To determine the risk factors and the potential relationship between body mass index (BMI) and the development of interstitial lung disease (ILD) in patients with systemic sclerosis.
MethodsA cross-sectional, correlational, study with a non-probabilistic sample was conducted on consecutive patients with scleroderma seen in the Rheumatology Division at Hospital Universitario de Caracas during the period April to September 2018. Patients underwent clinical and laboratory assessments, anthropometric measurements, pulmonary function tests, and high-resolution computed axial tomography scan of the chest.
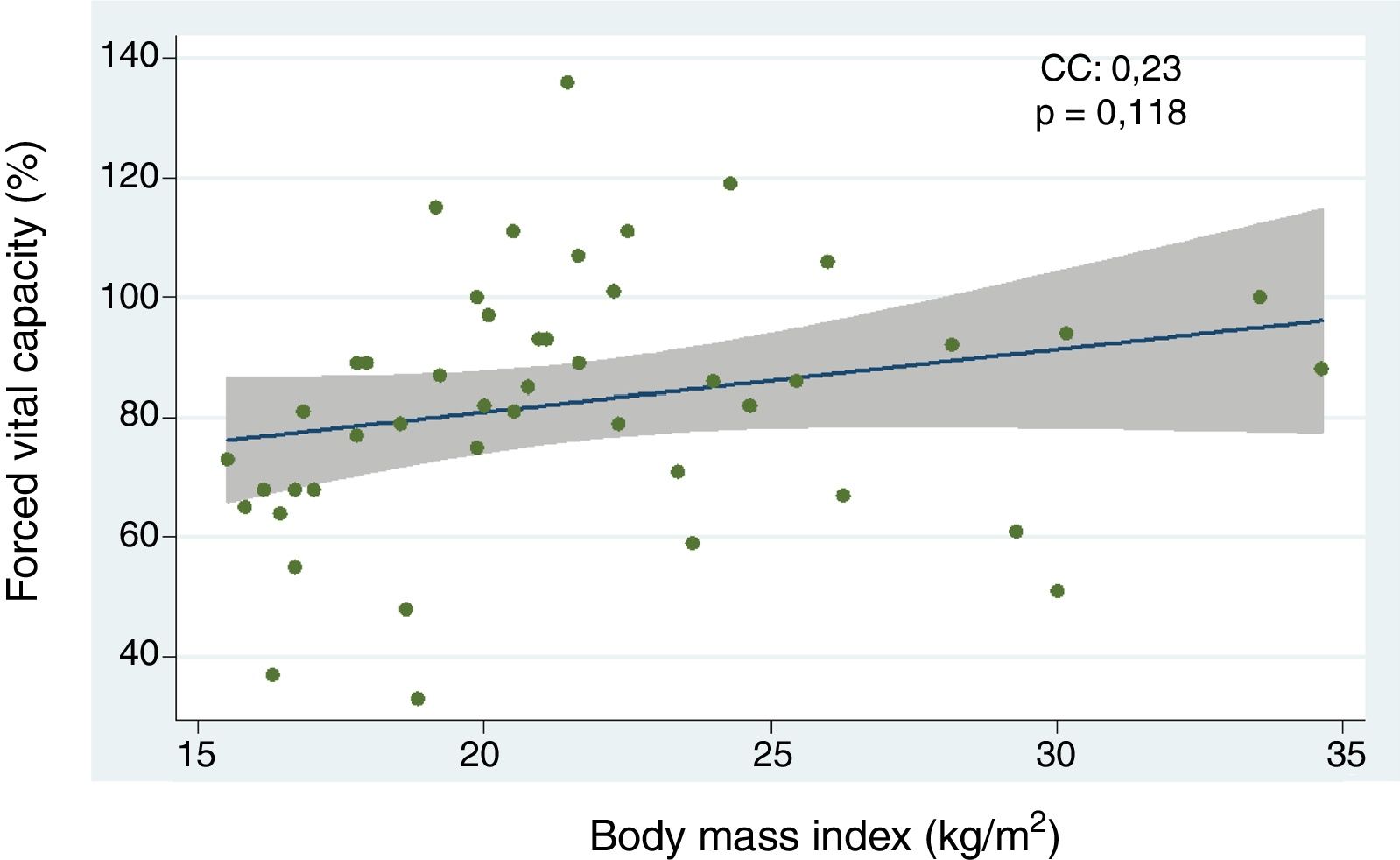
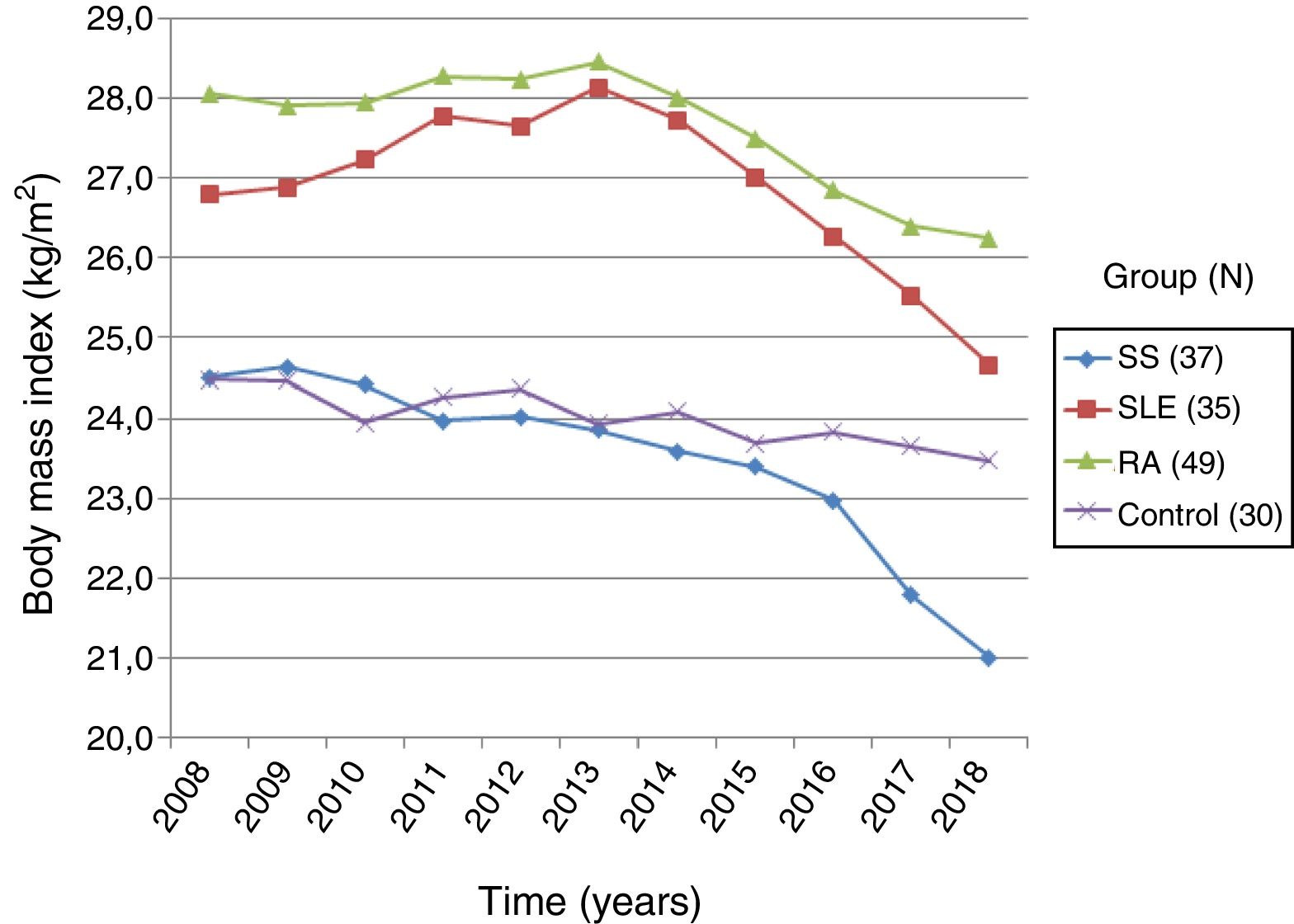
ResultsOf 48 patients, 95.8% were female, with a high prevalence of limited cutaneous systemic sclerosis (lcSSc) (64.6%). Underweight (BMI<18.5kg/m2) was a risk factor for the development of ILD (OR 4.60; 95% CI 1.12–18.86). There was directly proportional relationship between BMI and forced vital capacity, with no statistical significance (CC 0.23; p=0.1180). However, other predictors of ILD included diffuse systemic sclerosis (OR 6.91, 95% CI; 1.78–26.85), modified Rodnan skin score (mRSS) (OR 1.25; 95% CI; 1.04–1.30), and a speckled pattern of antinuclear antibody (OR 5.62; 95% CI; 1.33–23.62). Multivariate analysis showed that mRSS (OR 1.34; 95% CI; 1.04–1.72) and the presence of a speckled antinuclear antibody (OR 25.99; 95% CI; 1.02–16.02) were independent predictors of ILD. The percentage of BMI lost in a period of time of 2008–2018 was 14.3% (p=0.0021), which was higher than rheumatoid arthritis (p=0.0000), systemic lupus erythematosus (p=0.0025), and healthy patients (p=0.0331).
