Cor pulmonale is the right ventricular dysfunction that results from pulmonary hypertension secondary to alterations of the pulmonary structure and/or function, by various aetiologies, mainly those that affect the parenchyma and pulmonary vasculature. The case is presented of a 62-year-old woman with right ventricular dysfunction secondary to parenchymal and functional pulmonary alterations, as a result of systemic autoimmune disease.
Cor pulmonale es la disfunción ventricular derecha que resulta de hipertensión pulmonar secundaria a alteraciones de la estructura o función pulmonar, por diversas etiologías, principalmente aquellas que afecten el parénquima y la vasculatura pulmonar. Se presenta el caso de una mujer de 62 años de edad, con disfunción ventricular derecha secundaria a alteraciones parenquimatosas y funcionales pulmonares como resultado de enfermedad sistémica autoinmune.
Autoimmune diseases represent a broad spectrum of consequences with multiple effects. In the case of rheumatoid arthritis, there may be up to 41% of pulmonary involvement (parenchyma, pleura, or vasculature). Notwithstanding its frequent presentation, the approach continues to be late, affecting the patient's quality of life and leading to reduced therapeutic options. A case of cor pulmonale secondary to rheumatoid arthritis as a rare complication is discussed.
Clinical observation62-Year-old female patient with a history of rheumatoid arthritis (RA) since she was 20 years old. The patient has been treated from the onset of the disease with prednisone 5mg/day as monotherapy. She was referred from Cartagena for multidisciplinary management due to heart failure and interstitial lung disease (ILD), presenting with around 8 days of dyspnea on mild to low exertion, mMRC 4, edema of the lower extremities, and increased waist circumference, with no other relevant medical history.
At admission the patient required continuous oxygen support, with a tolerance of 1 hour without supplemental oxygen, tachypnea, postural limitation due to breathlessness, jugular engorgement at 90° and hepatojugular reflux, decreased breathing noises, rales, and universal rhonchus, multiple bone malformations of the metacarpophalangeal and interphalangeal joints, with significant ulnar deviation and limitation for flexion and extension with symmetrical involvement, pain and swelling with functional limitation of the elbows and knees. The patient denies ever being followed by rheumatology.
The echocardiogram showed severe pulmonary arterial hypertension (PAH), Pulmonary Artery Systolic Pressure 100mgHg, severely dilated right ventricle, 15% fractional area change, severely diminished systolic function, severely dilated right atrium, and severe dilatation of the inferior vena cava, without inspiratory collapse; the septum, ventricle and left atrium are normal.
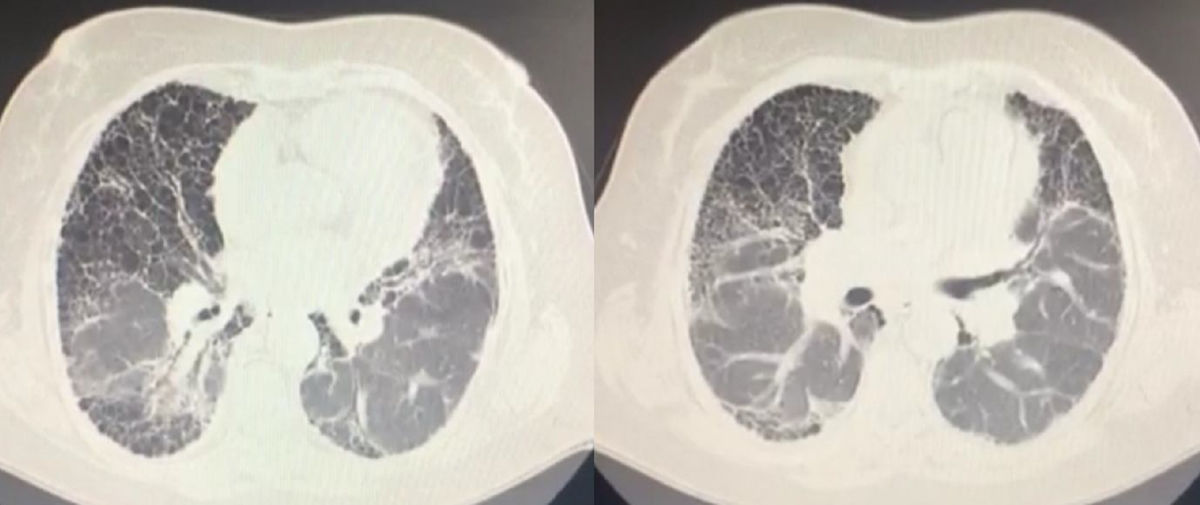
The high resolution chest CT showed a thick reticular lineal pattern, with multiple air-filled cystic spaces ranging from 3 to 10mm in diameter, with a broad distribution and fine walls without any areas of consolidation or masses, with a honeycomb appearance, with apicobasal gradient and traction bronchiectasis (Fig. 1).
