shown as means ± standard deviation or median (range).
HMGB1 are proteins that play an important role as late mediators of inflammation. Just as in other rheumatic diseases, the serum of patients with SLE has shown high levels of HMGB1 and of anti-HMGB1, as compared against patients without SLE. Similarly, the serum, urine, and renal biopsies of patients with LN have shown higher levels of HMGB1. These findings, in addition to the role of HMGB1 proteins in inflammation, lead to a number of questions such as: Is it possible to use HMGB1 as a biomarker of disease activity? Or, Could these proteins be considered as therapeutic targets for future therapies? For the moment, further evidence is required in order to answer these questions.
Conflict of interestsThe authors have no conflict of interests to disclose.
Please cite this article as: Aragón Cristian C, Llanos Julio-Alfredo, Posso-Osorio Iván, Nieto-Aristizábal Ivana, Perea-Cardona Natalia, Ortiz Andrés-Felipe, Tobón Gabriel J. Papel de las proteínas de alta movilidad (HMGB1) en el lupus eritematoso sistémico. Rev Colomb Reumatol. 2020;27:37–45.