ConclusionBMI and ILD are inversely correlated. Age, underweight, higher mRSS, and speckled pattern of antinuclear antibody were risk factors for development of ILD in patients with systemic sclerosis. The mRSS could be used as a clinical predictive tool for ILD in systemic sclerosis.
Determinar los factores de riesgo y la asociación entre el índice de masa corporal (IMC) y enfermedad pulmonar intersticial difusa (EPID) en pacientes con esclerosis sistémica.
MétodosEstudio transversal, correlacional, con muestra no probabilística constituida por pacientes consecutivos con esclerosis sistémica que acudieron al Servicio de Reumatología del Hospital Universitario de Caracas durante el período abril-septiembre de 2018. Se realizó evaluación clínica, paraclínica, mediciones antropométricas, pruebas de función pulmonar y tomografía de tórax de alta resolución.
ResultadosDe 48 pacientes, el 95.8% fueron del sexo femenino, con mayor prevalencia de esclerosis sistémica limitada (64.6%). Un peso bajo (IMC<18.5kg/m2) fue un factor de riesgo de EPID (OR 4.60; IC 95% 1.12-18.86). El IMC se relacionó de forma directamente proporcional con la capacidad vital forzada sin alcanzar significación estadística (CC 0.23; p=0.1180), sin embargo, otros predictores de EPID fueron: la esclerosis sistémica difusa (OR 6.91; IC 95% 1.78-26.85), el índice modificado de Rodnan (mRSS) (OR 1,25; IC 95% 1.04-1.30) y el patrón moteado de anticuerpos antinucleares (OR 5,62; IC 95% 1.33-23.62). El análisis multivariado demostró asociación independiente con EPID de: mRSS (OR 1.34; IC 95% 1.04-1.72) y el patrón moteado de anticuerpos antinucleares (OR 25.99; IC 95% 1.02-16.02). El estudio del IMC durante 10 años (2008-2018) reveló una pérdida de 14.3% (p=0.0021), que fue mayor en comparación con pacientes con artritis reumatoide (p=0.0000), lupus eritematoso sistémico (p=0.0025) e individuos sanos (p=0.0331).
ConclusiónEl IMC y la EPID se correlacionan de forma inversamente proporcional. La edad, un bajo peso corporal, un puntaje alto en mRSS y el patrón moteado de anticuerpos antinucleares fueron factores de riesgo para el desarrollo de EPID en pacientes con esclerosis sistémica. El mRSS podría ser utilizado como herramienta clínica predictiva de EPID en esclerosis sistémica.
Systemic sclerosis is a systemic inflammatory autoimmune disease, characterized by skin and organ fibrosis that may result in varying levels of severity in systems such as the GI tract, lungs, heart, and kidneys. While the characteristic hallmark is skin fibrosis, vital organs impairment is what reduces the life expectancy and quality of life of patients.1,2 Lung involvement in systemic sclerosis may affect up to 80% of the cases and is one of the major factors affecting the morbidity and mortality in this disease.3,4
The pathogenesis of the fibrotic process is still being investigated. Recent research highlights the role of the adipocyte and the fact that it is replaced by myofibroblasts in experimental murine systemic sclerosis models.5–8 These studies indicate that the myofibroblast is the major fibrogenic inductor by promoting excessive fibrillar collagen deposition and other extracellular matrix proteins in the dermis. Its origin in fibrotic tissue may be accounted for by a process called adipocyte-myofibroblast transition, observed in bleomycin-induced systemic sclerosis in mice, in which fluorochrome-labeled adipocytes underwent a differentiation process into myofibroblasts when influenced by transforming growth factor β (TGF-β). Adipocytes lost their phenotypical characteristics and developed fibroblast-like characteristics, preserving the marker after the transition was over.8 This process if further enhanced by the loss of the repair and protective properties of the adipose tissue on the dermis.9,10
Lung fibrosis is one of the principal causes of morbidity and mortality in systemic sclerosis.11–13 This architectural disruption of the lung parenchyma in systemic sclerosis corresponds to a diffuse interstitial lung disease (ILD) and may be present in up to 80–90% of the patients.1,3 Some lung fibrosis studies in murine models also show changes in the phenotype of the lung fibroblasts from lipogenic to myogenic during the fibrosis process.14,15 This process involves a certain type of fibroblast identified in the lung parenchyma of rodents, called lung lipofibroblast, which is responsible for alveolar maturation and the production of lung surfactant. These lipofibroblasts under the effects of profibrotic mediators such as TGF-β differentiate into myofibroblasts.14,16 This is a reversible process through the action of the peroxisome proliferator-activated receptor γ (PPARγ), the factor responsible for adipocyte differentiation.
The dermal white adipose tissue (dWAT) is a particular adipose tissue in the skin and is different from the subcutaneous adipose tissue and its loss is characteristic of systemic sclerosis in humans and experimental mice.9,17,18 The dWAT has repair, renewing and angiogenic properties on the dermis and its depletion precedes the onset of fibroblast activation and its differentiation into myofibroblast. A fat-rich diet or PPARγ stimulation are both responsible for the proliferation of this adipose tissue fraction.18,19 In fact, the negative regulation of the PPARγ pathway is associated with accelerated fibrogenesis and a higher modified Rodnan score, a clinical tool for estimating the level of dermal fibrosis is systemic sclerosis.20
Weight loss is a major health and disease predictor in the general population. The prevalence of malnutrition in patients with systemic sclerosis varies between 15% and 55.7% of the cases,21,22 and there are other causes involved in addition to intestinal malabsorption, such as musculoskeletal disease and mood disorders leading to decreased appetite. Moreover, studies on the relationship between body mass index (BMI) and the development of pneumopathy indicate that an optimal nutritional status favors the respiratory system and represents a good postoperative prognosis of patients undergoing lung transplantation.23,24 A causal relationship has also been established between low BMI and increased respiratory mortality in inflammatory and infectious diseases.24–26
A disrupted adipose tissue homeostasis in systemic sclerosis translates into decreased integumentary fat mass and the onset of pulmonary fibrosis, which clinically manifests with weight loss and respiratory symptoms, respectively. It is yet unclear if the BMI reduction observed in these patients is associated with the loss of fat mass, and the latter with increased differentiation of lung lipo-fibroblasts into myofibroblasts, which could contribute to the development of ILD in systemic sclerosis.
In accordance with the data collected from the National Center for Rheumatic Diseases of Venezuela (Centro Nacional de Enfermedades Reumáticas), the annual incidence of systemic sclerosis has been estimated at one case per one million inhabitants (unpublished data). This number may underestimate the actual incidence of the disease in our country, since it is based on information from one single center.