Spirometry with severe restriction (FVC, 36%=0.91) was not reversible; pulmonary volumes with air restriction and entrapment, with no improvement upon the administration of a beta-2 adrenergic; carbon monoxide diffusion (DLco) 13%, decreased alveolar volume, decreased DLco/VA ratio. Arterial gases PO2: 56.8mm Hg and DAaO2 of 56mm Hg (estimated: 16mm Hg). The paraclinical tests at admission showed evidence of polyglobulia (hemoglobin 15.8g/dL), ESR 15, CRP 0.68, RA TEST 9.1; the rest of the infectious and immunological profile was within the normal range. The patient was assessed by the department of rheumatology that considered a non-inflammatory sequel condition, currently controlled and with multiple deformities. The decision was to de-escalate corticosteroids and based on the patient's evolution, add a modifier drug because of the significant pulmonary compromise.
Some changes with regard to the usual interstitial pneumonia secondary to the baseline disease are considered. The PHT may be linked to the extensive compromise of the lung parenchyma, and the possibility to include the patient in the transplant list shall be considered in the medium term, since the patient is not a candidate for specific PHT therapy and requires constant oxygen therapy, with significant quality of life limitations, and no possibility of improving the lung function as a result of the fibrotic condition; for these reasons the patient is a candidate for lung transplantation. A biopsy to confirm fibrosis does not represent any additional benefits over the diagnostic images, but increases the risk of complications and the imminent need for invasive mechanical ventilation; Moreover, it is contraindicated because of PHT, current cor pulmonale management, and high risk of cardiovascular complications due to depressed ejection fraction from area change.
DiscussionRA is a chronic inflammatory disease with multiple extra-articular manifestations. In terms of the respiratory system, changes tend to be more frequent as the disease turns chronic following immune imbalance.
The association between ILD and RA was documented more than 60 years ago, reaching up to 33–41%.1 Patients at risk of ILD are mostly elderly men with a history of cigarette smoking.2
The role of modifier-drug therapy is unclear in the progression of EPI.3 Usually the symptoms are associated with dyspnea on exertion, presenting sometimes with PHT, right ventricular dysfunction, and cor pulmonale, as in the case in this discussion.
The lung function evidences restrictive and exchange disorders, with decreased DLco which translates into a reduction of capillary recruitment and hence, of the exchange zone, representing the primary component in hypoxia.4–7 The imaging studies are broad, ranging from beveled glass areas and mostly a peripheral reticular pattern, to traction bronchiectasis and honeycombing.8–9
The mortality is high, with a mean survival of 3.5 years and a 5-year survival of 39%.10 The treatment will depend on the patient's condition and involvement.
ConclusionsThe spectrum of immune diseases is broad, and requires knowledgeable specialists able to adopt a multidisciplinary approach for diagnosis and timely therapy, in order to reduce the number of lesions and morbidity to ensure the best possible quality of life.
Conflicts of interestThe authors have no conflicts of interest to disclose.
We want to extend our gratitude to Clínica General del Norte for their contribution to medical training.
Please cite this article as: Castellanos-Gutiérrez MÁ, Maestre-Serrano R, Santiago-Henríquez E. Manifestaciones pulmonares de artritis reumatoide: cor pulmonale. Rev Colomb Reumatol. 2019;26:129–131.