It is well known that up to 90% of the patients with systemic sclerosis present varying degrees of interstitial lung changes, and 40 to 75% of them exhibit altered pulmonary function tests.3 Since systemic sclerosis is a multi-system autoimmune disease that often leads to ILD, and also impacts the patient's body weight in a way directly proportional to the severity of the disease, it is then quite important to study this potential association. All of the above leads to the following question: Is there a relationship between BMI and the development of ILD in patients with systemic sclerosis?
In order to answer this question, a research project was conducted in patients with systemic sclerosis that were being treated at the Rheumatology unit of the Hospital Clínico Universitario de Caracas, between April and September, 2018.
MethodsA cross-sectional, correlational study was conducted. The study population included patients with a diagnosis of systemic sclerosis treated at the Rheumatology clinic of Hospital Universitario de Caracas, between April and September, 2018. The sample was non-probabilistic and circumstantial, and the following inclusion criteria were used: patients with a diagnosis of systemic sclerosis based on the classification criteria of the American College of Rheumatology and the 2013 European League Against Rheumatism, over 18-years old, both males and females, who had signed an informed consent. Patients with other concurrent autoimmune conditions were excluded, as well as patients with any of the following pre-existing chronic lung diseases: emphysema, asthma, chronic obstructive pulmonary disease, pneumoconiosis and sarcoidosis; patients with any of the following co-existing endocrine conditions: hyperthyroidism, hypothyroidism, hypoadrenalism and diabetes mellitus, as well patients with cancer.
A demographic and clinical assessment was conducted to collect socio-economic information, physical examination, anthropometric indexes, pulmonary function tests, high-resolution chest CT (HRCT) and autoantibodies profile. The patients were assessed by a rheumatologist who completed the data sheet with socio-economic information, clinical characteristics of the disease, anthropometric data and laboratory tests.
Body measurements recordEach patient's medical record was reviewed in order to establish the evolution of body weight, height, and BMI from the first outpatient visit to the Rheumatology unit, until the completion of the study. The actual bodyweight measurement used a standard stadiometer.
Modified Rodnan skin score [mRSS]The score was measured during the physical examination of the patients. Two trained rheumatologists assessed the patients using the mRSS in a room with adequate lighting and controlled temperature. The skin thickness was assessed in 17 body areas distributed over the face, trunk, and extremities, by forming a skin fold with the thumb and index fingers. Based on the findings, the following scores were assigned: 0=no thickening, 1=mild thickening, 2=moderate thickening, and 3=severe thickening preventing the formation of a skin fold.20 Each patient's assessment was collected in a data sheet, and the estimated total score was the sum of all the scores in the different body areas.
Lung disease examinationBased on the forced vital capacity measured with spirometry, the six-minute walk (6MW) and the chest HRCT. The spirometry was done in a controlled-temperature environment, with calibrated equipment and adjusted sensors. The patient was educated about the procedure and was asked to avoid intensive exercising and smoking prior to the test. The patient was placed in a fixed chair with arms rest, sitting straight and with the head slightly elevated; a mouthpiece and nasal forceps were used. The patient was asked to take a deep breath and then exhale forcefully and sustained. The values obtained were recorded in the datasheet.
The 6-minute walk was conducted in an obstacle-free area, in a flat surface with clearly demarcated start and end points stretching over a 30-m distance. The patient was wearing comfortable workout cloths and had not been engaged in vigorous physical activity 2h prior to the test. The patient's weight and height were recorded, as well as baseline blood pressure and resting heart rate. A programmed 6-min stop watch was used and the patient walked for 6min but was allowed to stop at any point if needed to take a break. The instructions to interrupt the test were: chest pain, intolerable dyspnea, sweating, pallor, paresthesia, pulse oximetry below 80% or at the patient's request. At the end of the 6min, the number of rounds and meters walked were recorded.27 The reference value for the 6-M walk in patients with systemic sclerosis is estimated between 465 and 480m.28 Less than 465m was interpreted as an altered test. The test was conducted at the laboratory of the Pulmonology Department of the Caracas University Hospital.
Control groupUsing the medical record, the BMI of the patients with autoimmune disease and of a group of healthy controls was recorded. Three groups were set up: the first group was made up by healthy individuals matched by age, gender and family background; the second group included patients with systemic lupus erythematous (SLE), and the third group comprised patients with rheumatoid arthritis (RA).
The study complied with the guidelines of the Declaration of Helsinki and had the approval of the Ethics Committee of the Caracas University Hospital. Upon admitting the patients to the trial, they were informed about the objectives of the trial, its non-interventional and harmless nature, and were asked to sign their informed consent.
Statistical analysisThe average and the standard deviation of the continuous variables were estimated. In the case of the nominal variables, the frequency and the percentages were estimated. The Student-t test was used to compare the quantitative variables with normal distribution and Mann–Whitney's U test in the absence of a normal distribution. The categorical variables were compared using Chi square or Fisher's test. To compare the means of the ordinal variables, a variance analysis was used (ANOVA). The dependent variables were FVC measured with spirometry, the 6-M walk value and the high resolution chest CT. The independent variable was BMI. The following co-variables were taken into consideration: age, gender, age of onset of the disease, duration of the disease, smoking, educational level, socio-economic level, subtype of systemic sclerosis, mRSS score, and the presence of anti-Scl70. Using a univariate logistic regression analysis, the odds ratio of the independent variables was estimated. Then a multivariate logistic regression analysis of the regression variables was conducted, with predetermined statistical significance, using the backward stepwise regression technique. A p value <0.05 was considered significant.
The inter-observer variability of 2 trained rheumatologists who estimated the mRSS score was determined, using Cohen's kappa coefficient (concordance power), which was considered weak if (<0.40); moderate (0.40–0.60); good (0.60–0.80) or excellent (0.80–1). The statistical analysis used the Stata 12.0 software for Windows (StataCorp LP, TX, USA).
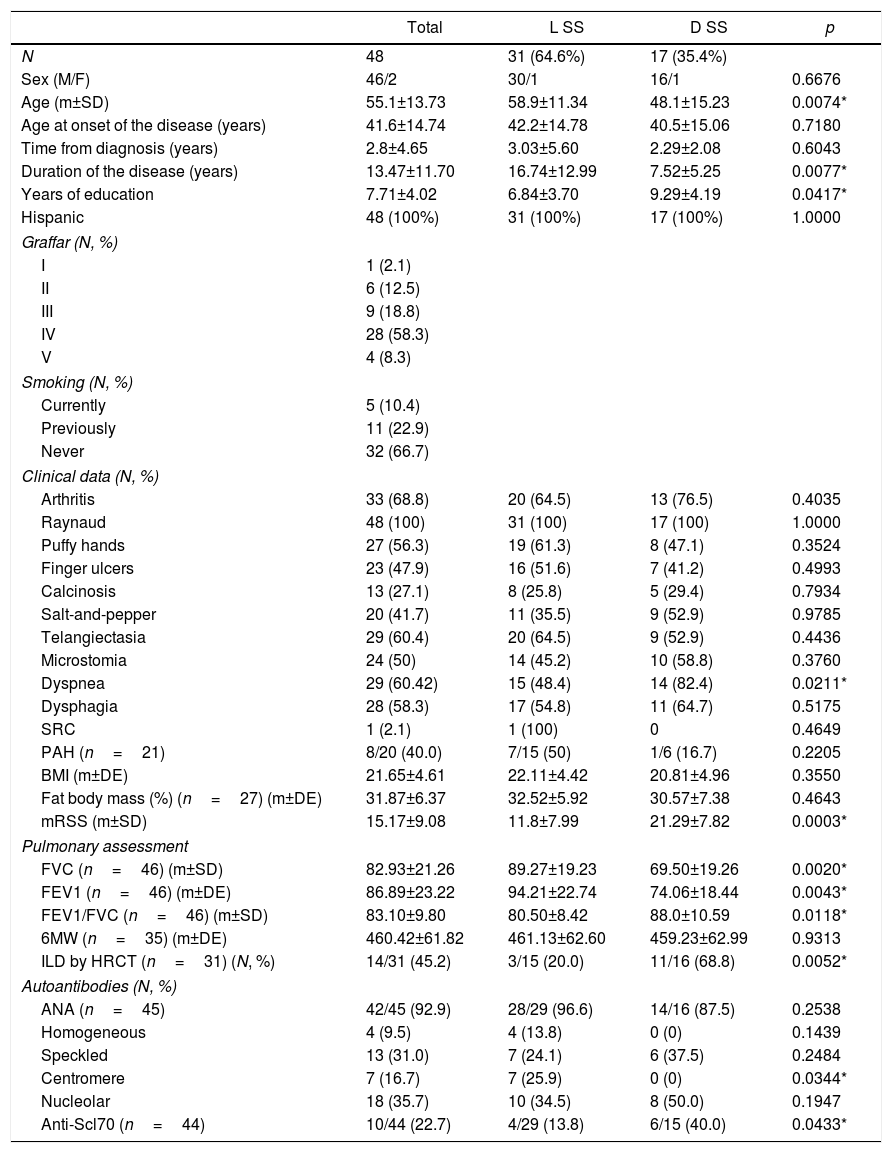
ResultsDemographic and clinical findings48 patients with systemic sclerosis were assessed, of which 46 (95.8%) were females. The mean age was 55.1±13.73 years and all were Hispanics. The most prevalent systemic sclerosis subtype was limited cutaneous (64,6%), while diffuse cutaneous was 35.4%. The mean duration of the disease was 13.47±11.70 years, but was significantly longer in the limited systemic sclerosis group (16.74±12.99 vs. 7.52±5.25; p=0.0077). With regards to the level of education measured in years, the mean was 7.71±4.02 years; the patients with limited systemic sclerosis had less education and the difference was statistically significant: 6.84±3.70 vs. 9.29±4.19 (p=0.0417). When assessing the patients using the Graffar scale, the prevailing socio-economic level was IV (58.3%), followed by III (18.8%), II (12.5%), V (8.3%) and I (2.1%). Thirty-two patients (66.7%) denied any smoking habits, while 11 (22.9%) declared having a history of smoking in the past, and finally 5 (10.4%) were active smokers at the time of the study (Table 1).
Clinical and demographic characteristics of patients with systemic sclerosis.
| Total | L SS | D SS | p | |
|---|---|---|---|---|
| N | 48 | 31 (64.6%) | 17 (35.4%) | |
| Sex (M/F) | 46/2 | 30/1 | 16/1 | 0.6676 |
| Age (m±SD) | 55.1±13.73 | 58.9±11.34 | 48.1±15.23 | 0.0074* |
| Age at onset of the disease (years) | 41.6±14.74 | 42.2±14.78 | 40.5±15.06 | 0.7180 |
| Time from diagnosis (years) | 2.8±4.65 | 3.03±5.60 | 2.29±2.08 | 0.6043 |
| Duration of the disease (years) | 13.47±11.70 | 16.74±12.99 | 7.52±5.25 | 0.0077* |
| Years of education | 7.71±4.02 | 6.84±3.70 | 9.29±4.19 | 0.0417* |
| Hispanic | 48 (100%) | 31 (100%) | 17 (100%) | 1.0000 |
| Graffar (N, %) | ||||
| I | 1 (2.1) | |||
| II | 6 (12.5) | |||
| III | 9 (18.8) | |||
| IV | 28 (58.3) | |||
| V | 4 (8.3) | |||
| Smoking (N, %) | ||||
| Currently | 5 (10.4) | |||
| Previously | 11 (22.9) | |||
| Never | 32 (66.7) | |||
| Clinical data (N, %) | ||||
| Arthritis | 33 (68.8) | 20 (64.5) | 13 (76.5) | 0.4035 |
| Raynaud | 48 (100) | 31 (100) | 17 (100) | 1.0000 |
| Puffy hands | 27 (56.3) | 19 (61.3) | 8 (47.1) | 0.3524 |
| Finger ulcers | 23 (47.9) | 16 (51.6) | 7 (41.2) | 0.4993 |
| Calcinosis | 13 (27.1) | 8 (25.8) | 5 (29.4) | 0.7934 |
| Salt-and-pepper | 20 (41.7) | 11 (35.5) | 9 (52.9) | 0.9785 |
| Telangiectasia | 29 (60.4) | 20 (64.5) | 9 (52.9) | 0.4436 |
| Microstomia | 24 (50) | 14 (45.2) | 10 (58.8) | 0.3760 |
| Dyspnea | 29 (60.42) | 15 (48.4) | 14 (82.4) | 0.0211* |
| Dysphagia | 28 (58.3) | 17 (54.8) | 11 (64.7) | 0.5175 |
| SRC | 1 (2.1) | 1 (100) | 0 | 0.4649 |
| PAH (n=21) | 8/20 (40.0) | 7/15 (50) | 1/6 (16.7) | 0.2205 |
| BMI (m±DE) | 21.65±4.61 | 22.11±4.42 | 20.81±4.96 | 0.3550 |
| Fat body mass (%) (n=27) (m±DE) | 31.87±6.37 | 32.52±5.92 | 30.57±7.38 | 0.4643 |
| mRSS (m±SD) | 15.17±9.08 | 11.8±7.99 | 21.29±7.82 | 0.0003* |
| Pulmonary assessment | ||||
| FVC (n=46) (m±SD) | 82.93±21.26 | 89.27±19.23 | 69.50±19.26 | 0.0020* |
| FEV1 (n=46) (m±DE) | 86.89±23.22 | 94.21±22.74 | 74.06±18.44 | 0.0043* |
| FEV1/FVC (n=46) (m±SD) | 83.10±9.80 | 80.50±8.42 | 88.0±10.59 | 0.0118* |
| 6MW (n=35) (m±DE) | 460.42±61.82 | 461.13±62.60 | 459.23±62.99 | 0.9313 |
| ILD by HRCT (n=31) (N, %) | 14/31 (45.2) | 3/15 (20.0) | 11/16 (68.8) | 0.0052* |
| Autoantibodies (N, %) | ||||
| ANA (n=45) | 42/45 (92.9) | 28/29 (96.6) | 14/16 (87.5) | 0.2538 |
| Homogeneous | 4 (9.5) | 4 (13.8) | 0 (0) | 0.1439 |
| Speckled | 13 (31.0) | 7 (24.1) | 6 (37.5) | 0.2484 |
| Centromere | 7 (16.7) | 7 (25.9) | 0 (0) | 0.0344* |
| Nucleolar | 18 (35.7) | 10 (34.5) | 8 (50.0) | 0.1947 |
| Anti-Scl70 (n=44) | 10/44 (22.7) | 4/29 (13.8) | 6/15 (40.0) | 0.0433* |
Data presented as means (±standard deviation). To establish the statistical differences among groups, Student t and Chi square were used as appropriate.
ANA: antinuclear antibodies detected through indirect immunofluorescence with HEp-2 cell substrate; Anti-Scl70: anti-topoisomerase antibody through enzyme-immunoassay; SRC: scleroderma renal crisis; FVC: forced vital capacity; 6MW: 6-minute walk; ILD: diffuse interstitial lung disease; D SS: diffuse systemic sclerosis; L SS: limited systemic sclerosis; FEV1: forced expiratory volume in the first second; PAH: pulmonary arterial hypertension; BMI: body mass index; mRSS: modified Rodnan skin score; HRCT: high resolution computed tomography.
In terms of the level of skin fibrosis, estimated with mRSS, the mean was 15.17±9.08 points, with higher severity in patients with diffuse scleroderma: 21.29±7.82 vs. 11.8±7.99, with a statistically significant difference (p=0.0003). The interobserver variability of mRSS between the two rheumatologists that assessed the patients with systemic sclerosis, was estimated using the Kappa coefficient, reporting a value of 0.64 (good concordance). Raynaud's phenomenon was present in 100% of the patients from both groups of systemic sclerosis. The most frequent clinical signs of limited systemic sclerosis were: arthritis (64.5%), telangiectasia (64.5%), and puffy hands (61.3%), while in diffuse systemic sclerosis the following signs prevailed: dyspnea (82.4%), arthritis (76.5%) and dysphagia (64.7%). Dyspnea showed a statistically significant difference between the subtypes of systemic sclerosis (p=0.0211) (Table 1).
Antinuclear antibodies (ANA) were present in 92.9% of the patients. In 22.7% of the patients, anti-topoisomerase I (anti-Scl70) antibodies were also identified, 40% in the diffuse systemic sclerosis group and 13.8% in the limited scleroderma patients, with limited statistical significance (p=0.0433). Anti-centromere antibodies were detected in 16.7% of the patients, all of them in the limited phenotype group, with a statistically significant difference versus the diffuse phenotype (p=0.0344) (Table 1).
The spirometry results of the total sample of patients were: FVC, 82.93%±21.26 of the average value and FEV1: 86.89%±23.22 of the average value, respectively. Patients with diffuse systemic sclerosis reported lover FVC levels (69.50±19.26 vs. 89.27±19.23) and FEV1 (74.06±18.44 vs. 94.21±22.74) as compared to patients with limited scleroderma, observing a significant difference for both parameters between the 2 subgroups (p=0.0020 and p=0.0043, respectively). The 6-minute walk reported an average of 460.42±61.82m. Patients with diffuse systemic sclerosis walked less meters in the 6-minute walk (459.23±62.99) as compared to the limited systemic sclerosis group (461.13±62.60); however there was no statistically significant difference between the two groups. In accordance with the HRCT of the chest conducted in 31 patients, 45.2% presented diffuse ILD, of which 11 patients belonged to the diffuse scleroderma group and 3 patients to the limited scleroderma group, representing 68.8% and 20.0% of each group, respectively, with a statistically significant difference (p=0.0052) (Table 1).
The mean BMI in systemic sclerosis was 21.65±4.61kg/m2, and it was lower in the diffuse subtype (20.81±4.96 vs. 22.11±4.42), with no statistically significant difference between the 2 subgroups (Table 1). With regards to the BMI-based nutritional status according to the World Health Organization (WHO) criteria, 25% of the patients had low weight, 56.3% had an optimal weight, 10.4% were overweight and 8.3% were obese. Low weight was observed in 16.7% of the patients with diffuse systemic sclerosis and 8.33% of the limited subtype, with statistical significance (p=0.0082).
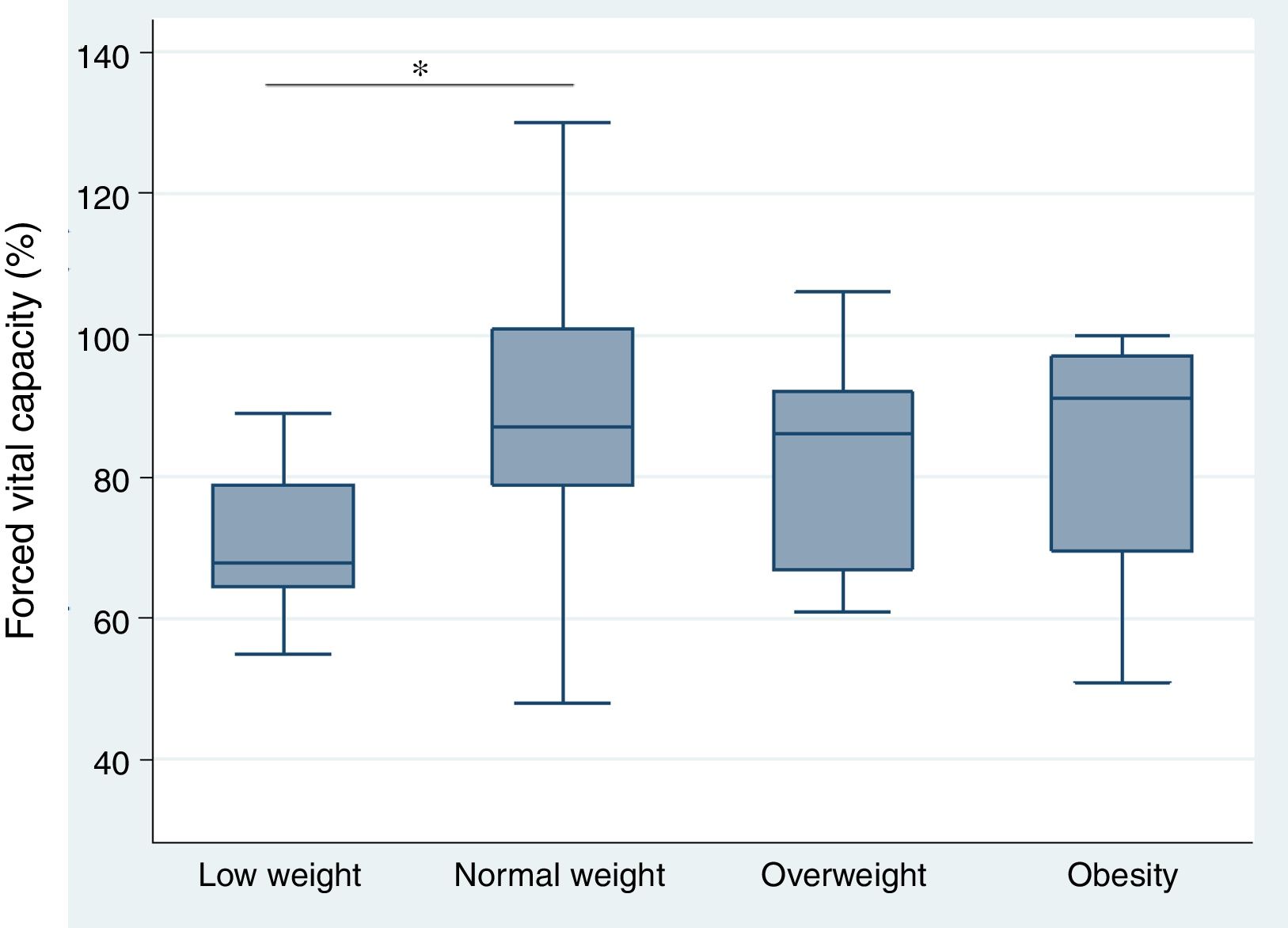
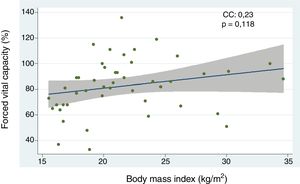
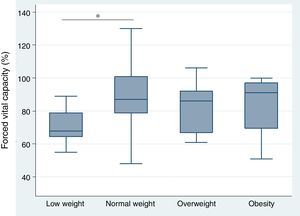
Relationship between body mass index and interstitial lung disease, based on forced vital capacity, 6-minute walk, and high resolution computed tomography of the chestThe analysis of the correlation between FVC and BMI showed a tendency toward a directly proportional relationship (CC=0.23) not reaching statistical significance (p=0.118) (Fig. 1). When assessing the correlation between BMI and the 6-minute walk and HRCT of the chest, the relationship for each comparison was inversely proportional and was not statistically significant in both cases (CC=−0.09 and −0.07, p=0.571 and p=0.672, respectively). Furthermore, the BMI analysis by groups, based on WHO criteria, showed that the low weight group of patients had lower FVC levels as compared to the optimum weight group, with a statistically significant difference (p=0.046) (Fig. 2).
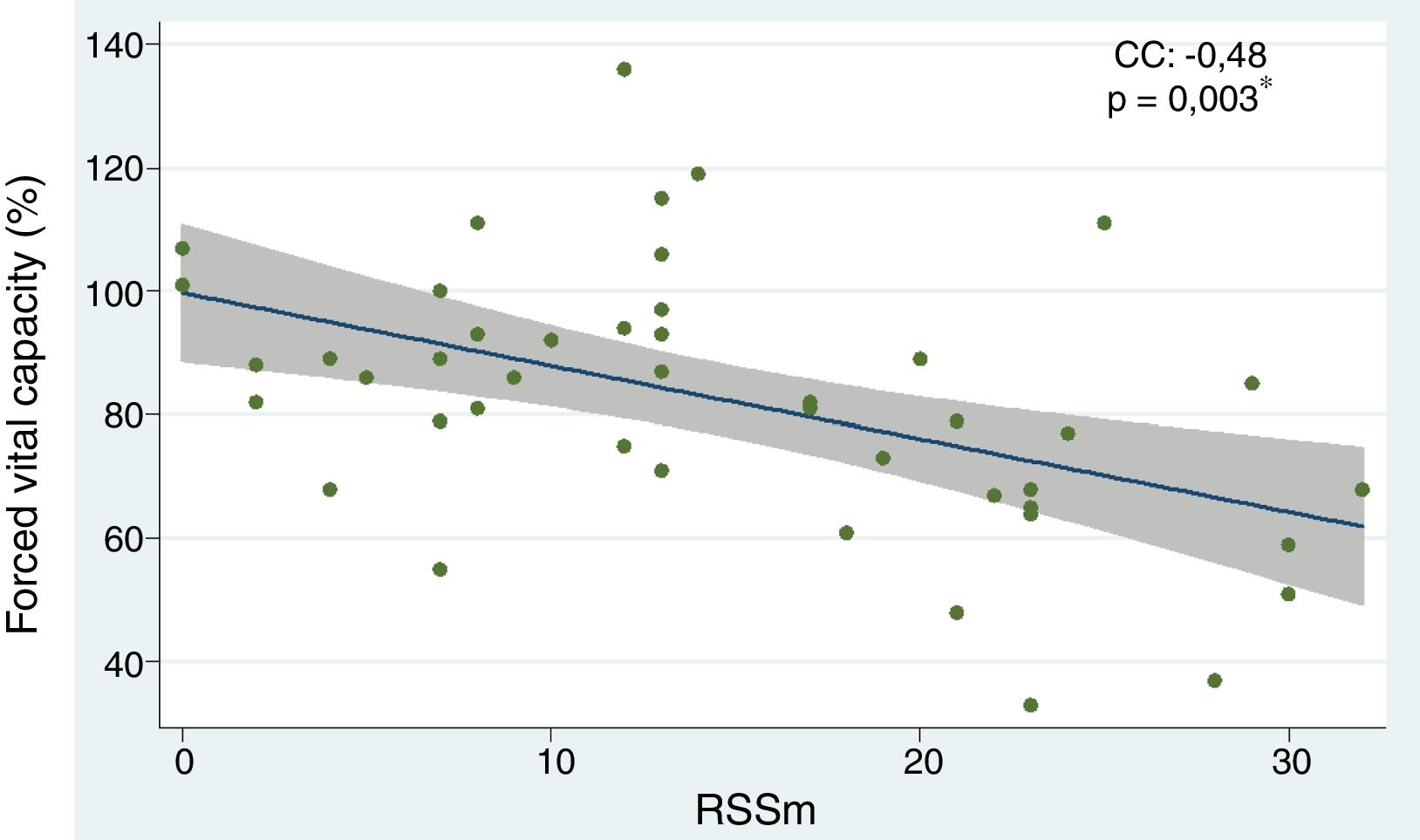
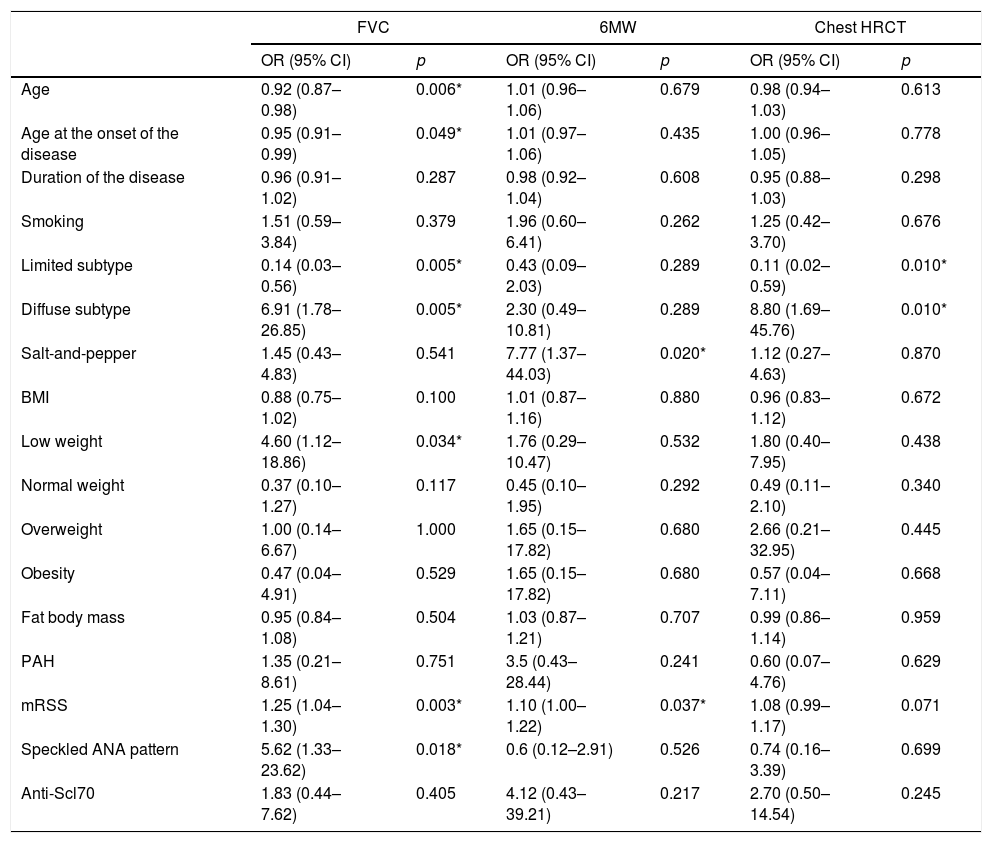
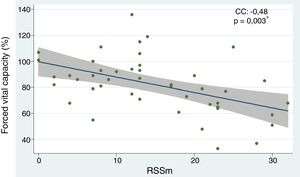
The independent predictors of interstitial lung disease based on a reduction in FVC were: the subtype of diffuse systemic sclerosis (OR 6.91; 95% CI 1.78–26.85; p=0.005), having low weight (OR 4.60; 95% CI 1.12–18.86; p=0.034), the presence of speckled pattern ANA (OR 5.62; 95% CI 1.33–23.62; p=0.018) and the mRSS scale (OR 1.25; 95% CI 1.04–1.30; p=0.003). The following factors showed a protective effect: age (OR 0.92; 95% CI 0.87–0.98; p=0.006), age of onset of the disease (OR 0.95; 95% CI 0.91–0.99; p=0.049), and the limited subtype (OR 0.14; 95% CI 0.03–0.56; p=0.005). When the risk factors that disrupted the results of the 6-minute walk were assessed, the following were considered negative predictors: “salt-and-pepper” skin lesions (OR 7.77; 95% CI 1.37–44.03; p=0.020) and mRSS (OR 1.10; 95% CI 1.00–1.22; p=0.037). With regards to the chest HRCT, the diffuse subtype behaved as a risk factor for ILD (OR 8.80; 95% CI 1.69–45.76; p=0.010) (Table 2). The correlation analysis between FVC and mRSS showed an inversely proportional tendency with statistical significance (CC=−0.48; p=0.003) (Fig. 3).
Univariate analysis of predictors of diffuse interstitial lung disease in patients with systemic sclerosis.
| FVC | 6MW | Chest HRCT | ||||
|---|---|---|---|---|---|---|
| OR (95% CI) | p | OR (95% CI) | p | OR (95% CI) | p | |
| Age | 0.92 (0.87–0.98) | 0.006* | 1.01 (0.96–1.06) | 0.679 | 0.98 (0.94–1.03) | 0.613 |
| Age at the onset of the disease | 0.95 (0.91–0.99) | 0.049* | 1.01 (0.97–1.06) | 0.435 | 1.00 (0.96–1.05) | 0.778 |
| Duration of the disease | 0.96 (0.91–1.02) | 0.287 | 0.98 (0.92–1.04) | 0.608 | 0.95 (0.88–1.03) | 0.298 |
| Smoking | 1.51 (0.59–3.84) | 0.379 | 1.96 (0.60–6.41) | 0.262 | 1.25 (0.42–3.70) | 0.676 |
| Limited subtype | 0.14 (0.03–0.56) | 0.005* | 0.43 (0.09–2.03) | 0.289 | 0.11 (0.02–0.59) | 0.010* |
| Diffuse subtype | 6.91 (1.78–26.85) | 0.005* | 2.30 (0.49–10.81) | 0.289 | 8.80 (1.69–45.76) | 0.010* |
| Salt-and-pepper | 1.45 (0.43–4.83) | 0.541 | 7.77 (1.37–44.03) | 0.020* | 1.12 (0.27–4.63) | 0.870 |
| BMI | 0.88 (0.75–1.02) | 0.100 | 1.01 (0.87–1.16) | 0.880 | 0.96 (0.83–1.12) | 0.672 |
| Low weight | 4.60 (1.12–18.86) | 0.034* | 1.76 (0.29–10.47) | 0.532 | 1.80 (0.40–7.95) | 0.438 |
| Normal weight | 0.37 (0.10–1.27) | 0.117 | 0.45 (0.10–1.95) | 0.292 | 0.49 (0.11–2.10) | 0.340 |
| Overweight | 1.00 (0.14–6.67) | 1.000 | 1.65 (0.15–17.82) | 0.680 | 2.66 (0.21–32.95) | 0.445 |
| Obesity | 0.47 (0.04–4.91) | 0.529 | 1.65 (0.15–17.82) | 0.680 | 0.57 (0.04–7.11) | 0.668 |
| Fat body mass | 0.95 (0.84–1.08) | 0.504 | 1.03 (0.87–1.21) | 0.707 | 0.99 (0.86–1.14) | 0.959 |
| PAH | 1.35 (0.21–8.61) | 0.751 | 3.5 (0.43–28.44) | 0.241 | 0.60 (0.07–4.76) | 0.629 |
| mRSS | 1.25 (1.04–1.30) | 0.003* | 1.10 (1.00–1.22) | 0.037* | 1.08 (0.99–1.17) | 0.071 |
| Speckled ANA pattern | 5.62 (1.33–23.62) | 0.018* | 0.6 (0.12–2.91) | 0.526 | 0.74 (0.16–3.39) | 0.699 |
| Anti-Scl70 | 1.83 (0.44–7.62) | 0.405 | 4.12 (0.43–39.21) | 0.217 | 2.70 (0.50–14.54) | 0.245 |
To calculate the OR and 95% confidence interval, the logistic regression analysis was used.
ANA: antinuclear antibodies; anti-Scl70: anti-topoisomerase antibody; FVC: forced vital capacity; 6MW: 6-minute walk; PHP: pulmonary arterial hypertension; CI: confidence interval; BMI: body mass index; mRSS: modified Rodnan skin score; OR: odds ratio; HRCT: high resolution computed tomography.
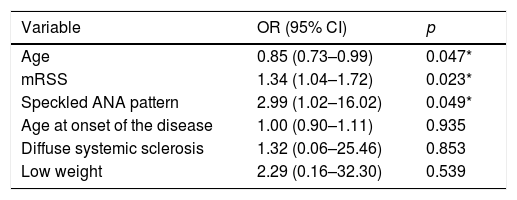
For the multivariate analysis of predictors of lung disease based on FVC, the variables showing a significant association in the univariate analysis were included in a model; these variables were: age, age of onset of the disease, systemic sclerosis subtype, low weight, mRSS and speckled ANA pattern. The independently ILD-associated variables were age (OR 0.85; 95% CI 0.73–0.99; p=0.047), the mRSS score (OR 1.34; 95% CI 1.04–1.72, p=0.023) and the speckled ANA pattern (OR 25.99; 95% CI 1.02–16.02; p=0.049) (Table 3).
Multivariate analysis of variables predictive of lung disease based on forced vital capacity.
| Variable | OR (95% CI) | p |
|---|---|---|
| Age | 0.85 (0.73–0.99) | 0.047* |
| mRSS | 1.34 (1.04–1.72) | 0.023* |
| Speckled ANA pattern | 2.99 (1.02–16.02) | 0.049* |
| Age at onset of the disease | 1.00 (0.90–1.11) | 0.935 |
| Diffuse systemic sclerosis | 1.32 (0.06–25.46) | 0.853 |
| Low weight | 2.29 (0.16–32.30) | 0.539 |
ANA: antinuclear antibodies; CI: confidence interval; mRSS: modified Rodnan skin score; OR: odds ratio.
Multivariate logistic regression was used in the analysis.
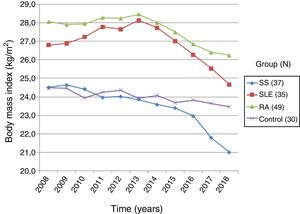
Based on the data collected from the medical record, the changes in BMI over the last 10 years in patients with systemic and controls were recorded. The group of patients with systemic sclerosis (n=37) had an initial BMI of 24.50±4.65kg/m2 and a final BMI of 21.00±4.43kg/m2, showing a 14.3% BMI reduction, with a statistically significant difference (p=0.0000). The group of patients with RA (n=49) had an initial and final BMI of 28.05±4.58kg/m2 and 26.23±5.32kg/m2, respectively, with a BMI loss of 6.5%, which was statistically significant (p=0.000). Then the group of patients with SLE (n=35) showed an initial BMI of 26.79±4.94kg/m2 and a final BMI of 24.65±4.83kg/m2, with 7.9% BMI loss that was statistically significant (p=0.0005). Finally, the group of healthy controls (n=30) had an initial BMI of 24.47±3.08kg/m2, and a final BMI of 23.46±3.08kg/m2; the percentage BMI loss was 4.1%, with no statistical significance (p=0.1327). The comparative analysis of the BMI loss in patients with systemic sclerosis versus the other groups showed a significant difference with SLE (p=0.0025), RA (p=0.0000), and the group of healthy subjects (p=0.0331) (Fig. 4).
DiscussionSystemic sclerosis is an autoimmune disease which due its low prevalence in the population29 apparently has received less attention in terms of research, as compared to other more frequent inflammatory autoimmune conditions, such as systemic lupus erythematous and RA. The available epidemiological studies on systemic sclerosis conducted in Latin America also indicate a low incidence in the continent.30,31 There are no demographic studies available in Venezuela on this population. However, over the last few years, significant progress has been made in the knowledge of the various immunopathogenic mechanisms that contribute to the development of the disease,32,33 which may therefore promote the research interest in systemic sclerosis.
According to the figures from the National Center of Rheumatic Diseases in 2011, the incidence of systemic sclerosis in Venezuela was estimated at about one case per million inhabitants (unpublished data), which certainly reflects under-registration, since these are data collected from only one center. With regards to the demographic characteristics, the study showed a prevalence of females and an age of onset of the disease similar to that reported in Caucasian patients,34,35 which indicates a late onset of the disease, as compared to Afro-Americans or individuals of Afro-American descent.29,34–36 The level of education of the patients in our study was low; two thirds of them said not having completed their school education, which was reflected in the socio-economic status – two thirds of the group belonged to levels IV and V of the Graffar Méndez-Castellanos scale. These findings, similar to the findings in other epidemiological studies,37 have implications in terms of the patients’ awareness of the disease, seeking early medical care, treatment compliance, and ensuring regular medical follow-up.38
Among the clinical manifestations of patients, Raynaud's phenomenon was the most frequent, which is consistent with the reports on clinical and experimental systemic sclerosis trials.39,40 The prevalence of this sign and the fact that it precedes by years the onset of the other clinical manifestations of the disease, stress the importance of microvascular endothelial damage as a primary event in the pathogenesis of the disease.41,42 In our study, the distribution of the various clinical characteristics among the limited and diffuse systemic sclerosis subtypes was homogeneous, with the exception of dyspnea, the lung function tests, and the mRSS score, which showed increased visceral lung involvement and severity of the cutaneous fibrosis in diffuse systemic sclerosis; this finding has been observed in various systemic sclerosis cohort studies.43 This shows that Hispanic patients with diffuse systemic sclerosis also present with respiratory and physical dysfunction, and hence will have a shorter life expectancy associated to this subtype.43–45
The study showed a direct correlation between BMI and FVC, though with no statistical significance. However, when the BMI was analyzed by groups, based on the WHO criteria, it was evident that low body weight (IMC <18.5kg/m2) is a risk factor for the development of ILD, since the FVC levels were lower in this group. In other words, malnourished patients with systemic sclerosis have a higher frequency of ILD, as compared to patients with BMI levels equal or higher than 18.5kg/m2. This finding is consistent with the study by Caimmi et al.46, which also proved that muscle mass loss was not involved in FVC dysfunction. So, under the premise that fat has a direct influence on BMI, and probably to a larger extent than muscle mass, this could explain the impact of low body weight on the predisposition to develop ILD in our patients with systemic sclerosis. Therefore, adipose tissue – through antifibrotic lipid mediators such as adiponectin,47 could represent a protective factor on the lung parenchyma, and hence on the pulmonary function.9,14 Some studies support the correlation between this group of mediators and BMI.48,49 So the repair, angiogenic, anti-inflammatory characteristics of the release of antifibrotic cytokines attributed to adipose tissue,17,50 could have an impact not just on the integument, but probably also on the respiratory cell units.14,16,17
The degree of skin fibrosis in systemic sclerosis is associated with the severity of the disease and with increased visceral involvement.51 Our study reported that high mRSS scores and the diffuse subtype had a negative influence on FVC levels, an important marker for ILD and the prognosis of short term survival in systemic sclerosis.52 In this regard, our results are consistent with other studies of the disease.53 Furthermore, since systemic sclerosis is a systemic connective tissue disease, the association observed between low weight and FVC could account for a substantial loss of non-functional fibrotic tissue.14 It is then suggested that just as it is the case in mice, the lipid nature of the lung parenchyma16 makes it susceptible to be replaced by myofibroblasts.14 A study measuring the cutaneous adipose mass index and the level of fibrosis in lung tissue samples would enable a more direct examination of this potential association. Additionally, other study outcomes indicated an average body mass loss of 14.3%, and this was correlated with the low FVC. The association between the loss of adipose mass and the fibrotic process has been observed in various studies in experimental murine models of systemic sclerosis, in which the loss of adipose tissue preceded the development of fibrosis.8,15 Our study showed that the extend of the skin involvement measured with mRSS was associated to the development of ILD. Hence, mRSS could be a clinical tool to indirectly assess the level of adipose atrophy and its replacement by fibrotic tissue in the subcutaneous tissue, and to predict visceral lung disease. The reproducibility, the interobserver consistency, the feasibility and the correlation with other tools such as ultrasound,20 make mRSS an extremely valuable instrument for prognosis and patient follow-up.
The 6-minute walk shall be part of the comprehensive assessment of patients with systemic sclerosis.54 The 6-minute walk evaluates different biomechanical, organic and body composition factors, which accounts for its diagnostic and predictive ability.55 In our study, the BMI had a negative influence on the number of meters walked in the 6MW, and although the difference was not significant, studies by Sanges et al.56 and Deuschle et al.28 showed a strong association. This may be due to the fact that in these studies the average age and BMI were higher and these factors are associated with reduced physical capacity.57,58 However, the results of the 6MW in systemic sclerosis shall be interpreted with caution due to multiple factors that may alter the test results.56 Thus, a higher prevalence of pulmonary arterial pressure, ILD, or musculoskeletal manifestations in varying degrees, are confounding factors. Moreover, our results indicated that the severity of the cutaneous fibrosis measured with mRSS and the “salt-and-pepper” skin lesions are negatively correlated with the 6MW. This is explained by the association described between mRSS and such lesions leading to severe fibrosis, and are related to joint contractures59 that may hinder locomotion.
There are multiple risk factors identified for the development of ILD in systemic sclerosis.3 In our study, mRSS and the speckled ANA pattern were independent risk factors associated with lung disease. With regards to mRSS, there are studies supporting its association with ILD.60,61 Similarly, Ashmore et al.44 have reported the relationship existing between speckled ANA pattern and ILD, which may be because the antigen specificity of this type of pattern is 20 a 60% against topoisomerase I.2,62 Therefore, in both cases, mRSS and the speckled ANA pattern could indirectly be associated with diffuse systemic sclerosis. Age however was a protective factor for the development of ILD. This may be due to the fact that a late onset, and a longer duration of the disease, are characteristics of patients with limited systemic sclerosis, in whom pulmonary involvement is less frequent.53,63
The amount of weight loss evidenced during a 10-year follow-up in patients with systemic sclerosis was more significant when compared against other inflammatory diseases and against a healthy control group. The greater weight loss of patients with systemic sclerosis may be explained by the replacement of adipose mass by fibrotic tissue affecting integuments and potentially other visceral tissues as well, such as the GI tract which compromises intestinal absorption.64 Additionally, patients present with muscle atrophy due to joint and integument involvement resulting in contractures of the extremities that progressively limit the patient's mobility and promote sarcopenia.27 Finally, the combination of integumentary damage and its cosmetic implications and visceral disease, frequently result in major depressive disorder, dysthymia and anxiety disorder, further compromising the patient's nutrition.64–66 The combination of all of these manifestations contributes to a phenotype typical of systemic sclerosis, which differentiates the condition from the rest of the inflammatory autoimmune diseases, and is characterized by wasting of the visible body fat reserves and, in extreme cases, may lead to cachexia. Consequently, recent studies based on the stimulation of the proliferation of fatty tissue,6,14,19,67,68 blocking of the adipocyte-myofibroblast transition6,7,15,69 and therapies based on adipose tissue transplantation,70–72 have opened a potential research field for the development of new therapies for the disease.
Our results should be considered preliminary results because of the small sample size since only one center was involved; there was a bias toward lower socioeconomic level patients, and there were no histological studies of the skin and the lung. The advantages of the study include a homogeneous population in terms of ethnicity, and as far as we know, this is the first study of this type to be conducted in the country, and probably in Latin America.
ConclusionThe study suggests that age, low body weight, high mRSS scores and the specked ANA patter are all risk factors for the development of ILD in patients with systemic sclerosis. ILD may be conditioned by significant adipose tissue wasting and its replacement by fibrotic tissue, both in the skin and in the lungs. The extent of cutaneous fibrosis of patients with diffuse systemic sclerosis and, consequently, the higher mRSS score, are also predictors of ILD. mRSS is a clinical tool able to predict visceral lung disease in systemic sclerosis. There is a need to conduct multicenter studies with a larger number of patients, in order to establish the role of body mass in the pathogenesis of systemic sclerosis through specific body composition studies and its correlation with lung disease in systemic sclerosis. This knowledge will allow for monitoring the progress of the disease using BMI and the fat composition index as predictors of lung disease in our patients.
Conflict of interestThe author has no conflicts of interest to disclose.
Please cite this article as: Rivas-Vargas D. Análisis del índice masa corporal y factores de riesgo de neumopatía intersticial en pacientes con esclerosis sistémica. Rev Colomb Reumatol. 2020;27:9–19.